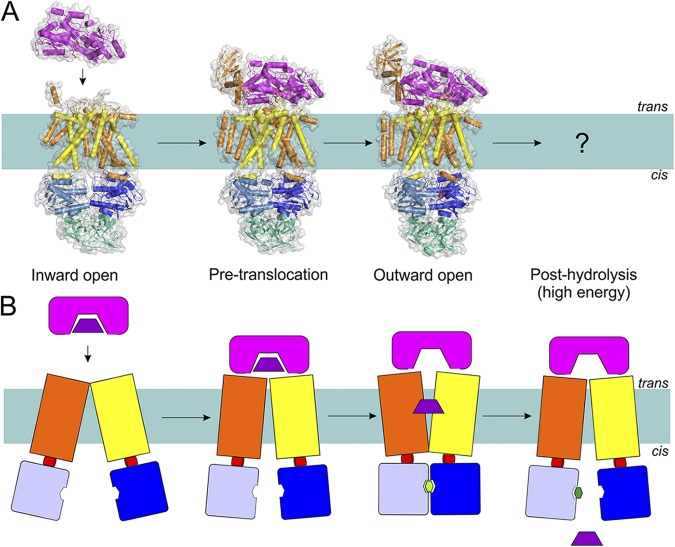
Figure 7.
The transport mechanism of Type I importers (exemplified by MalEFGK2) based on the available structures (A) and in schematic representation (B). Coloring is as in Fig. 1. Structures have been determined for the inward-facing, pre-translocation, and outward-facing conformations (Protein Data Bank accession nos.: 4JBW, 4KHZ, and 4KI0; see Table 1 for details of all available structures of ABC transporters). Substrate-loaded MalE docks onto the resting MalFGK2 transporter. This brings the NBDs closer to each other, allows ATP binding, and consequently causes MalK closure and TMD reorientation (outward-facing), leading to MBP opening. The released substrate diffuses toward the binding site in TMD, followed by ATP hydrolysis and resetting of the transporter into the inward-facing state.