Figure 1. Biological clocks organize glucose metabolism.
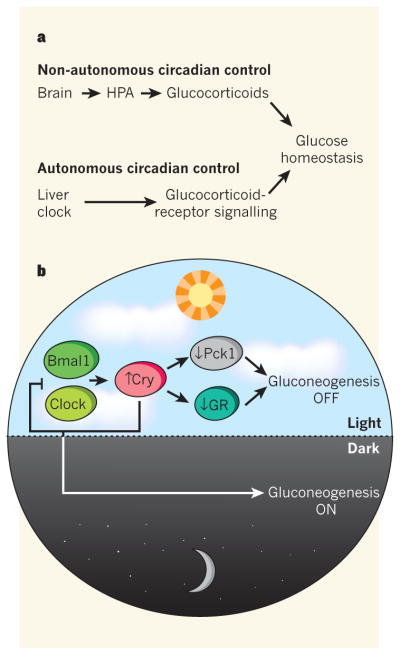
a, Glucose homeostasis is regulated by the interplay between non-autonomous circadian control — which affects glucocorticoid hormone levels through the brain and the hypothalamic–pituitary–adrenal (HPA) axis — and autonomous oscillatory regulation of these hormones’ signalling in peripheral tissues such as the liver. Lamia et al.4 report that the clock-repressor protein Cry plays a central part at both of these levels. b, In the liver, the clock-activator proteins Clock and Bmal1 increase Cry transcription, which downregulates gluconeogenesis both by interacting with — and so inhibiting — glucocorticoid receptors (GRs), and by reducing the expression of the Pck1 enzyme. As is typical of the clock machinery, high levels of Cry also downregulate Clock and Bmal1 transcription in a negative feedback loop, thus reducing its own levels and so promoting gluconeogenesis during the dark phase of the light–dark cycle.
