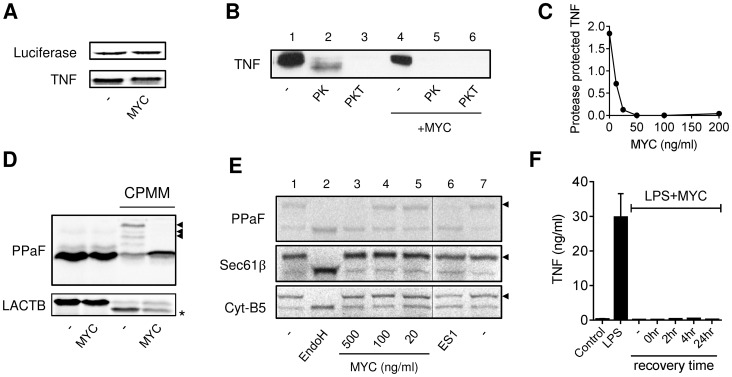
Figure 4. Mycolactone inhibits co-translational translocation of proteins into the ER via a mechanism that does not disrupt the structural integrity of the ER.
A–E. In vitro translation (IVT) reactions of different capped transcripts were performed as described +/− mycolactone (MYC; 200 ng/ml unless indicated otherwise), Eeyarestatin 1 (ES1; 250 µM) or DMSO (-; 0.02%). Data representative of 3 independent experiments. A. IVT of luciferase and TNF mRNAs detected by 35S incorporation and Western blot (∼26 kDa) respectively. B. IVT of TNF mRNA was performed in the presence of semi-permeabilised RAW264.7 cells then incubated with no addition (-), or Proteinase K (PK) +/−0.1% Triton-x-100 (PKT) for 1 hr at 4°C before stopping the reaction. C. Dose dependence of loss of the PK protected band. Signal intensity was quantified by ImageJ analysis of non-saturated blots and the protected band (PK) was normalised to total pro-TNF (-). D. IVT of prepro-α Factor (PPAF) and β-lactamase (LACTB) mRNAs in the absence or presence of canine pancreatic microsomal membranes (CPMM). Black arrowhead, glycosylated forms of α Factor; *, signal peptide-cleaved LACTB. E. IVT of PPAF, Sec61β and cytochrome B5 (Cyt-B5) mRNAs in the presence CPMM. After labelling with 35S as described, membranes were isolated by centrifugation through a sucrose cushion [31]. Endoglycosidase H (EndoH) is used to confirm glycosylation of proteins (black arrowhead), the thin line represents where an empty lane was removed from the image for ease of interpretation. F. RAW264.7 cells were incubated +/−125 ng/ml mycolactone (MYC), washed and incubated without mycolactone for various periods (recovery time) before stimulating with LPS for 4 hrs. Supernatant TNF levels were measured by ELISA (mean±SEM of triplicate assays).