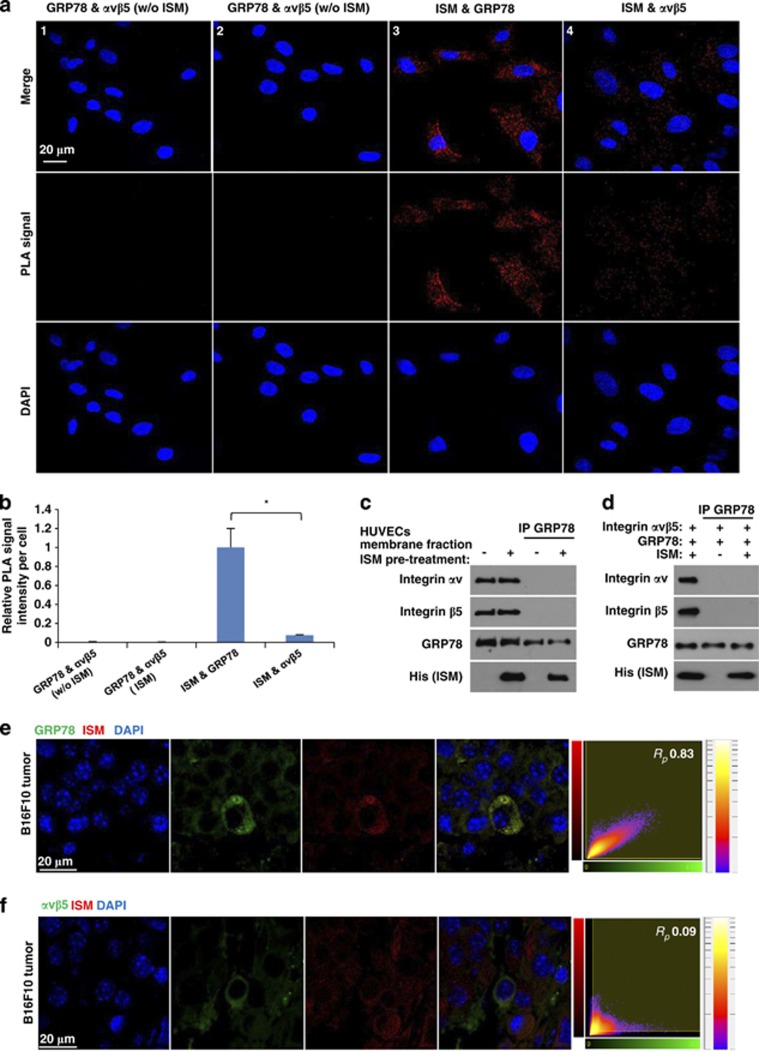
Figure 6.
GRP78 and αvβ5 integrin function as independent cell-surface receptor for ISM on ECs and mediating apoptosis through distinct signaling pathways. (a and b) GRP78 and αvβ5 integrin do not interact in HUVECs. (a) Determination of the interaction between GRP78 and αvβ5 integrin, ISM and GRP78, and ISM and integrin αvβ5 by Duolink in situ PLA. PLA signals (red) indicate two proteins in close proximity (nucleus). GRP78 and αvβ5 integrin did not exist in close proximity in HUVECs in the absence (1) or presence (2) of ISM; (3) ISM interacts with GRP78 on the cell surface and in the cytoplasm. (4) ISM interacts with αvβ5 integrin in HUVECs. (b) Quantification of PLA signals. PLA signal intensities of three microscopic fields from each group were calculated by ImageJ and then normalized by cell number. All photos were obtained under the same exposure time and laser power. *P<0.05, n=3. Error bars denote S.E.M. (c) GRP78 and αvβ5 integrin did not bind to each other on the surface of HUVECs with or without ISM pretreatment. (d) Recombinant GRP78 and αvβ5 integrin do not bind to each other with or without the presence of ISM. (e and f) Systemically delivered rISM is co-localized with GRP78, but not with αvβ5 integrin, in B16F10 tumor cells. Paraffin sections of rISM-treated B16F10 tumors were probed for both GRP78 and ISM through IF using anti-his (red) and anti-GRP78 or αvβ5 integrin (green) double staining. Nuclei were counterstained by DAPI (blue). Representative photos are shown. Quantitative measurements of the Pearson's correlation coefficient (Rp) between red and green fluorescent labeled channels were indicated in the far-right