Abstract
Aims/hypothesis
Vascular endothelial growth factor (VEGF) is essential for proper pancreatic development, islet vascularisation and insulin secretion. In the adult pancreas, VEGF is thought to be predominantly secreted by beta cells. Although human duct cells have previously been shown to secrete VEGF at angiogenic levels in culture, an analysis of the kinetics of VEGF synthesis and secretion, as well as elucidation of an in vivo role for this ductal VEGF in affecting islet function and physiology, has been lacking.
Methods
We analysed purified duct cells independently prepared by flow cytometry, surgical isolation or laser-capture microdissection. We infected duct cells in vivo with Vegfa short hairpin RNA (shRNA) in an intrapancreatic ductal infusion system and examined the effect of VEGF knockdown in duct cells in vitro and in vivo.
Results
Pancreatic duct cells express high levels of Vegfa mRNA. Compared with beta cells, duct cells had a much higher ratio of secreted to intracellular VEGF. As a bioassay, formation of tubular structures by human umbilical vein endothelial cells was essentially undetectable when cultured alone and was substantially increased when co-cultured with pancreatic duct cells but significantly reduced when co-cultured with duct cells pretreated with Vegfa shRNA. Compared with islets transplanted alone, improved vascularisation and function was detected in the islets co-transplanted with duct cells but not in islets co-transplanted with duct cells pretreated with Vegfa shRNA.
Conclusions/interpretation
Human islet preparations for transplantation typically contain some contaminating duct cells and our findings suggest that the presence of duct cells in the islet preparation may improve transplantation outcomes.
Keywords: Beta cell, Duct cell, FACS, Intrapancreatic ductal infusion, Islet transplantation, Laser-capture microdissection, shRNA, Vascular endothelial growth factor
Introduction
Vascular endothelial growth factor A (VEGF-A, hereafter referred to as VEGF) plays a crucial role during organ development, regeneration and carcinogenesis [1–5]. Alternative Vegfa mRNA splicing results in three protein isoforms (VEGF120, VEGF164 and VEGF188 in mice), of which VEGF164 has been shown to have the highest bioavailability and biological potency [3, 6, 7]. In the pancreas, beta cells have long been regarded as the major source of VEGF, which has been shown to be essential for proper pancreatic organogenesis, islet-specific capillary formation and beta cell function [8–19]. However, we have recently shown that VEGF secretion in beta cells is regulated differently from its production, suggesting that neither cellular mRNA levels nor cytoplasmic protein content of VEGF accurately reflects actual VEGF activity [20].
Islet transplantation is a promising treatment for diabetes, but its outcome has been hampered by poor graft vascularisation [21, 22]. Indeed, the survival and proper function of human islet cell grafts largely depend on a rapid and adequate revascularisation, during which VEGF plays a pivotal role [19, 23–28]. Of note, contaminating duct cells in human islet preparations for transplantation have been reported to express threefold more VEGF than islet endocrine cells and release VEGF at angiogenic levels in culture, suggesting that duct cells may be an important source of VEGF in the pancreas [29]. However, an analysis of the kinetics of VEGF synthesis and secretion, as well as elucidation of an in vivo role for this ductal VEGF in affecting islet function and physiology, has been lacking.
Here we analysed VEGF in purified cell types in the mouse adult pancreas, and showed that Vegfa transcript levels in duct cells are about 40% of the level in beta cells. We further showed that VEGF secreted by duct cells may play an important role in transplanted islet graft vascularisation and function.
Methods
Mouse manipulation
All mouse experiments were approved by the Animal Research and Care Committee at the Children’s Hospital of Pittsburgh and the University of Pittsburgh IACUC. BAC transgenic glucagon promoter Cre reporter (GCG-Cre) mice were purchased from MMRRC (Chapel Hill, NC, USA) [30]. The BAC transgenic elastase promoter CreERT reporter (Ela-CreERT) mouse has been described before [31, 32]. C57/6, Rosa26CAGTomato (Tomato) and BAC transgenic mouse insulin promoter green fluorescent protein reporter (MIP-GFP) mice [20] were all purchased from Jackson ImmunoResearch (Bar Harbor, Maine, USA). Tamoxifen induction of Tomato expression in acinar cells in Ela-CreERT; Tomato mice have been described before [32]. All experiments used 8-week-old male mice. Fasting blood glucose monitoring and intraperitoneal glucose tolerance test (IPGTT) were performed as described previously [20, 33, 34].
Pancreatic intraductal virus infusion was performed as described previously [32, 35], but here we delivered 50 μl adeno-associated virus serotype6 vectors (AAV6) (titration of 109) via catheter at a rate of 10 μl/min to optimise infection of duct cells in the current study. Islet transplantation was performed on a heated plate. The left kidney of the mouse was exposed through a lumbar incision. The kidney capsule was then incised with a Hamilton syringe (Fisher Scientific, Pittsburgh, PA, USA) and a pocket was made with polyethylene tubing, after which islets were placed under the kidney capsule. The incision in the kidney capsule was cauterised and the peritoneum and skin were closed with suture.
Production of AAV6 vectors expressing Vegfa small hairpin RNA under Sox9 promoter
The Vegfa small hairpin RNA (shRNA) sequence is 5′-TGTGAATGCAGACCAAAGA-3′. The scrambled sequence is 5′-GGTATCTACTAGATGTACT-3′. Vegfa shRNA or scrambled oligonucleotides were generated and cloned into the TOPO plasmid (Invitrogen, Carlsbad, CA, USA), which was then used to produce adeno-associated virus serotype6 vectors (AAV6) expressing AAV6-Sox9p-shVegf-2A-GFP (simplified as shVegf) or scrambled sequence AAV6-Sox9p-shCtr-2A-GFP (simplified as shCtr), followed by 2A driving GFP reporter under control of the Sox9 promoter, as described before [35–37].
Pancreatic digestion and FACS
Pancreatic duct perfusion, subsequent digestion of pancreas and FACS were performed as described before [20, 32–34]. The purity of sorted cell fractions was evaluated by expression of cell-type specific markers with RT-qPCR.
Primary cell culture and analysis of VEGF or insulin by ELISA
Primary beta cells, duct cells or islets were isolated and cultured as described previously [20]. The conditioned media and the cells were harvested for ELISA (RayBiotech, Norcross, GA, USA) to examine released VEGF and cellular VEGF, respectively. Cells were also harvested for RNA extraction to examine Vegfa transcript levels, and for DNA extraction to determine the cell number. Vegfa levels were normalised against the DNA content of the cells, as described previously [20]. Insulin levels in the serum or graft were determined using an insulin ELISA kit (ALPCO, Salem, NH, USA).
Human umbilical vein endothelial cell culture and collagen gel assay
Human umbilical vein endothelial cells (HUVECs) were grown in M-200 supplemented medium with low-serum growth supplement (Invitrogen) for 48 h before use. HUVECs were embedded in a collagen gel, and plated on a culture plate. Once the collagen polymerised (by keeping the plate at 37°C in a CO2 incubator for 10–15 min), the same number of purified primary duct cells (infected with shVegf or shCtr), or a piece of microdissected duct, was added to the culture plate. An additional 100–200 μl of collagen was layered on top of the existing gel and epithelium and the plate was returned to the CO2 incubator. The medium was replenished every day, and culture images were taken after 3 days.
Laser-capture microdissection and RT-qPCR
Laser-capture microdissection (LCM) of duct cells and Quantitative reverse transcription polymerase chain reaction (RT-qPCR) were performed as described before [20, 32]. RT-qPCR primers were all purchased from Qiagen (Hilden, Germany), except for VEGF isoforms (ESM Fig. 1). Values of genes were normalised against Cyclophilin A, which proved to be stable across the samples. Fold change under various conditions are shown, compared with the control condition (= 1).
Immunohistochemistry
All pancreas samples were fixed and cryo-protected in 30% sucrose overnight before freezing, as described before [20, 33, 34]. Staining with 3,3′-Diaminobenzidine (DAB) was performed using a DAB chromogen system (Dako, Carpinteria, CA, USA). A Biotin-TSA or Cy5-TSA enhancer kit (Fisher Scientific) was used to amplify VEGF signals. GFP was detected by direct fluorescence. Primary antibodies for immunostaining were: guinea pig polyclonal anti-insulin and anti-pancreatic polypeptide (Dako), goat polyclonal anti-VEGF-A (R&D systems, Minneapolis, MN, USA) and anti-insulin (Santa Cruz, CA, USA); rabbit polyclonal anti-somatostatin (Dako) and anti-VEGF-A (Santa Cruz); rat polyclonal anti-CD31 (BD, San Jose, CA, USA) and mouse monoclonal anti-glucagon (Sigma, St Louis, MO, USA). Secondary antibodies were all purchased from Jackson ImmunoResearch. Nuclear staining was performed with Hoechst (BD). When duct cells were labelled with biotin-dolichos biflorus agglutinin (DBA) (Vector Lab, Burlingame, CA, USA), they were detected with Cy5-conjugated streptavidin. Staining, imaging of sections and quantifications were performed as described previously [20, 33, 34].
Data analysis
All values are depicted as mean ± SEM. Each in vitro experimental condition contains five repeats. Each in vivo experimental group used five mice and each mouse pancreas was analysed on the basis of five sections that were 100 μm apart. All RT-qPCR data are from five samples for each condition. All data were statistically analysed using a two-tailed Student’s t test. Significance was considered when p < 0.05.
Results
Duct cells produce significant levels of Vegfa transcript
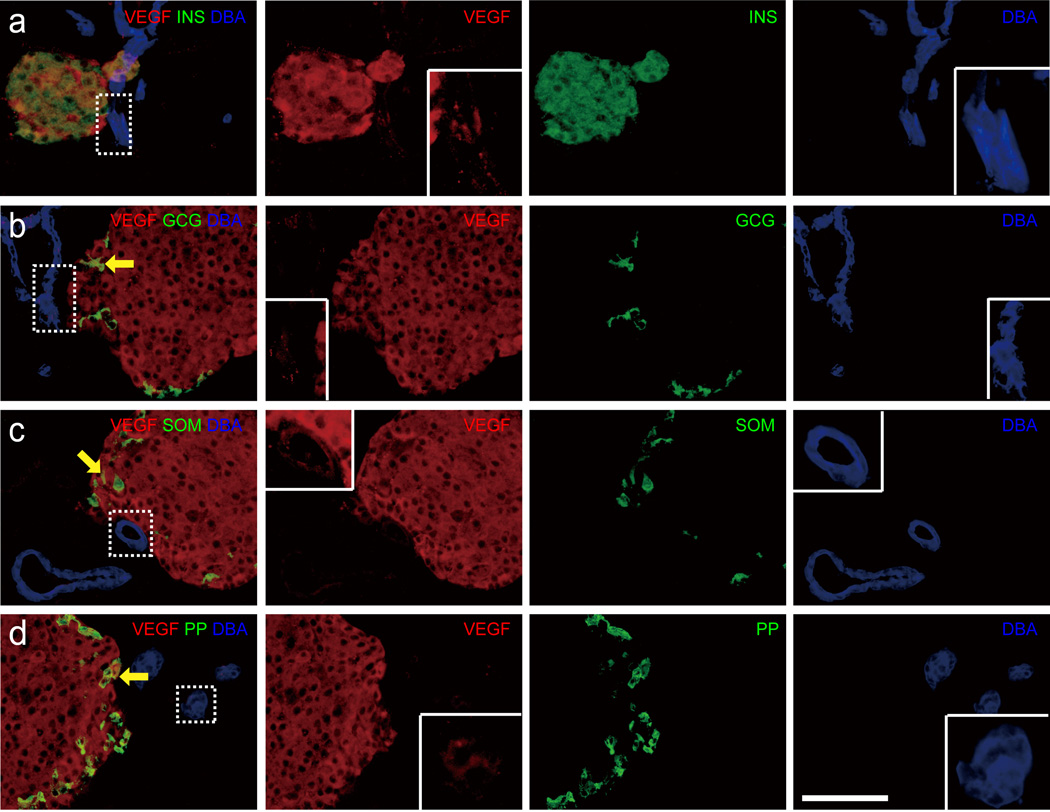
We examined the expression of VEGF in normal mouse pancreases by immunostaining and found that 98.4% of beta cells co-stained for VEGF (Fig. 1a), as the major source of VEGF in islets. Of note, VEGF was also detected in a small proportion of other endocrine cells (alpha cells, 14.3%; delta cells, 1.2%; pancreatic polypeptide [PP] cells, 34.5%) (Fig. 1 b–d).
Fig. 1. Expression of VEGF in mouse pancreas.
(a–d) Representative images of mouse pancreas immunostaining. Confocal triple fluorescent images for VEGF (red), DBA (blue) and different endocrine markers (INS, insulin; GCG, glucagon; SOM, somatostatin; PP, pancreatic polypeptide) (green). The areas in merged channels outlined with a dashed line in the first column of panels are shown in the insets in the VEGF and DBA channels at higher magnification. The insets show low VEGF positivity in DBA-positive (DBA+) duct cells. Arrows point to VEGF+GCG+ (b), VEGF+SOM+ (c) and VEGF+PP+ (d) cells. Scale bars, 50 μm
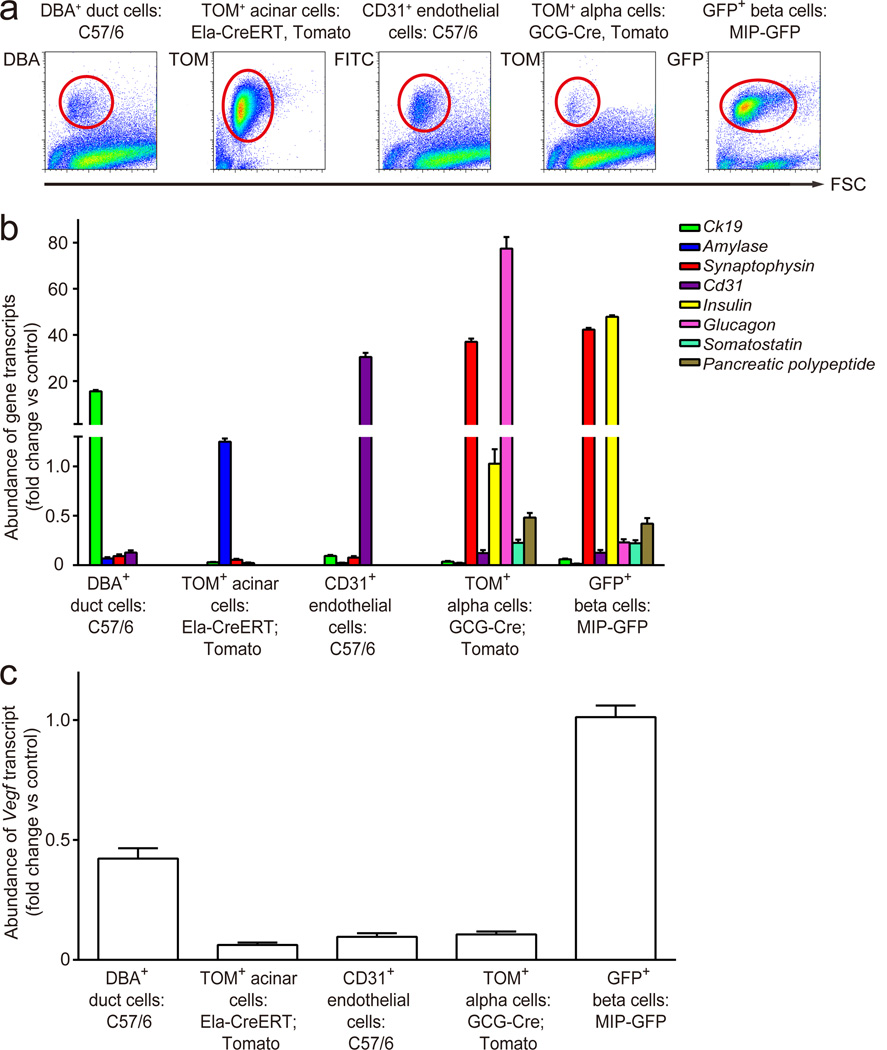
Messenger RNA levels of Vegfa have been thought to be an accurate reflection of protein levels [38–40]. Recently, we reported that, specifically in pancreatic beta cells, secretion of VEGF is differently regulated from translation, suggesting that neither cellular mRNA levels nor cytoplasmic VEGF protein necessarily reflect VEGF signalling activity. This differential regulation may also explain the high VEGF immunostaining signal in beta cells [20]. Interestingly, weak VEGF immunostaining was readily detected in DBA-positive (DBA+) duct cells (Fig. 1 a–d, insets), and could be better visualised with biotin-TSA enhancer staining (ESM Fig. 2). We then compared VEGF transcript levels among different FACS-purified pancreatic cell types from digested pancreas, with sorting based on labelling either genetically or by specific membrane markers. Three transgenic mice (MIP-GFP [41], GCG-Cre;Tomato [30] and Ela-CreERT;Tomato [31], with a labelling efficiency of 93.5%, 67.5% and 98.6%, respectively) were used for isolating beta cells, alpha cells and acinar cells, respectively. Essentially, zero (less than 0.01%) non-specific labelling was seen in any of the three strains. In parallel, fluorescein-DBA or FITC-conjugated CD31 antibody was applied to pancreatic digests from C57/6 mice to label duct cells or endothelial cells for sorting, respectively (Fig. 2a). Expression of genes encoding CK19 (specific to duct cells), amylase (specific to acinar cells), synaptophysin (specific to pan-islet-endocrine cells) and CD31 (specific to endothelial cells) was evaluated in sorted cells to confirm the purity of fluorescence-activated cell sorting. Additionally, for beta cell isolation from MIP-GFP mice and alpha cell isolation from GCG-Cre; Tomato mice, individual endocrine genes (encoding insulin, glucagon, somatostatin and PP) were further checked to assure the endocrine cell-type specificity of the sorted cells (Fig. 2b). Finally, Vegfa transcript levels were analysed in these five cell populations, and showed much lower levels in alpha cells, acinar cells and endothelial cells, but a relatively high level in duct cells (42.2 ± 2.8%), compared with beta cells (Fig. 2c).
Fig. 2. Duct cells express high levels of Vegfa mRNA.
(a) MIP-GFP, GCG-Cre;Tomato (TOM) and Ela-CreERT;Tomato (TOM) mice were used for isolating either beta cells based on green fluorescence, or for alpha cells, or acinar cells based on red fluorescence, respectively. Fluorescein-DBA or FITC-conjugated CD31 antibody was applied to label duct cells or endothelial cells, respectively, from C57/6 mice. Pancreatic or islet digests were analysed by flow cytometry, in which the target cell populations were sorted (circled). (b) Expression of genes encoding CK19, amylase, synaptophysin and CD31 was evaluated in sorted cells to confirm purity. Expression of individual endocrine genes (encoding insulin, glucagon, somatostatin and PP) was further checked to assure the purity of sorted beta cells and alpha cells. Fold change over total pancreas (= 1) is shown. (c) Vegfa transcript levels were examined in various cell types. Fold change over beta cells (= 1) is shown. FSC, forward-scattered light
To confirm this relatively high level of Vegfa mRNA expression in duct cells, the duct cells were isolated by two other methods. First, the main pancreatic ducts were microdissected from the surrounding tissues and placed in RNA-later (Qiagen) to preserve RNA (Fig. 3a). In another approach, duct cells were sampled by LCM, following an intrapancreatic ductal injection with fluorescein-DBA prior to harvesting (Fig. 3b). Specific markers were evaluated in the mRNA from sampled duct cells to confirm purity (Fig. 3c). Our data showed that Vegfa transcript levels were similarly elevated in duct cells isolated by all three methods (Fig. 3d), suggesting that pancreatic duct cells express a relatively high level of Vegfa mRNA compared with other non-beta pancreatic cells.
Fig. 3. A high level of Vegfa mRNA was detected in duct cells purified by different methods.
(a) Representative image of surgically dissected pancreatic duct connected to the common pancreatobiliary duct. (b) Representative image showing isolation of duct cells by LCM following an intraductal DBA injection prior to harvesting and histology. (c) Levels of mRNA for genes encoding CK19 (white bars), amylase (light-grey bars), synaptophysin (dark-grey bars) and CD31 (black bars) were evaluated in the microdissected or LCM ducts, and then compared with purified beta cells to assure purity. Fold change over total pancreas (= 1) is shown. (d) Relatively high levels of Vegfa transcripts were detected in the microdissected or LCM ducts. Fold change over beta cells (= 1) is shown. *p<0.05. Scale bar, 200 μm
Cultured duct cells release more VEGF and retain lower intracellular VEGF than beta cells
The relatively high levels of Vegfa mRNA in duct cells despite the relatively low VEGF immunostaining signal might result from a more rapid VEGF release from duct cells compared with beta cells. Thus, we compared VEGF protein synthesis and release by duct cells with that by beta cells in culture. Our data showed that, compared with beta cells, duct cells secreted a significantly higher level of VEGF protein into conditioned media, but contained a much lower intracellular level of VEGF protein in cell lysates (Fig. 4a). These data suggest that, compared with beta cells, although duct cells produce less VEGF, they release the VEGF at a more rapid rate. In contrast, beta cells produce more VEGF but seem to retain it longer within the cells [20].
Fig. 4. Duct cell VEGF is rapidly secreted, and does not differ in isoforms from beta cell VEGF. VEGF synthesis and release were compared between purified duct cells and beta cells.
(a) Both the conditioned media and cells were harvested to examine VEGF levels by ELISA. Genomic DNA was used to determine the number of cells. A significantly higher level of VEGF in conditioned media, but a much lower intracellular VEGF in cell lysates, was detected in duct cells compared with beta cells. (b) Gene expression analysis on VEGF isoform VEGF120 (grey bars) and VEGF164 (white bars). Fold change over beta cells (= 1) is shown. *p<0.05
Differences in VEGF secretion by duct cells and beta cells are not due to differences in expression of VEGF isoforms
Mouse VEGF includes VEGF120, VEGF164 and VEGF188 isoforms, which vary in localisation, accessibility, receptor binding and activity, and appear to co-ordinately orchestrate the processes of vascular growth and development [3, 15, 42]. Isoform-specific primers were thus designed (ESM Fig. 1) and applied to gene expression analysis. Our data showed that VEGF164 is the major isoform produced by both duct cells and beta cells, and that the ratio of VEGF120 to VEGF164 is similar in duct cells and beta cells (Fig. 4b). VEGF188 was not detected, consistent with previous reports [11]. These data suggest that the differences in VEGF synthesis and release in duct cells and beta cells are not due to differential expression of VEGF isoforms.
VEGF secreted by duct cells promotes tube formation by HUVECs in culture
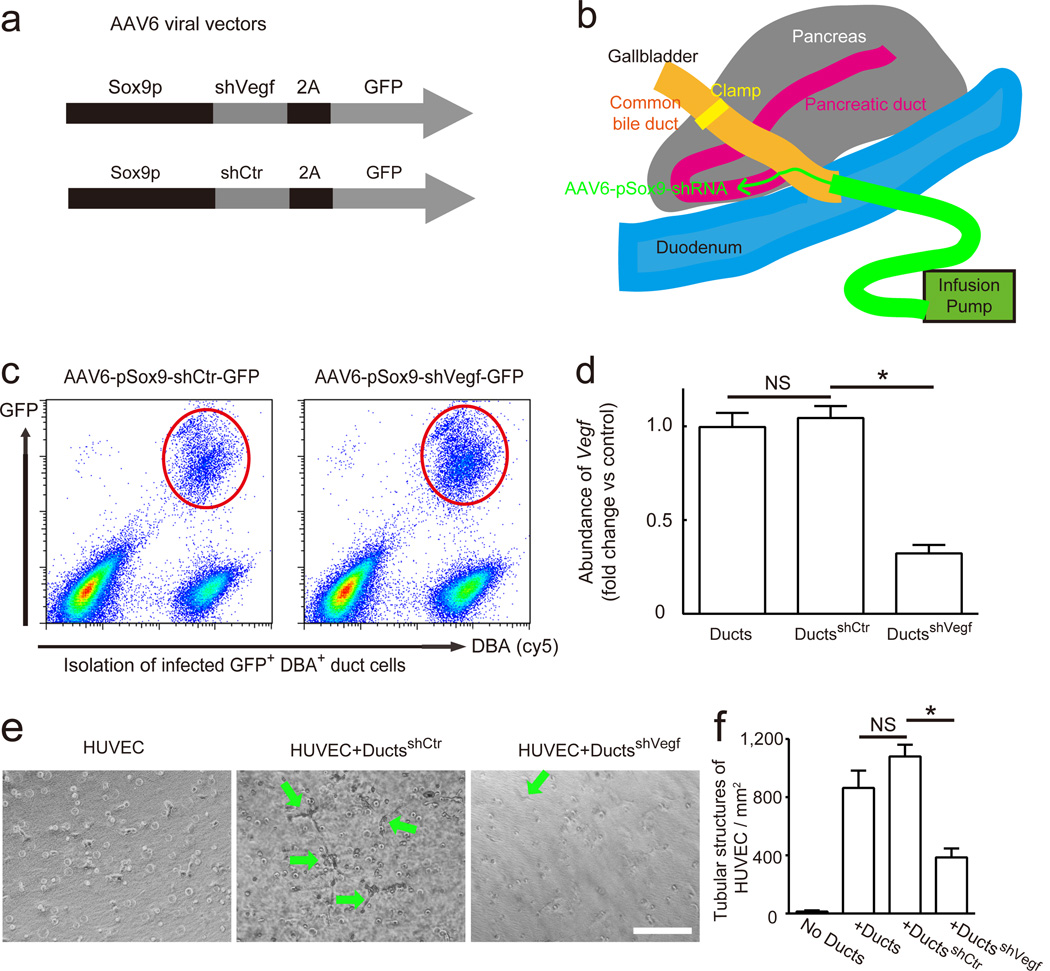
To confirm the vasculogenic effect of the VEGF secreted by duct cells, we knocked down VEGF production in primary duct cells using shRNA. We generated AAV6 vectors carrying Vegfa shRNA and a GFP reporter under the Sox9 promoter (shVegf). Vectors with a scrambled sequence were also generated as controls (shCtr) (Fig. 5a). AAV6 was used because it has the highest infection efficiency in duct cells among all adeno-associated virus serotypes [35, 43]. We used the Sox9 promoter to drive shRNA to increase the specificity of duct cell infection, since Sox9 is predominantly expressed by duct cells in the adult mouse pancreas [35, 44, 45]. Then we performed an intrapancreatic ductal infusion with shRNA viruses (Fig. 5b), as we described before [32, 35]. A positive correlation between GFP signal intensity (representing successful infection of duct cells) and decreased VEGF immunostaining signal in duct cells by shVegf confirmed the proper targeting of the virus to the pancreatic ducts, and confirmed the ability of the shVegf to inhibit the expression of VEGF (ESM Fig 3). The shRNA-infected pancreatic duct cells were isolated by flow cytometry based on GFP and DBA 5 days after viral infusion (Fig. 5c). Analysis of mRNA confirmed the immunostaining findings, and showed a 70% knockdown of Vegfa transcripts in duct cells by shVegf, compared with shCtr-infected or untreated duct cells (Fig. 5d). When untreated duct cells and duct cells infected with shVegf or shCtr were subsequently co-cultured with HUVECs in a tube formation assay, the HUVECs substantially increased their tube formation when co-cultured with pancreatic duct cells, with or without shCtr infection. Furthermore, this tube formation was significantly reduced when the co-cultured duct cells had been infected with shVegf (Fig. 5 e, f). To exclude the possibility that changes in ductal structure or integrity due to the single-cell dispersion of the duct cells may have affected VEGF synthesis and release, HUVECs were co-cultured with microdissected intact pancreatic ducts. Similar levels of tube formation were observed, compared with HUVECs co-cultured with fluorescence-activated sorted duct cells (ESM Fig. 4). Taken together, our results suggest that VEGF secreted by duct cells specifically promotes tube formation by HUVECs in culture.
Fig. 5. VEGF secreted by duct cells promotes tube formation by HUVECs in culture.
(a) Schematic representation of generation of AAV6 carrying shVegf and a GFP reporter under the Sox9 promoter (shVegf). Vectors with a scrambled sequence were also generated as controls (shCtr). (b) Schematic representation of injection of AAV vectors carrying shVegf or shCtr by intrapancreatic ductal infusion. (c) shRNA-infected pancreatic duct cells were isolated by flow cytometry based on GFP and DBA 5 days after viral infusion. (d) mRNA in duct cells sorted by FACS were analysed by RT-qPCR and showed a 70% knockdown of Vegfa transcripts by shVegf, compared with controls. Fold change over untreated duct cells (= 1) is shown. (e, f) In a tube formation assay, HUVECs substantially increased their tube formation when co-cultured with pancreatic duct cells pretreated with shCtr (arrows), but not when co-cultured with duct cells pretreated with shVegf. These data are shown in representative images (e) and in quantification (f). DuctsshVegf, duct cells infected with shVegf; DuctsshCtr, duct cells infected with shCtr. *p < 0.05. Scale bar, 50 μm
VEGF secreted by duct cells improves vascularisation and function of grafted islets
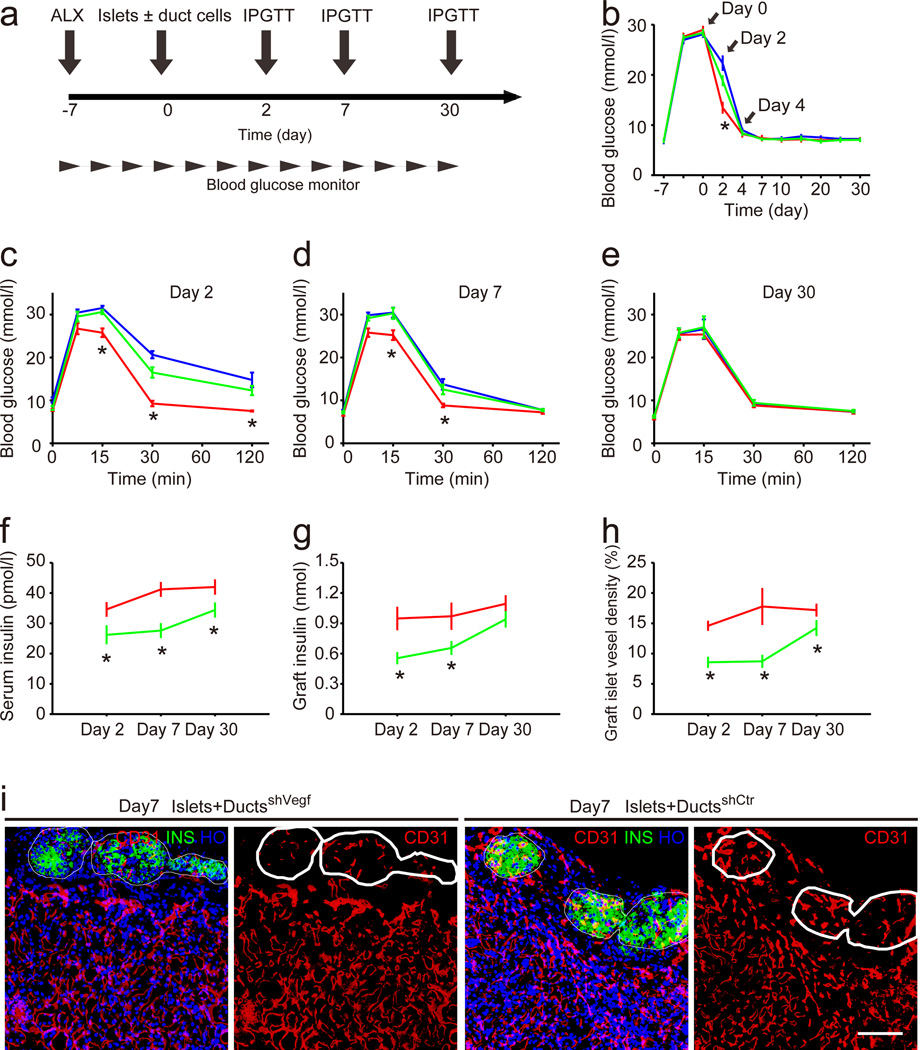
Since duct cells are often present in human islet preparations for transplantation [29], and since VEGF is well known for its effect on the revascularisation and survival of islet grafts [19, 24, 26], we co-transplanted 105 duct cells, infected with either shVegf or shCtr, along with 300 islets, under the kidney capsule of isogeneic mice 7 days after alloxan injection to ablate recipient beta cells (Fig. 6a). The grafted islets normalised the blood glucose more rapidly (Fig. 6b), had a better glucose response (Fig. 6 c–e), greater serum insulin levels (Fig. 6f) and greater graft insulin content (Fig. 6g) when co-transplanted with either normal duct cells (not shown) or with shCtr-infected duct cells, compared with islets transplanted alone (no ducts) or transplanted together with shVegf-infected duct cells. These data suggest that VEGF secreted by ducts improves islet function in the grafts. Immunostaining further showed significantly higher islet vessel density at 2 and 7 days after transplantation when the islets were co-transplanted with untreated ducts (not shown) or ducts infected with shCtr, compared with islets transplanted alone (not shown) or with ducts infected with shVegf (Fig. 6 h, i). These results suggest that VEGF secreted by ducts accelerates islet graft function by improving islet vascularisation, possibly through preventing the loss of pre-existing islet vessels and promoting islet graft revascularisation.
Fig. 6. VEGF secreted by duct cells accelerates grafted islet vascularisation and function.
(a) Schematic representation of experiment. Seven days after alloxan (ALX) treatment to establish hyperglycaemia, 300 isogeneic islets were transplanted alone, or co-transplanted with 105 duct cells infected with either shVegf or shCtr. (b–g) Functional evaluation of the islet grafts. The blood glucose (b), glucose response (IPGTT) on days 2 (c), 7 (d) and 30 (e) after transplantation, fasting serum insulin content (f) and graft insulin content (g), were examined in the mice that received islets alone (blue line), or co-transplanted with shCtr-infected ducts (red line) or shVegf-infected ducts (green line). (h–i) Graft islet vessel density was evaluated. (h) Quantification of islet graft vessel density. (i) Representative immunostaining of CD31 and insulin on day 7 after transplantation. DuctsshVegf, duct cells infected with shVegf; DuctsshCtr, duct cells infected with shCtr. *p<0.05, DuctsshCtr vs DuctsshVegf or no ducts. Scale bar, 50 μm
Discussion
Pancreatic beta cells have long been regarded as the only significant source of VEGF in the pancreas, and VEGF expression by beta cells is reported to be crucial for proper pancreatic organogenesis and proper insulin secretion [8–19]. Of note, we recently showed that synthesis and release of VEGF are uncoupled in beta cells [20]. In the current study, we further show that pancreatic duct cells produce significant levels of VEGF but display a low cellular immunostaining signal due to prompt secretion of the VEGF. Our data thus confirm previous findings regarding VEGF expression by human duct cells [29], and further reveal an interesting cell-type-dependent regulation of VEGF secretion, separate from the regulation of VEGF synthesis. As endocrine cells, beta cells by necessity are closely associated with capillaries; this association allows accurate glucose sensing and prompt secretion of hormones into the blood. As non-endocrine cells, duct cells would not seem to need such close association with endothelial cells. Therefore, our finding that VEGF release is regulated by glucose levels in beta cells might represent a generalisable difference between endocrine and non-endocrine cells [20]. Interestingly, here we also found that a smaller percentage of non-beta endocrine cells stain positive for VEGF, but contain significantly fewer Vegfa transcripts than duct cells. Hence, examining the secretion and synthesis of VEGF in other endocrine and non-endocrine cell types may confirm whether this differential regulation of VEGF synthesis and secretion is seen in other endocrine cell types.
Also, our findings have important ramifications for pancreatic development, diabetes and pancreatic ductal carcinoma in that pancreatic ducts appear to be an important but overlooked source of VEGF. First, there is variability in the literature regarding the role of VEGF during pancreas development, depending on whether the Pdx1 or insulin promoter is used to drive VEGF expression. This inconsistency may be simply explained by VEGF expression in the embryonic ducts/epithelial cells (Pdx1 positive, but insulin negative), and thus suggests that embryonic ductal/epithelial VEGF is a key regulator of vasculogenesis [8, 16–18, 46, 47]. Second, VEGF has been suggested as a key regulator during the onset of autoimmune diabetes, but there is controversy in the literature concerning the exact role of VEGF in this pathogenesis [15, 48]. Secretion of VEGF by duct cells could be an important factor in the pathogenesis of diabetes. Moreover, it is thought that pancreatic cancer can develop from acinar-ductal metaplasia [49, 50] and that metaplastic cells may allow tumour progression by secreting VEGF to induce tumour angiogenesis. Finally, it has been reported that pancreatic duct cell growth and differentiation is dependent on VEGF [51].
It is well established that VEGF is essential for proper islet revascularisation and islet function after islet transplantation. Previous studies have shown improved outcomes after islet transplantation by either overexpressing VEGF in beta cells or by co-transplanting endothelial (progenitor) cells with the beta cells [19, 23–26]. Here, we show that the presence of duct cells significantly improved tube formation by HUVECs in culture and that this effect was significantly decreased when VEGF production in duct cells was specifically inhibited by shRNA targeting VEGF. Furthermore, we found that co-transplantation with duct cells significantly shortened the time lag before proper function of grafted islets, apparently due to improved VEGF-dependent islet graft vascularisation. Unlike the results from insulin content measurement and glucose tolerance testing, the difference in fasting blood glucose is only significant in the early stages after transplantation. This may be due to the fact that with time even the relatively poorly functioning islets grafted with VEGF-depleted duct cells are still adequate to prevent significant hyperglycaemia. Experiments with a more severe reduction in the number of transplanted islets may result in a greater difference in fasting blood glucose between mice transplanted with islets plus VEGF-depleted ducts and mice transplanted with islets plus normal ducts.
Taken together, our data suggest that duct cells may improve the vascularisation of the islet grafts and, subsequently, islet function after transplantation, consistent with the findings of a previous study [29]. Since duct cells are commonly present in islet preparations for human islet transplantation and since poor graft revascularisation can contribute to inferior outcomes after islet transplantation, our data suggest that transplanted human islets may be better vascularised when duct cells are present in the islet preparations. Further studies are needed to evaluate whether duct cells may also increase immune reactivity to grafted islets in addition to any pro-angiogenic effect [29].
Supplementary Material
Acknowledgements
Special thanks go to A. J. Styche, R. J. Lakomy, M. Branca and L. Brink (all from Children’s Hospital of Pittsburgh, Pittsburgh, PA, USA) for technical assistance in flow cytometry, LCM, confocal microscopy and mouse genotyping.
Funding
This work was supported, in whole or in part, by the NIH (GKG; R01 DK083541, R01 DK098196) and Cochrane-Weber Endowed Fund in Diabetes Research (XX; NO19831).
Abbreviations
- AAV6
Adeno-associated virus serotype6 vectors
- DBA
Dolichos biflorus agglutinin
- Ela
Elastase promoter
- FACS
Fluorescence-activated cell sorting
- GCG
Glucagon promoter
- GFP
Green fluorescent protein
- HUVEC
Human umbilical vein endothelial cell
- IPGTT
Intraperitoneal glucose tolerance test
- LCM
Laser-capture microdissection
- MIP
Mouse insulin promoter
- PP
Pancreatic polypeptide
- RT-qPCR
Quantitative reverse transcription PCR
- shCtr
Vector expressing AAV6-Sox9p-shCtr-2A-GFP
- shRNA
Short hairpin RNA
- shVegf
Vector expressing AAV6-Sox9p-shVegf-2A-GFP
- VEGF
Vascular endothelial growth factor
Footnotes
Duality of interest
The authors declare that there is no duality of interest associated with this manuscript.
Contribution statement
The study was conceived and designed by XX and GKG. Acquisition of data was by XX, KP, PG, YEG, SF, JW, IG and CS, and analysis of data was carried out by XX, KP, PG and YEG. XX and GKG interpreted the data. XX drafted the article, and all authors revised the article and approved the final version to be published.
References
- 1.Cleaver O, Melton DA. Endothelial signaling during development. Nat Med. 2003;9:661–668. doi: 10.1038/nm0603-661. [DOI] [PubMed] [Google Scholar]
- 2.Carmeliet P, Jain RK. Molecular mechanisms and clinical applications of angiogenesis. Nature. 2011;473:298–307. doi: 10.1038/nature10144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669–676. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 4.Carmeliet P, Ferreira V, Breier G, et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature. 1996;380:435–439. doi: 10.1038/380435a0. [DOI] [PubMed] [Google Scholar]
- 5.Ferrara N. Vascular endothelial growth factor. Arterioscler Thromb Vasc Biol. 2009;29:789–791. doi: 10.1161/ATVBAHA.108.179663. [DOI] [PubMed] [Google Scholar]
- 6.Stalmans I, Ng YS, Rohan R, et al. Arteriolar and venular patterning in retinas of mice selectively expressing VEGF isoforms. J Clin Invest. 2002;109:327–336. doi: 10.1172/JCI14362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dor Y, Djonov V, Keshet E. Making vascular networks in the adult: branching morphogenesis without a roadmap. Trends Cell Biol. 2003;13:131–136. doi: 10.1016/s0962-8924(03)00022-9. [DOI] [PubMed] [Google Scholar]
- 8.Lammert E, Cleaver O, Melton D. Induction of pancreatic differentiation by signals from blood vessels. Science. 2001;294:564–567. doi: 10.1126/science.1064344. [DOI] [PubMed] [Google Scholar]
- 9.Lammert E, Gu G, McLaughlin M, et al. Role of VEGF-A in vascularization of pancreatic islets. Curr Biol. 2003;13:1070–1074. doi: 10.1016/s0960-9822(03)00378-6. [DOI] [PubMed] [Google Scholar]
- 10.Cleaver O, Dor Y. Vascular instruction of pancreas development. Development. 2012;139:2833–2843. doi: 10.1242/dev.065953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Inoue M, Hager JH, Ferrara N, Gerber HP, Hanahan D. VEGF-A has a critical, nonredundant role in angiogenic switching and pancreatic beta cell carcinogenesis. Cancer Cell. 2002;1:193–202. doi: 10.1016/s1535-6108(02)00031-4. [DOI] [PubMed] [Google Scholar]
- 12.Christofori G, Naik P, Hanahan D. Vascular endothelial growth factor and its receptors, flt-1 and flk-1, are expressed in normal pancreatic islets and throughout islet cell tumorigenesis. Mol Endocrinol. 1995;9:1760–1770. doi: 10.1210/mend.9.12.8614412. [DOI] [PubMed] [Google Scholar]
- 13.Watada H. Role of VEGF-A in pancreatic beta cells. Endocr J. 2010;57:185–191. doi: 10.1507/endocrj.k09e-035. [DOI] [PubMed] [Google Scholar]
- 14.Bonner-Weir S. Morphological evidence for pancreatic polarity of beta-cell within islets of Langerhans. Diabetes. 1988;37:616–621. doi: 10.2337/diab.37.5.616. [DOI] [PubMed] [Google Scholar]
- 15.Akirav EM, Baquero MT, Opare-Addo LW, et al. Glucose and inflammation control islet vascular density and beta-cell function in NOD mice: control of islet vasculature and vascular endothelial growth factor by glucose. Diabetes. 2011;60:876–883. doi: 10.2337/db10-0793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Agudo J, Ayuso E, Jimenez V, et al. Vascular endothelial growth factor-mediated islet hypervascularization and inflammation contribute to progressive reduction of beta-cell mass. Diabetes. 2012;61:2851–2861. doi: 10.2337/db12-0134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Magenheim J, Ilovich O, Lazarus A, et al. Blood vessels restrain pancreas branching, differentiation and growth. Development. 2011;138:4743–4752. doi: 10.1242/dev.066548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Iwashita N, Uchida T, Choi JB, et al. Impaired insulin secretion in vivo but enhanced insulin secretion from isolated islets in pancreatic beta cell-specific vascular endothelial growth factor-A knock-out mice. Diabetologia. 2007;50:380–389. doi: 10.1007/s00125-006-0512-0. [DOI] [PubMed] [Google Scholar]
- 19.Brissova M, Shostak A, Shiota M, et al. Pancreatic islet production of vascular endothelial growth factor--a is essential for islet vascularization, revascularization, and function. Diabetes. 2006;55:2974–2985. doi: 10.2337/db06-0690. [DOI] [PubMed] [Google Scholar]
- 20.Xiao X, Guo P, Chen Z, et al. Hypoglycemia reduces vascular endothelial growth factor a production by pancreatic Beta cells as a regulator of Beta cell mass. J Biol Chem. 2013;288:8636–8646. doi: 10.1074/jbc.M112.422949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pipeleers D, Keymeulen B, Chatenoud L, et al. A view on beta cell transplantation in diabetes. Ann N Y Acad Sci. 2002;958:69–76. doi: 10.1111/j.1749-6632.2002.tb02948.x. [DOI] [PubMed] [Google Scholar]
- 22.Halban PA, German MS, Kahn SE, Weir GC. Current status of islet cell replacement and regeneration therapy. J Clin Endocrinol Metab. 2010;95:1034–1043. doi: 10.1210/jc.2009-1819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brissova M, Fowler M, Wiebe P, et al. Intraislet endothelial cells contribute to revascularization of transplanted pancreatic islets. Diabetes. 2004;53:1318–1325. doi: 10.2337/diabetes.53.5.1318. [DOI] [PubMed] [Google Scholar]
- 24.Nyqvist D, Kohler M, Wahlstedt H, Berggren PO. Donor islet endothelial cells participate in formation of functional vessels within pancreatic islet grafts. Diabetes. 2005;54:2287–2293. doi: 10.2337/diabetes.54.8.2287. [DOI] [PubMed] [Google Scholar]
- 25.Lai Y, Schneider D, Kidszun A, et al. Vascular endothelial growth factor increases functional beta-cell mass by improvement of angiogenesis of isolated human and murine pancreatic islets. Transplantation. 2005;79:1530–1536. doi: 10.1097/01.tp.0000163506.40189.65. [DOI] [PubMed] [Google Scholar]
- 26.Zhang N, Richter A, Suriawinata J, et al. Elevated vascular endothelial growth factor production in islets improves islet graft vascularization. Diabetes. 2004;53:963–970. doi: 10.2337/diabetes.53.4.963. [DOI] [PubMed] [Google Scholar]
- 27.Shimoda M, Chen S, Noguchi H, Matsumoto S, Grayburn PA. In vivo non-viral gene delivery of human vascular endothelial growth factor improves revascularisation and restoration of euglycaemia after human islet transplantation into mouse liver. Diabetologia. 2010;53:1669–1679. doi: 10.1007/s00125-010-1745-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim SC, Kim TH, We YM, Park HY, Cho KM, Han DJ. Study for improvement of early implantation and long-term graft survival in pancreatic islet cell transplantation by induction of angiogenesis with gene transfection of vascular endothelial growth factor. Transplant Proc. 2003;35:486–487. doi: 10.1016/s0041-1345(02)03916-7. [DOI] [PubMed] [Google Scholar]
- 29.Movahedi B, Gysemans C, Jacobs-Tulleneers-Thevissen D, Mathieu C, Pipeleers D. Pancreatic duct cells in human islet cell preparations are a source of angiogenic cytokines interleukin-8 and vascular endothelial growth factor. Diabetes. 2008;57:2128–2136. doi: 10.2337/db07-1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Herrera PL. Adult insulin- and glucagon-producing cells differentiate from two independent cell lineages. Development. 2000;127:2317–2322. doi: 10.1242/dev.127.11.2317. [DOI] [PubMed] [Google Scholar]
- 31.Ji B, Song J, Tsou L, et al. Robust acinar cell transgene expression of CreErT via BAC recombineering. Genesis. 2008;46:390–395. doi: 10.1002/dvg.20411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Xiao X, Guo P, Shiota C, et al. Neurogenin3 activation is not sufficient to direct duct-to-beta cell transdifferentiation in the adult pancreas. J Biol Chem. 2013;288:25297–25308. doi: 10.1074/jbc.M113.484022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Xiao X, Wiersch J, El-Gohary Y, et al. TGFbeta receptor signaling is essential for inflammation-induced but not beta-cell workload-induced beta-cell proliferation. Diabetes. 2013;62:1217–1226. doi: 10.2337/db12-1428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Xiao X, Chen Z, Shiota C, et al. No evidence for beta cell neogenesis in murine adult pancreas. J Clin Invest. 2013;123:2207–2217. doi: 10.1172/JCI66323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Guo P, Xiao X, El-Gohary Y, et al. Specific transduction and labeling of pancreatic ducts by targeted recombinant viral infusion into mouse pancreatic ducts. Lab Invest. 2013;93:1241–1253. doi: 10.1038/labinvest.2013.113. [DOI] [PubMed] [Google Scholar]
- 36.Guo P, El-Gohary Y, Prasadan K, et al. Rapid and simplified purification of recombinant adeno-associated virus. Journal of virological methods. 2012;183:139–146. doi: 10.1016/j.jviromet.2012.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Guo P, Xiao X, El-Gohary Y, et al. A simplified purification method for AAV variant by polyethylene glycol aqueous two-phase partitioning. Bioengineered. 2013;4:103–106. doi: 10.4161/bioe.22293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Shima DT, Adamis AP, Ferrara N, et al. Hypoxic induction of endothelial cell growth factors in retinal cells: identification and characterization of vascular endothelial growth factor (VEGF) as the mitogen. Mol Med. 1995;1:182–193. [PMC free article] [PubMed] [Google Scholar]
- 39.Cheng SY, Nagane M, Huang HS, Cavenee WK. Intracerebral tumor-associated hemorrhage caused by overexpression of the vascular endothelial growth factor isoforms VEGF121 and VEGF165 but not VEGF189. Proc Natl Acad Sci U S A. 1997;94:12081–12087. doi: 10.1073/pnas.94.22.12081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Huez I, Creancier L, Audigier S, Gensac MC, Prats AC, Prats H. Two independent internal ribosome entry sites are involved in translation initiation of vascular endothelial growth factor mRNA. Mol Cell Biol. 1998;18:6178–6190. doi: 10.1128/mcb.18.11.6178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Hara M, Wang X, Kawamura T, et al. Transgenic mice with green fluorescent protein-labeled pancreatic beta -cells. Am J Physiol Endocrinol Metab. 2003;284:E177–E183. doi: 10.1152/ajpendo.00321.2002. [DOI] [PubMed] [Google Scholar]
- 42.Ng YS, Rohan R, Sunday ME, Demello DE, D’Amore PA. Differential expression of VEGF isoforms in mouse during development and in the adult. Dev Dyn. 2001;220:112–121. doi: 10.1002/1097-0177(2000)9999:9999<::AID-DVDY1093>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 43.Jimenez V, Ayuso E, Mallol C, et al. In vivo genetic engineering of murine pancreatic beta cells mediated by single-stranded adeno-associated viral vectors of serotypes 6, 8 and 9. Diabetologia. 2011;54:1075–1086. doi: 10.1007/s00125-011-2070-3. [DOI] [PubMed] [Google Scholar]
- 44.Kopp JL, Dubois CL, Schaffer AE, et al. Sox9+ ductal cells are multipotent progenitors throughout development but do not produce new endocrine cells in the normal or injured adult pancreas. Development. 2011;138:653–665. doi: 10.1242/dev.056499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Lynn FC, Smith SB, Wilson ME, Yang KY, Nekrep N, German MS. Sox9 coordinates a transcriptional network in pancreatic progenitor cells. Proc Natl Acad Sci U S A. 2007;104:10500–10505. doi: 10.1073/pnas.0704054104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Cai Q, Brissova M, Reinert RB, et al. Enhanced expression of VEGF-A in beta cells increases endothelial cell number but impairs islet morphogenesis and beta cell proliferation. Dev Biol. 2012;367:40–54. doi: 10.1016/j.ydbio.2012.04.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhou Q, Law AC, Rajagopal J, Anderson WJ, Gray PA, Melton DA. A multipotent progenitor domain guides pancreatic organogenesis. Dev Cell. 2007;13:103–114. doi: 10.1016/j.devcel.2007.06.001. [DOI] [PubMed] [Google Scholar]
- 48.Villalta SA, Lang J, Kubeck S, et al. Inhibition of VEGFR-2 reverses type 1 diabetes in NOD mice by abrogating insulitis and restoring islet function. Diabetes. 2013;62:2870–2878. doi: 10.2337/db12-1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Crawford HC, Scoggins CR, Washington MK, Matrisian LM, Leach SD. Matrix metalloproteinase-7 is expressed by pancreatic cancer precursors and regulates acinar-to-ductal metaplasia in exocrine pancreas. J Clin Invest. 2002;109:1437–1444. doi: 10.1172/JCI15051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Morris JP, 4th, Cano DA, Sekine S, Wang SC, Hebrok M. Beta-catenin blocks Kras-dependent reprogramming of acini into pancreatic cancer precursor lesions in mice. J Clin Invest. 2010;120:508–520. doi: 10.1172/JCI40045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Rooman I, Schuit F, Bouwens L. Effect of vascular endothelial growth factor on growth and differentiation of pancreatic ductal epithelium. Lab Invest. 1997;76:225–232. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.