Table 3.
In vivo studies in rodent cancer models treated with vitamin D analogs (intraperitoneal i.p.; subcutaneous s.c.) published between 2007 and 2013.
| Cancer type | Dosage vitamin D analog | Duration of treatment | Outcome | References |
|---|---|---|---|---|
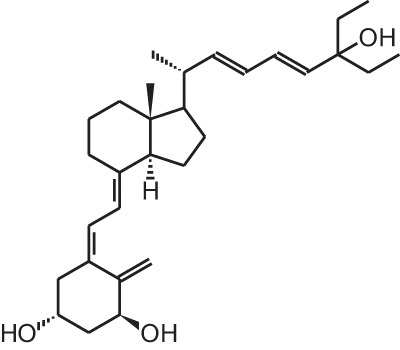
Seocalcitol (22,24-diene-24,26,27-trishomo-1 α,25(OH)2D3)
| ||||
| Chemically-induced breast cancer | 7 μg/kg/week | Approximately 80 days | Decreased tumor burden and volume | Liska et al., 2012 |
| Chemically-induced breast cancer | Oral, 7 μg/kg/week | 116 or 156 days | Prolonged latency of mammary gland tumors | Macejova et al., 2011 |
| Prostate cancer xenograft | i.p., 0.5 μg/kg every other day | 45 days | Reversal of growth stimulatory effects of PTHrP | Bhatia et al., 2009 |
| Hepatocellular carcinoma xenograft | Oral and i.p., 0.02/0.1/0.5 μg/kg/d | Approximately 21 days | Inhibition of tumor growth | Ghous et al., 2008 |
| Inoculation with mice breast cancer cells | i.p., 20 ng 3×/week | 6 weeks | Inhibition of tumor growth, no inhibition of tumor angiogenesis | Valrance et al., 2007 |
HY-11 (2-amino-3-deoxy-19-nor-22-ene-26-dihomo-27-dihomo-25(OH)D3) 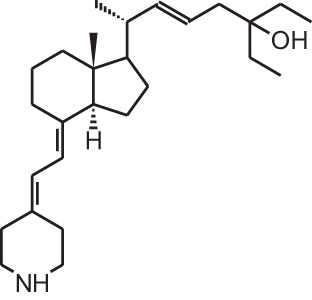
| ||||
| Inoculation with mice leukemia cells | i.p., 10−5 M/d | 26 days | 50% increase in survival | Yoon et al., 2008 |
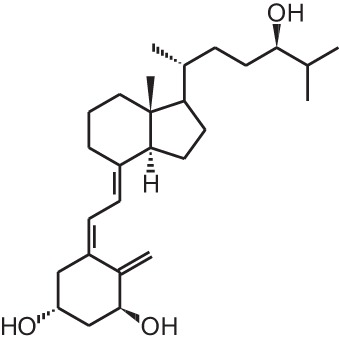
Tacalcitol (1 α,24(R)(OH)2D3)
| ||||
| Inoculation with mice colorectal cancer cells | Different concentrations s.c. (3 or 5×/week) or oral (3×/week) in combination with different concentrations of 5-fluorouracil | Variable duration | 1 μg/kg/d optimal dose + prolongation of life span of mice (synergistic effect when combined with chemotherapy) | Milczarek et al., 2013a |
| Inoculation with mice or human colorectal cancer cells | s.c., Different concentrations in combination with different concentraties of irinotecan or oxaliplatin | Variable duration | Under certain experimental conditions vitamin D analogs and chemotherapy can work synergistically | Milczarek et al., 2013b |
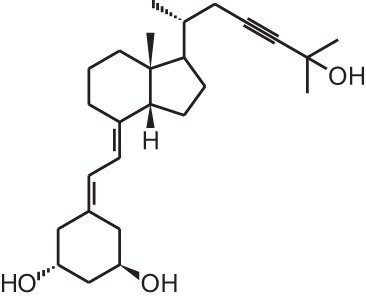
Inecalcitol (19-nor-14-epi-23-yne-1 α,25(OH)2D3)
| ||||
| Squamous cell carcinoma xenograft | i.p., 80/160/320 μg/mouse/d | 3 days | Inhibition of tumor growth, increased apoptosis, decreased proliferation | Ma et al., 2013 |
| Prostate cancer xenograft | i.p., 1300 μg/kg 3×/week | 42 days | Delay of tumor growth, 50% decrease in tumor weight and decreased tumor vascularity | Okamoto et al., 2012 |
TX527 (19-nor-14,20-bisepi-23-yne-1 α,25(OH)2D3) 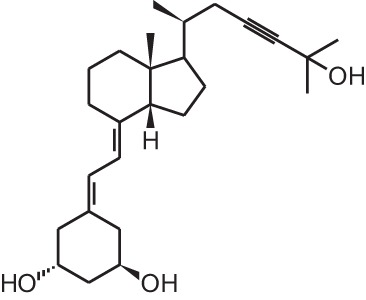
| ||||
| Kaposi sarcoma xenograft | i.p., 10 μg/kg/d | 4 days | Decreased tumor progression | Gonzalez-Pardo et al., 2010 |
Paricalcitol (19-nor-1 α,25(OH)2D2)
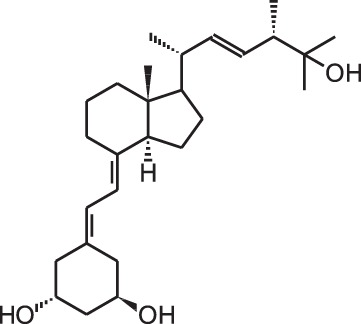
| ||||
| Gastric cancer xenograft | s.c., 100 ng/d 3×/week | 4 weeks | Lower tumor volume, reduced growth of intraperitoneal metastasis | Park et al., 2012 |
| Pancreatic cancer xenograft | s.c., 2.5 μg/kg 3×/week | Variable duration | Inhibition of tumor growth | Schwartz et al., 2008 |
Doxercalciferol (1 α (OH)D2) 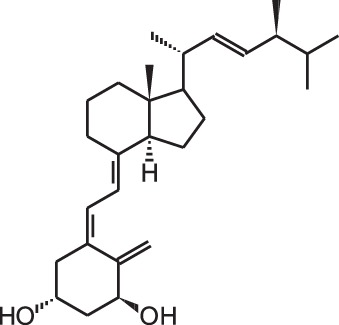
| ||||
| Neuroblastoma xenograft | Oral, 0.15/0.3 μg/d | 5 weeks | Lower tumor volume | van Ginkel et al., 2007 |
Maxacalcitol (22oxa-1 α,25(OH)2D3)
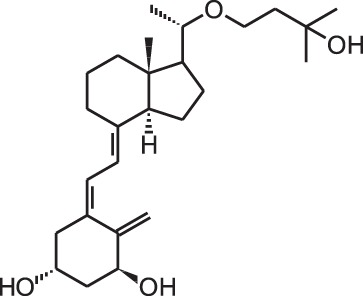
| ||||
| Cholangiocarcinoma xenograft | i.p., 15 μg/kg/d | 17 days | Inhibition of tumor growth | Seubwai et al., 2010 |
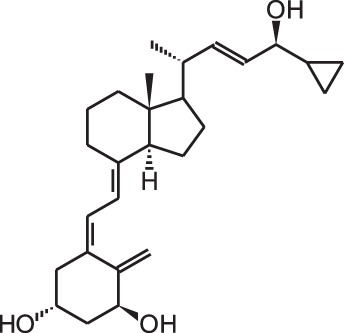
Calcipotriol (22-ene-26,27-dehydro-1 α,25(OH)2D3)
| ||||
| UV-induced non-melanoma skin cancer | Topical application in combination with diclofenac and difluoromethylornithine | 17 weeks | Decrease in number and area of tumors when combined with diclofenac | Pommergaard et al., 2013 |
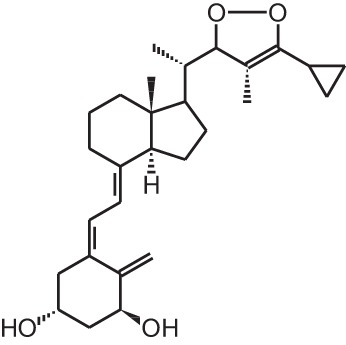
BGP-13 (1R, 3S, 5Z)-5-((8E)-2-((3R)-3-((2R, 3S)-3-(5-cyclopropyl-3H-1,2-dioxol-3-yl)-2-ethyl-3-methylcyclohexylidene) ethylidene)-4-methylenecyclohexane-1,3-diol)
| ||||
| Colorectal cancer xenograft | i.p., 2 μg/kg every 2 days | 8 days | Inhibition of tumor growth | Berkovich et al., 2013 |
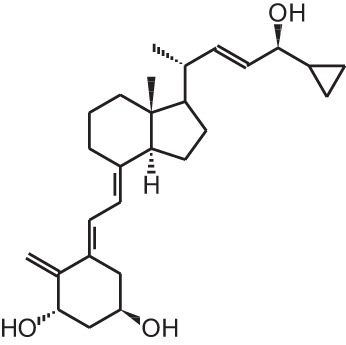
PRI-2205 ((5E,7E)-22-ene-26,27-dehydro-1 α, 25(OH)2D3)
| ||||
| Breast and lung cancer xenograft | s.c., 10 μg/kg 2 or 3×/week + cytostatics | 18–21 days | Combination of analogs with low doses of cytostatics is not effective | Wietrzyk et al., 2007 |
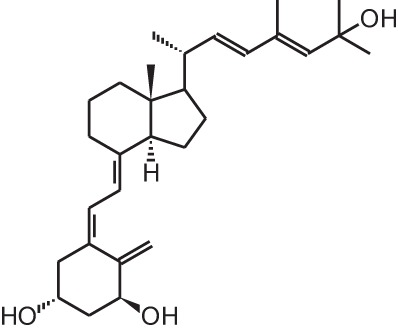
PRI-1906 ((24E)-(1S)-24-dehydro-24a-homo-1α,25(OH)2D3)
| ||||
| Inoculation with mice breast cancer cells | s.c., 0.1 or 1 μg/kg/d | 9 or 11 days | No effects | Wietrzyk et al., 2008 |
BXL-01-0126 (20R-(4-hydroxy-4-methylpentyl)-23-yne-26,27-hexafluoro-19-nor-1 α, 25(OH)2D3)
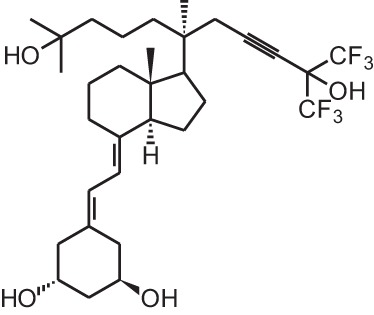
| ||||
| Acute myeloid leukemia xenograft | i.p., 0.0625 μg | 1 injection | Cathelicidin antimicrobial peptide present in systemic circulation | Okamoto et al., 2014 |
BXL0124 (20R-21(3-hydroxy-3-deuteromethyl-4,4,4-trideuterobutyl)-23-yne-26,27-hexafluoro-1 α, 25(OH)2D3)
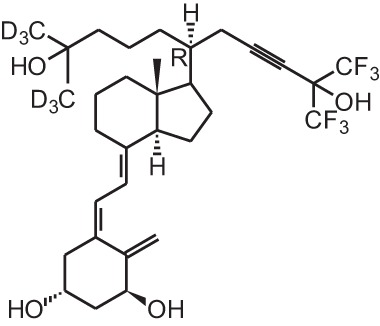
| ||||
| Breast cancer xenograft | i.p., 0.1 μg/kg or oral 0.03/0.1 μg/kg 6 days/week | 5 weeks | Suppressed tumor growth | So et al., 2011 |
| Transgenic mice with breast cancer (ErbB2/Her-2/neu overexpressing tumors) | i.p., 0.3 μg/kg 3×/week | Approximately 38 weeks | Inhibition of tumor growth and regulation of ErbB2/AKT/ERK pathway | Lee et al., 2010 |
Gemini0097 (20R-21(3-trideuteromethyl-3-hydroxy-4,4,4-trideuterobutyl)-23-yne-26,27-hexafluoro-19-nor-1 α, 25(OH)2D3)
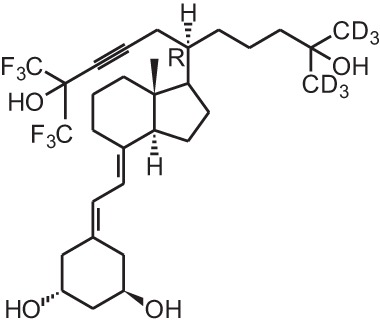
| ||||
| ER-negative breast cancer xenograft | i.p., 0.1 μg/kg/d | 9 weeks | Suppressed tumor growth | Lee et al., 2008 |
| Chemically-induced breast cancer (ER positive) | i.p., 0.03/0.1/0.3 μg/kg 5days/week | 9 weeks | Inhibition of tumor burden | |
MART-10 (19-nor-2 α-(3-hydroxypropyl)-1 α, 25(OH)2D3)
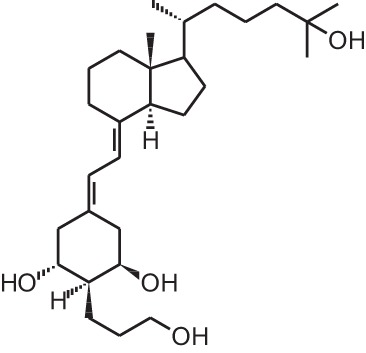
| ||||
| Pancreatic cancer xenograft | i.p., 0.3 μg/kg 2×/week | 3 weeks | Inhibition of tumor growth | Chiang et al., 2013 |
1 α,25(OH)2D3-3-bromoacetate
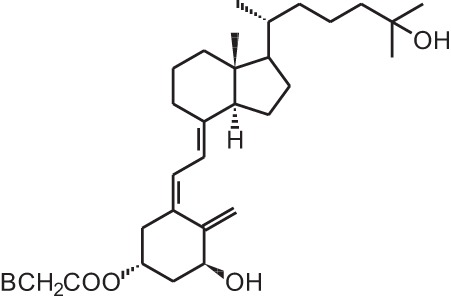
| ||||
| Kidney cancer xenograft | i.p., 0.75 μg/kg every third day | 80 days | Reduced tumor size and increased apoptosis | Lambert et al., 2010 |
Ro26-2198 (16,23Z-diene-26,27-F6-19-nor-1 α, 25(OH)2D3)
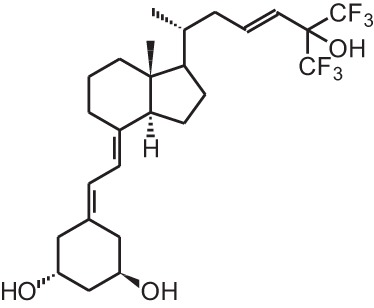
| ||||
| Chemically-induced colorectal cancer | 0.01 μg/kg/d via mini-osmotic pump | 28 days | Inhibition of dysplasia progression and inhibition of proliferation and pro-inflammatory signals | Fichera et al., 2007 |
For studies preceding 2007 the reader is referred to other reviews (Eelen et al., 2007).
