Abstract
DNA methylation (5-methylcytosine, 5mC) plays critical biological functions in mammals and plants as a vital epigenetic marker. The Ten-Eleven translocation dioxygenases (TET1, 2, and 3) have been found to oxidize 5mC to 5-hydroxymethylcytosine (5hmC) and then to 5-formylcytosine (5fC) and 5-carboxylcytosine (5caC) in mammalian cells. We report herein three mushroom TET homologues from Coprinopsis cinerea that can mediate 5mC oxidation. Specifically, one homologue (CC1G_05589, CcTET) shows similar activity to its mammalian TET homologues. Biochemically, CcTET actively converts 5mC to 5hmC, 5fC, and 5caC under natural conditions (pH 7.0). Interestingly, CcTET also converts the majority of 5mC to 5fC under slightly acidic (pH 5.8) and neutral conditions. Kinetics analyses of the oxidation by CcTET under neutral conditions indicate that conversion of 5mC to 5hmC and 5hmC to 5fC are faster than that of 5fC to 5caC, respectively. Our results provide an example of a TET homologue in a non-mammalian organism that exhibits full 5mC-to-5caC oxidation activity and a slight preference to producing 5fC. The preferential accumulation of 5fC in the in vitro oxidation reactions under both neutral and acidic conditions may have biological implications for 5mC oxidation in fungi species.
DNA methylation (5mC) at the 5-position of cytosine occurs mostly on CpG dinucleotides and is widespread among eukaryotic species.1,2 This methylation plays broad and critical biological functions especially in transcriptional regulation, gene silencing, and genome reprogramming2−4 and exhibits dynamic distribution patterns at different developmental stages in eukaryotic genomes.5−7 Abnormal 5mC distributions have been linked to human diseases such as cancer.8
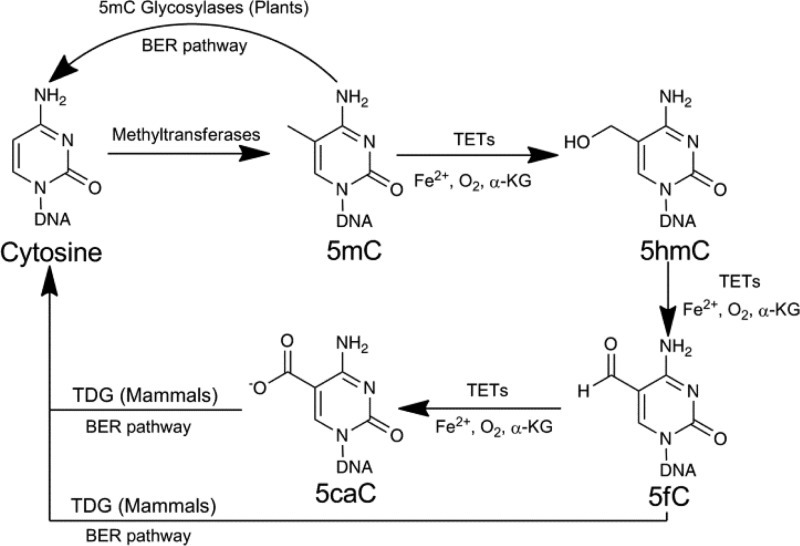
Cytosine methylation is generated and maintained by a family of methyltransferases (DNMTs).1,8 However, demethylation pathways vary among different species and are poorly understood. For instance, plants employ the Demeter family of 5mC glycosylases to excise the methylated cytosine base and replace it with an unmethylated base through the DNA base-excision repair pathway (BER).9−14 Lacking in 5mC glycosylases, mammals must make use of different pathways. In 2009, a family of Ten-Eleven translocation dioxygenases (TET1, 2, and 3) was discovered, which led to a new paradigm of active DNA demethylation in mammals.15 These proteins belong to the iron(II)/α-ketoglutarate (α-KG)-dependent dioxygenase family and contain a highly conserved C-terminal DNA-binding and catalytic domain (CD domain) that shares a similar, core catalytic domain with AlkB family proteins and HIF prolyl-hydroxylases.15−20 TET proteins utilize oxygen, iron(II), and α-KG in order to oxidize the methyl group of 5mC stepwise to 5-hydroxymethylcytosine (5hmC),15,19 5-formylcytosine (5fC), and then 5-carboxylcytosine (5caC).21−24 The oxidation products of 5fC and 5caC are subsequently recognized and excised by mammalian thymine DNA glycosylase (TDG) and repaired back to unmethylated cytosine through the BER pathway (Figure 1).25−27 The 5hmC modification is relatively abundant in embryonic stem cells and neuronal cells,28−31 whereas cellular levels of 5fC and 5caC are much lower, presumably due to their constant removal through BER.9,21,25,27
Figure 1.
Mammalian cytosine methylation and the active demethylation pathway.
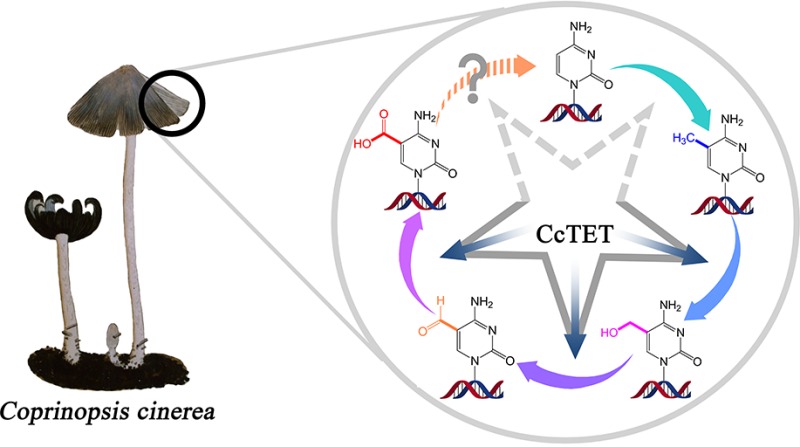
Other than plants, mammals and certain unicellular eukaryotes, fungi contain 1–4% 5mC of the total cytosines in the genome,32,33 but the roles of methylation in fungi remain incompletely understood. Through use of sequence profile searches, multiple TET/JBP homologues have been identified in several eukaryotes other than animals, including fungi, certain chlorophyte algae, stramenopile algae, the heterolobosean amoeboflagellate Naegleria.35,36 One of the eight TET/JBP homologues from Naegleria has been recently crystallized.34 In fungi the TET/JBP proteins are associated with a novel class of DNA transposons.35 Here we report a newly discovered TET enzyme from Coprinopsis cinerea (CC1G_05589, hereafter named and referred to as CcTET), which has full activity to convert 5mC to 5hmC, 5fC, and 5caC.
C. cinerea belongs to the mushroom family Psathyrellaceae and is considered an important model organism of multicellular fungi. Its genome sequence was published in 2010.36 From a total of 47 TET homologues in strain Okayama 7#130, we selected and expressed 12 in E. coli based on computational predictions.37,38 The his-tagged recombinant proteins were purified via Ni-NTA, MonoS, and Superdex75 Gel-filtration. We employed a 5mC-oxidation assay in vitro to monitor the activity (see Supporting Information). A 9/11-mer duplex DNA containing a 5mCpG site on the 9mer was employed as the substrate (sequences: 5′-GAC(5mC)GGAGT-3′, 5′-GACTCCGGTCT-3′). The oxidation reactions were assayed at 37 and 25 °C under pH 5.8, 7.0, and 8.0, respectively, and the oxidation products were subsequently digested and monitored by quantitative mass spectrometry (HPLC-MS/MS). Although multiple sequence alignment demonstrated high similarity, particularly within the catalytic domain, the activity assay revealed three candidate proteins: CC1G_05589, Genbank id: XP_001838108.2; CC1G_10221, XP_001835294.1; and CC1G_02065, XP_001834329.1, which possess 5mC oxidation activity (Figure S1). All other proteins were well-expressed and could be purified; the possibility remains that some of these other proteins could be active in vivo.
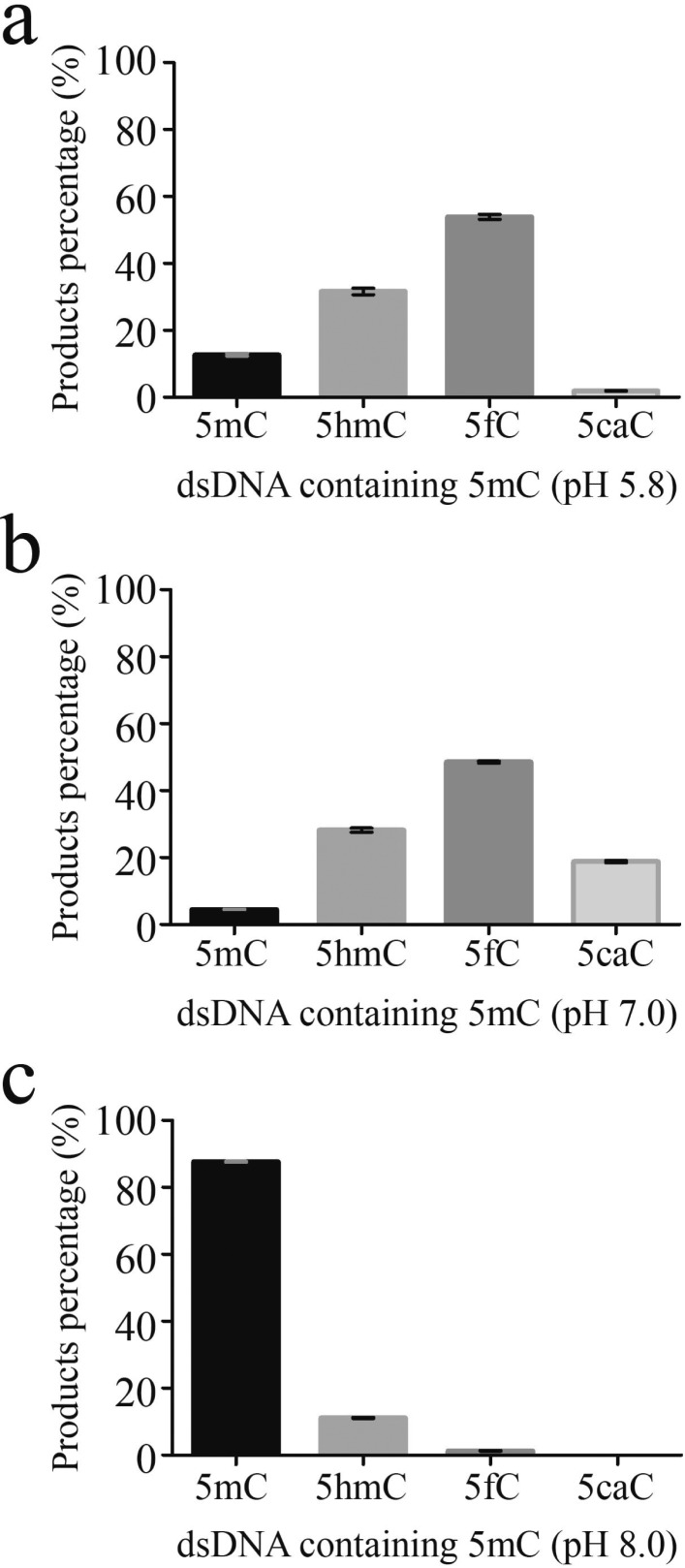
Among the three active proteins, CC1G_05589 exhibited the highest activity; the other two moderately convert 5mC to 5hmC and 5fC with almost no 5caC observed (Figure S2). We refer to CC1G_05589 as “CcTET” here and further characterized its enzymatic activity. CcTET has 430 amino acids and shares 10.7% identity (46/430 residues) and 33.7% similarity (145/430 residues) with human TET1 (Figures S3 and S4). Moreover, the catalytic domain of CcTET is highly similar to that of mammalian (human and mouse) TET/JBP family proteins with all the catalytic residues for iron- and α-KG-binding sites conserved (such as HxD···H···R; Figure S4). Unlike its mammalian TET homologues, CcTET exhibits weak activity under reported reaction conditions (pH 8.0).15,19,23,26,39 With an excess amount of protein (DNA:protein molar ratio of 1:2), treatment of CcTET with an 5mC-containing dsDNA produced only ∼11% 5hmC and ∼1.3% 5fC (Figure 2). Instead, CcTET exhibited much higher activity under neutral (pH 7.0) and slightly acidic conditions (pH 5.8) at 25 and 37 °C (Figures 2, S4, and S5). Under these conditions, CcTET generated ∼28% or ∼32% 5hmC and almost 2-fold more 5fC (∼49% or 54%) beginning with 5mC-containing dsDNA at 37 °C, respectively; ∼19% of 5caC can still be generated at neutral pH. Under slightly acidic conditions (pH 5.8), however, the reaction almost stalls at the 5fC stage, generating only ∼1.9% 5caC. Additionally, CcTET could also oxidize 5mC in single-stranded DNA (ssDNA) with similar oxidation activity and product contents as dsDNA (Figure S6).40 In contrast, the catalytic domain of mouse TET1 (mTET1) also exhibits higher activity at pH 5.8 and 7.0 than at pH 8.0 at 37 °C. mTET1 converts almost all 5mC to 5caC under slightly acidic and neutral conditions as well as under pH 8.0 with ATP in vitro (Figure S7).22,39 These biochemical data indicate that CcTET has full activity to convert 5mC to 5hmC, 5fC, and 5caC on both of single stranded and duplex DNA under neutral condition and is mostly inactive at pH 8.0 even with the presence of ATP in vitro (Figure S8). Notably, the accumulation of 5fC production by CcTET observed under both neutral and acidic reaction conditions may have interesting biological implications as 5fC accumulates in certain mammalian cells in vivo and could exhibit important functions.41,42 Interestingly, CcTET also exhibits weak thymine oxidation activity under pH 7.0 on A·T base pair in duplex DNA substrate and oxidizes thymine to 5-hydroxymethyluridine (5hmU) in our activity assay (Figure S9). This activity toward thymine is much lower than oxidation of 5mC. However, this is consistent with the recent prediction regarding existence of hypermodified thymines in fungi such as Coprinopsis38 and raises the possibility that ccTET, despite being in a distinct clade from JBP, has more JBP-like activity that mammalian TET.
Figure 2.
CcTET oxidation assays at 37 °C under pH 5.8, 7.0, and 8.0. The reaction mix contains 1 μM 5mC containing dsDNA, 2 μM CcTET, 50 mM Bis-Tris, pH 5.8 (or HEPES, pH 7.0 or 8.0), 75 μM Fe(NH4)2(SO4)2, 2 mM ascorbic acid, 1 mM α-KG, and 2.5 mM DTT for 30 min at 37 °C and then quenched by heating to 100 °C for 5 min and immediately cooled in an ice bath.
To further probe the intriguing 5mC oxidation activity, we also performed oxidation assays of CcTET on genomic DNA isolated from mouse embryonic stem cells (mESC) under neutral and slightly acidic conditions with similar results observed (Figures S10–S12). We then performed electrophoretic mobility shift assays (EMSA) against cytosine derivatives (C, 5mC, 5hmC, 5fC, and 5caC) contained in a 22mer dsDNA sequence (Figure S13). The results show that CcTET could bind to all these cytosine derivatives with very similar binding affinity (∼125 nM), suggesting that CcTET does not have noticeable selective preference to these modifications. It should be noted that the mammalian TET enzymes oxidize 5hmC all the way to 5caC in vitro with minimum 5fC observed (Figure S7).39 In the case of CcTET it is possible that 5fC bound in the active site of the enzyme cannot be effectively hydrolyzed to form hemiacetal, which could be critical for continued oxidation to 5caC.43
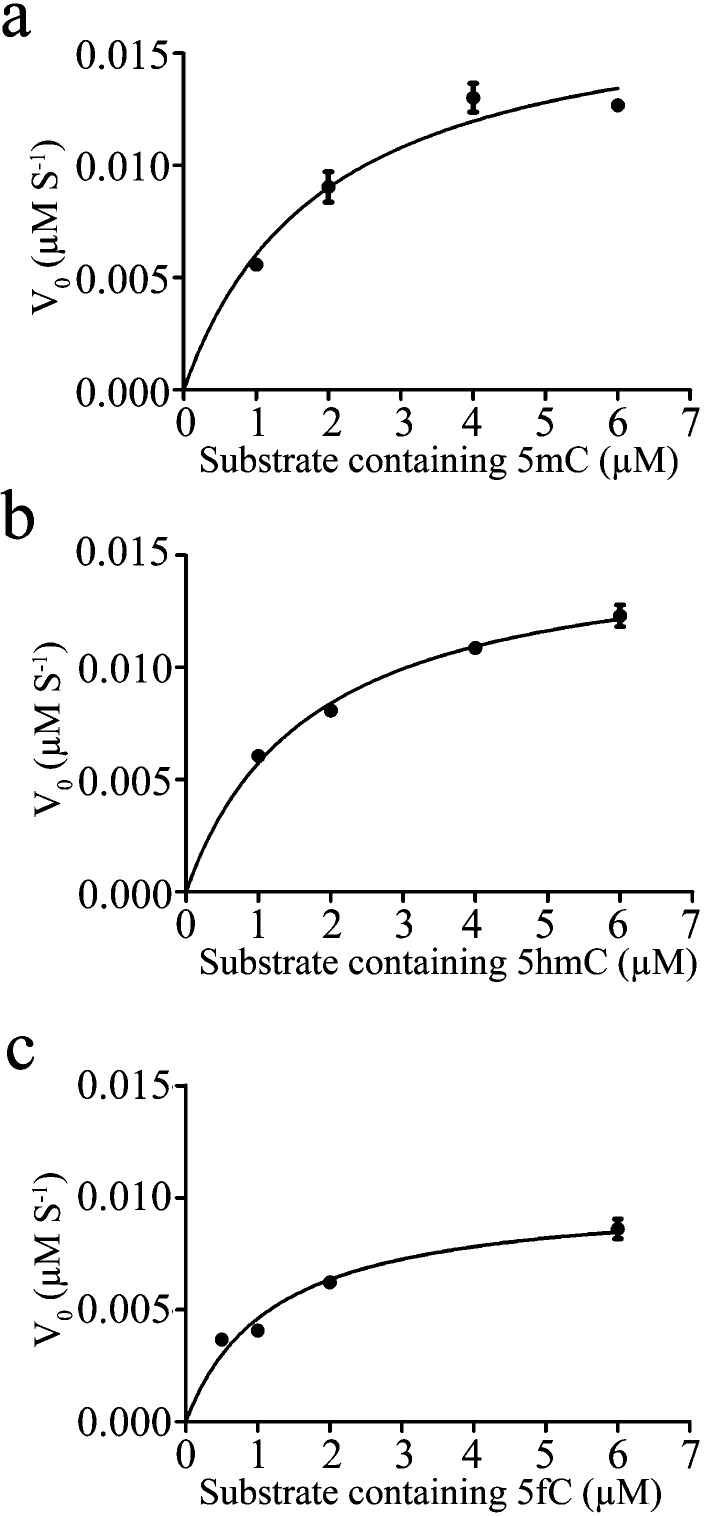
To further understand the catalytic mechanism of CcTET, we performed kinetic analyses on the reactions of 5mC to 5hmC, 5hmC to 5fC, and 5fC to 5caC at 37 °C using dsDNA under neutral conditions (pH 7.0), employing quantitative mass spectrometry. We first collected the reaction progress curve of substrate and fraction products (5hmC, 5fC, and 5caC) versus incubation times at 37 °C (Figure S14). Like mTET1,22 the CcTET-catalyzed reaction plateaus after ∼10 min of incubation; inactivation of CcTET at the reaction temperature (37 °C) is less likely the reason (Figure S15). The progress curve appears linear within 2.5 min, allowing for initial rates to be derived (Figure S16). We then performed kinetic study within 2.5 min at 37 °C and quenched the reactions at time points of 0, 30, 60, 90, 120, and 150 s. As shown in Figure 3 and Table S1, the oxidation activity of 5mC to 5hmC, 5hmC to 5fC, and 5fC to 5caC under pH 7.0 at 37 °C gradually decreases. Under these experimental conditions, the maximal rate of the oxidation of 5mC to 5hmC is slightly faster than that of 5hmC to 5fC, which is faster than that of the conversion of 5fC to 5caC. The half-maximal rate values (1/2 Vmax) of 5mC to 5hmC, 5hmC to 5fC, and 5fC to 5caC were obtained at similar substrate concentrations, suggesting that CcTET lacks binding preference to these modified cytosine bases, which is consistent with the EMSA results (Figure S13). The progress curves of 5mC to 5hmC and 5hmC to 5fC could be influenced by the subsequent oxidation step. However, the subsequent oxidation steps show slower reaction rates which may not significantly affect the kinetic analysis of the previous step (Figure S16).
Figure 3.
Michaelis–Menten’s plot of the kinetics of reactions of 5mC to 5hmC, 5hmC to 5fC, and 5fC to 5caC catalyzed by CcTET at 37 °C in pH 7.0 solution.
The discovery of CcTET and the other two homologues as well as the characterization may have several implications: (1) It suggests that fungal TET homologues can catalyze the oxidation of 5mC to further modified derivatives; these conversions might have significant biological functions as epigenetic marks or as intermediates in active demethylation in these fungi; (2) the fact that CcTET yields more 5fC over 5caC in vitro might suggest that different TET enzymes have distinctly tuned their active sites in order to generate different reaction products; this activity tuning perhaps arises from 5fC or its derivatives in genomic DNA playing specific roles in these fungi; and (3) mouse TET proteins have been shown to function as excellent tools to convert 5mC to either 5hmC (through in situ trapping) or 5caC in sequencing applications.39,44,45 The production of 5fC by CcTET under both neutral and acidic reaction conditions could be explored in order to develop new sequencing strategies for cytosine modifications. In addition, the molecular mechanism of CcTET-mediated oxidation can be compared with that of the mammalian mechanism in the future when structures of these enzymes become available. Importantly, these findings offer the first biochemical glimpses of the TET homologues, which display major lineage-specific expansions in several fungi and appear to be notable contributors to the genomic and epigenetic landscape of these organisms.35
Acknowledgments
This work is supported by National Institutes of Health HG006827, GM071440 (C.H.) and the intramural research grant of the DHHS, National Library of Medicine, NIH, USA (L.A. and L.M.I.). We thank S. F. Reichard for editing the manuscript.
Supporting Information Available
Experimental details and kinetic profiles. This material is available free of charge via the Internet at http://pubs.acs.org.
Author Contributions
∥ These authors contributed equally.
The authors declare no competing financial interest.
Funding Statement
National Institutes of Health, United States
Supplementary Material
References
- Law J. A.; Jacobsen S. E. Nat. Rev. Genet. 2010, 11, 204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu S. C.; Zhang Y. Nat. Rev. Mol. Cell Biol. 2010, 11, 607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhutani N.; Burns D. M.; Blau H. M. Cell 2011, 146, 866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klose R. J.; Bird A. P. Trends Biochem. Sci. 2006, 31, 89. [DOI] [PubMed] [Google Scholar]
- Weber M.; Schubeler D. Curr. Opin. Cell Biol. 2007, 19, 273. [DOI] [PubMed] [Google Scholar]
- Reik W.; Dean W.; Walter J. Science 2001, 293, 1089. [DOI] [PubMed] [Google Scholar]
- Sasaki H.; Matsui Y. Nat. Rev. Genet. 2008, 9, 129. [DOI] [PubMed] [Google Scholar]
- Robertson K. D.; Wolffe A. P. Nat. Rev. Genet. 2000, 1, 11. [DOI] [PubMed] [Google Scholar]
- Feng S.; Jacobsen S. E.; Reik W. Science 2010, 330, 622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suzuki M. M.; Bird A. Nat. Rev. Genet. 2008, 9, 465. [DOI] [PubMed] [Google Scholar]
- Gehring M.; Huh J. H.; Hsieh T. F.; Penterman J.; Choi Y.; Harada J. J.; Goldberg R. B.; Fischer R. L. Cell 2006, 124, 495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi Y.; Gehring M.; Johnson L.; Hannon M.; Harada J. J.; Goldberg R. B.; Jacobsen S. E.; Fischer R. L. Cell 2002, 110, 33. [DOI] [PubMed] [Google Scholar]
- Hsieh T. F.; Ibarra C. A.; Silva P.; Zemach A.; Eshed-Williams L.; Fischer R. L.; Zilberman D. Science 2009, 324, 1451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mok Y. G.; Uzawa R.; Lee J.; Weiner G. M.; Eichman B. F.; Fischer R. L.; Huh J. H. Proc. Natl. Acad Sci. U.S.A. 2010, 107, 19225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tahiliani M.; Koh K. P.; Shen Y.; Pastor W. A.; Bandukwala H.; Brudno Y.; Agarwal S.; Iyer L. M.; Liu D. R.; Aravind L.; Rao A. Science 2009, 324, 930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yi C.; Yang C. G.; He C. Acc. Chem. Res. 2009, 42, 519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hausinger R. P. Crit. Rev. Biochem. Mol. Biol. 2004, 39, 21. [DOI] [PubMed] [Google Scholar]
- Sedgwick B.; Bates P. A.; Paik J.; Jacobs S. C.; Lindahl T. DNA Repair 2007, 6, 429. [DOI] [PubMed] [Google Scholar]
- Ito S.; D’Alessio A. C.; Taranova O. V.; Hong K.; Sowers L. C.; Zhang Y. Nature 2010, 466, 1129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elkins J. M.; Hewitson K. S.; McNeill L. A.; Seibel J. F.; Schlemminger I.; Pugh C. W.; Ratcliffe P. J.; Schofield C. J. J. Biol. Chem. 2003, 278, 1802. [DOI] [PubMed] [Google Scholar]
- Pfaffeneder T.; Hackner B.; Truss M.; Munzel M.; Muller M.; Deiml C. A.; Hagemeier C.; Carell T. Angew. Chem., Int. Ed. Engl. 2011, 50, 7008. [DOI] [PubMed] [Google Scholar]
- Ito S.; Shen L.; Dai Q.; Wu S. C.; Collins L. B.; Swenberg J. A.; He C.; Zhang Y. Science 2011, 333, 1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krumova P.; Weishaupt J. H. Cell. Mol. Life Sci. 2013, 70, 2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yin R.; Mao S. Q.; Zhao B.; Chong Z.; Yang Y.; Zhao C.; Zhang D.; Huang H.; Gao J.; Li Z.; Jiao Y.; Li C.; Liu S.; Wu D.; Gu W.; Yang Y. G.; Xu G. L.; Wang H. J. Am. Chem. Soc. 2013, 135, 10396. [DOI] [PubMed] [Google Scholar]
- He Y. F.; Li B. Z.; Li Z.; Liu P.; Wang Y.; Tang Q.; Ding J.; Jia Y.; Chen Z.; Li L.; Sun Y.; Li X.; Dai Q.; Song C. X.; Zhang K.; He C.; Xu G. L. Science 2011, 333, 1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L.; Lu X.; Lu J.; Liang H.; Dai Q.; Xu G. L.; Luo C.; Jiang H.; He C. Nat. Chem. Biol. 2012, 8, 328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maiti A.; Drohat A. C. J. Biol. Chem. 2011, 286, 35334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kriaucionis S.; Heintz N. Science 2009, 324, 929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pastor W. A.; Pape U. J.; Huang Y.; Henderson H. R.; Lister R.; Ko M.; McLoughlin E. M.; Brudno Y.; Mahapatra S.; Kapranov P.; Tahiliani M.; Daley G. Q.; Liu X. S.; Ecker J. R.; Milos P. M.; Agarwal S.; Rao A. Nature 2011, 473, 394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Globisch D.; Munzel M.; Muller M.; Michalakis S.; Wagner M.; Koch S.; Bruckl T.; Biel M.; Carell T. PLoS ONE 2010, 5, e15367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellen M.; Ayata P.; Dewell S.; Kriaucionis S.; Heintz N. Cell 2012, 151, 1417. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Binz T. Mycologia 1998, 90, 785. [Google Scholar]
- Zemach A.; McDaniel I. E.; Silva P.; Zilberman D. Science 2010, 328, 916. [DOI] [PubMed] [Google Scholar]
- Hashimoto H.; Pais J. E.; Zhang X.; Saleh L.; Fu Z. Q.; Dai N.; Correa I. R.; Zheng Y.; Cheng X. Nature 2013, 506, 391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iyer L. M.; Zhang D.; de Souza R. F.; Pukkila P. J.; Rao A.; Aravind L. Proc. Natl. Acad Sci. U.S.A. 2014, 111, 1676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stajich J. E.; Wilke S. K.; Ahren D.; Au C. H.; Birren B. W.; Borodovsky M.; Burns C.; Canback B.; Casselton L. A.; Cheng C. K.; Deng J.; Dietrich F. S.; Fargo D. C.; Farman M. L.; Gathman A. C.; Goldberg J.; Guigo R.; Hoegger P. J.; Hooker J. B.; Huggins A.; James T. Y.; Kamada T.; Kilaru S.; Kodira C.; Kues U.; Kupfer D.; Kwan H. S.; Lomsadze A.; Li W.; Lilly W. W.; Ma L. J.; Mackey A. J.; Manning G.; Martin F.; Muraguchi H.; Natvig D. O.; Palmerini H.; Ramesh M. A.; Rehmeyer C. J.; Roe B. A.; Shenoy N.; Stanke M.; Ter-Hovhannisyan V.; Tunlid A.; Velagapudi R.; Vision T. J.; Zeng Q.; Zolan M. E.; Pukkila P. J. Proc. Natl. Acad Sci. U.S.A. 2010, 107, 11889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iyer L. M.; Tahiliani M.; Rao A.; Aravind L. Cell Cycle 2009, 8, 1698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iyer L. M.; Zhang D.; Burroughs A. M.; Aravind L. Nucleic Acids Res. 2013, 41, 7635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu M.; Hon G. C.; Szulwach K. E.; Song C. X.; Zhang L.; Kim A.; Li X.; Dai Q.; Shen Y.; Park B.; Min J. H.; Jin P.; Ren B.; He C. Cell 2012, 149, 1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L.; Yu M.; He C. Acta Chim. Sinica 2012, 70, 2123. [Google Scholar]
- Song C. X.; Szulwach K. E.; Dai Q.; Fu Y.; Mao S. Q.; Lin L.; Street C.; Li Y.; Poidevin M.; Wu H.; Gao J.; Liu P.; Li L.; Xu G. L.; Jin P.; He C. Cell 2013, 153, 678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen L.; Wu H.; Diep D.; Yamaguchi S.; D’Alessio A. C.; Fung H. L.; Zhang K.; Zhang Y. Cell 2013, 153, 692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fu Y.; He C. Curr. Opin. Chem. Biol. 2012, 16, 516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang L.; Szulwach K. E.; Hon G. C.; Song C. X.; Park B.; Yu M.; Lu X.; Dai Q.; Wang X.; Street C. R.; Tan H.; Min J. H.; Ren B.; Jin P.; He C. Nat. Comm. 2013, 4, 1517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu M.; Hon G. C.; Szulwach K. E.; Song C. X.; Jin P.; Ren B.; He C. Nat. Protoc. 2012, 7, 2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.