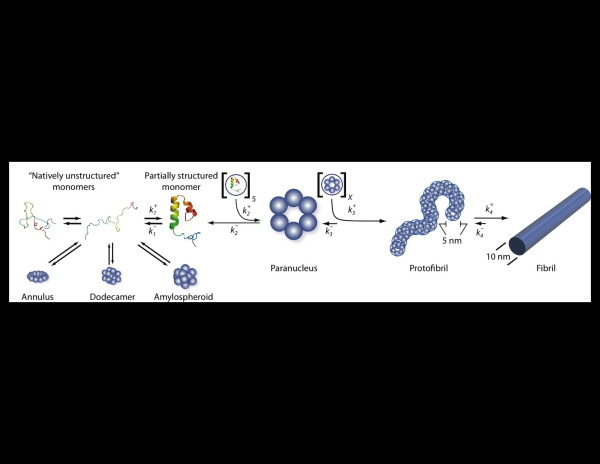
Figure 1.
Amyloid β-protein assembly. Amyloid β-protein appears to be an intrinsically disordered protein and thus exists in the monomer state as an equilibrium mixture of many conformers. ‘On-pathway’ fibril assembly requires the formation of a partially folded monomer that self-associates to form a nucleus for fibril elongation, a paranucleus, which in this case contains six monomers. Nucleation of monomer folding is a process distinct from fibril nucleation. Fibril nucleation is unfavorable kinetically (), which explains the lag phase of fibrillogenesis experiments, a period during which no fibril formation is apparent. Paranuclei self-associate readily () to form protofibrils, which are relatively narrow (approximately 5 nm), short (<150 nm), flexible structures. These protofibrils comprise a significant but finite number X of paranuclei. Maturation of protofibrils, through a process that is poorly understood but is favorable (), yields classical amyloid-type fibrils (approximately 10 nm diameter, indeterminate (but often >1 μm) length). Other assembly pathways produce a variety of oligomers, including annular, pore-like structures; globular dodecameric (and higher-order) structures; and amylospheroids. Adapted from [27].