Abstract
Aim:
Dilated cardiomyopathy (DCM) is the most common cause of heart failure, and pharmacological intervention is not currently available. Here we investigate the effect of tetramethylpyrazine phosphate (TMPP) on the progression of DCM in the cTnTR141W transgenic mouse model.
Methods:
The cTnTR141W transgenic mice aged 2 months were divided into model group and TMPP group, whereas age-matched nontransgenic mice were used as wild-type control. TMPP 45 mg·kg−1·d−1 was administered for 7 months. Following assessment of cardiac function by echocardiography, cardiac tissues were prepared for histology and electron microscopy. Levels of molecular markers for cardiomyocyte hypertrophy and fibrosis were detected by RT-PCR. Expression of structural proteins of the sarcomere and intercalated disc was determined by Western blot.
Results:
TMPP significantly prevented cardiac dilatation and dysfunction with the development of DCM, and decreased mortality by 54%. TMPP decreased HW/BW ratios and expression of hypertrophic markers BNP and ACTA1, as well as reduced interstitial collagen deposition and expression of profibrotic markers Col1a1 and Col3a1. TMPP attenuated ultrastructural disruption caused by cTnTR141W expression and decreased expression of structural proteins myotilin and E-cadherin which were up-regulated in the cTnTR141W heart. Moreover, TMPP reduced the mRNA expression of Calm1 and Camk2b in the cTnTR141W heart.
Conclusion:
Our results suggest that TMPP could be a promising drug for prevention and treatment of DCM.
Keywords: dilated cardiomyopathy, cTnTR141W transgenic mouse, survival, tetramethylpyrazine phosphate
Introduction
Dilated cardiomyopathy (DCM) is a primary heart muscle disease characterized by dilation of the heart chambers and markedly reduced contraction1. The estimated prevalence of idiopathic dilated cardiomyopathy is 1 per 2500 of the general population, and appears to be on the rise2. Despite recent advances in pharmacological and surgical therapies, it is still a leading cause of death worldwide3. Currently, heart transplant is thought to be the most effective therapy for end-stage DCM. However, this approach obviously cannot be used for all of the numerous affected patients and is not suitable for patients with a mild disease state. Therefore, there is increasing demand for new therapeutic targets and methods for DCM.
That familial dilated cardiomyopathy (FDCM) accounts for 20%–48% of DCM is principally a result of mutations of the genes encoding cytoskeletal and sarcomeric proteins in cardiac myocytes4, 5. Clinical research has revealed that a missense mutation, R141W, in the strong tropomyosin-binding region of cardiac troponin T (cTnT) causes FDCM6. Recently, we reported that a cTnTR141W transgenic mouse manifested progressive chamber dilation and contractile dysfunction, and had a pathologic phenotype similar to that of human DCM7. Here, using the cTnTR141W transgenic mouse model of FDCM, tetramethylpyrazine phosphate (TMPP) was selected as a potential medicine for FDCM from 11 kinds of compounds isolated from herbs.
Tetramethylpyrazine (TMP) is a biologically active alkaloid isolated in 1957 from the traditional herbal medicine Ligusticum wallichii Franch, which has long been used to improve circulation and prevent clot formation in traditional Chinese medicine. Its pharmacological actions include vasodilatation8, inhibition of platelet aggregation9 and strong antioxidant effect10. TMP has been verified to be a new-type antagonist of calcium channel11, 12. TMPP is one of the derivatives of tetramethylpyrazine. The synthetic TMPP has been put on the market and widely used for the treatment of patients with ischemic cerebrovascular diseases13 and cardiovascular diseases14. However, its potential effect on DCM has not been studied either in vivo or in vitro.
In the present study, the effect of long-term treatment with TMPP on the development of dilated cardiomyopathy was examined in the cTnTR141W transgenic mouse model established in our laboratory7. We found that TMPP had beneficial effects on the dilated cardiomyopathy caused by cTnTR141W expression, which was demonstrated at organ, tissue and molecular levels.
Materials and methods
Animals
The αMHC-cTnTR141W transgenic mouse was established in our laboratory. The mice were maintained on a C57BL/6J genetic background. It develops phenotypic features characteristic of human DCM by 4 months of age. Genotyping of transgenic mice was detected by the PCR using the primers, 5′-GAACAGGAGGAAGGCTGAGGATGAG-3′ and 5′-TATTTCCAGCGCCCGGTGACTTTAG-3′. All the mice were bred in an AAALAC-accredited facility and the use of animals was approved by the Animal Care and Use Committee of the Institute of Laboratory Animal Science of Peking Union Medical College. This investigation conforms with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No 85-23, revised 1996).
Groups and treatment
We performed M-mode echocardiography in 50 αMHC-cTnTR141W transgene-positive mice at the age of 2 months, which is considered to be at the pre-dilated stage of DCM7. We determined the mean and the 95% CI of LVIDs in the transgenic mice. We used the upper limit of the 95% CI (LVIDs of >2.93 mm) as indicative of cardiac dilation and thus excluded 2 αMHC-cTnTR141W mice. The remaining 48 transgenic mice were randomly divided into two groups: Model group (n=24) and TMPP group (n=24). TMPP (Tetramethylpyrazine Phosphate Tablets, Beijing Yanjing Pharmaceutical Co, Ltd) dissolved in water was administered at 45 mg·kg−1·d−1 (human-equivalent dosage) for 7 months. Twenty-four age-matched transgene-negative mice were used as WT group.
Echocardiography
M-mode echocardiography was performed with the small animal echocardiography analysis system (Vevo770, Canada). In brief, mice were anesthetized lightly by intraperitoneal injection of tribromoethanol at a dose of 0.18 mL/10 g body weight. Fur was shaved from the upper sternal and subxiphoid areas, which were then moistened for better acoustic coupling. Limbs were attached to electrodes for electrocardiographic monitoring and timing of cardiac cycles. M-mode echocardiography of left ventricular long-axis was recorded at the tip of the mitral valve apparatus with a 30 MHz transducer. Left ventricular end-systolic diameter (LVIDs), left ventricular end-diastolic diameter (LVIDd), interventricular septum during diastole (IVSd), interventricular septum during systole (IVSs), left ventricular posterior wall during diastole (LVPWd), left ventricular posterior wall during systole (LVPWs), heart rate (HR) were measured. Left ventricular end-systolic volume (LVESV), left ventricular end-diastolic volume (LVEDV), ejection fraction (EF%), fractional shortening (FS%) was calculated. To control for the influence of heart rate variability, the dose of mouse-specific anesthetic was strictly maintained.
Survival analysis
Cumulative percent mortality in each group of mice was calculated every month and the data from 2 to 9 months of age were summarized. Upon death of each mouse, the body was autopsied by a pathologist and the morphological and pathological changes of the heart were recorded. Heart dilation and mural thrombi were observed on postmortem examination. Kaplan-Meier curves for survival analysis were compared by the log-rank test (Spss 10.0 software).
Heart weight/body weight ratio, H&E, and Masson trichrome staining
Mice at the ages of 3 and 6 months were sacrificed and the hearts were excised, washed in PBS, blotted dry on tissue paper, and weighed. The HW/BW (heart weight/body weight) ratio was then calculated. Myocardial tissue was sampled in cross-sections 2 mm in thickness throughout the left and right ventricles. Sections were fixed in formalin and embedded in paraffin. Lengthwise sections were cut in 4 μm thickness. The sections were stained with H&E and Masson Trichrome, and examined under the light microscope by a pathologist who was not given the mouse genotypes.
Reverse transcription-polymerase chain reaction (RT-PCR)
Briefly, total RNA was isolated from the hearts of mice aged 6 months using TRIzol Reagent (Invitrogen, Carlsbad, USA); 2 μg of total RNA was used to synthesize the first strand of cDNA using random hexamer primers according to the manufacturer's protocol for Superscript III reverse transcriptase (Invitrogen). The genes expression of cardiac hypertrophic markers brain natriuretic peptide (Nppb) and skeletal α–actin (Acta1), cardiac fibrosis markers procollagens COL1 (α1) (Col1a1) and COL3 (α1) (Col3a1), signaling molecules calmodulin 1 (Calm1), calcium/calmodulin-dependent protein kinase II beta (Camk2b) were detected by the RT-PCR. GAPDH was used as an internal standard for the determination of targeted mRNA levels. Bands were quantified by the densitometry function of the Quantity One software. The primers used (sense/antisense) for amplification in the RT-PCR: Acta1 5′-CCGACCCCGTCACCAGGGTG-3′,5′-ATCCAACACGAT GCCGGTG-3′ Nppb 5′-ATGGATCTCCTGAAGGTGCTGTC-3′,5′-CTACAACAA CTTCAGTGCGTTAC-3′ Col1a1 5′-CCTGCCTGCTTCGTGTAAACT-3′,5′-TTGG GTTGTTCGTCTGTTTCC-3′ Col3a1 5′-TTGATGTGCAGCTGGCATTC-3′,5′-GCC ACTGGCCTGATCCATAT-3′ Calm1 5′-CTGACTGAAGAGCAGATTGCTGAAT-3′,5′-CCCATCCTTGTCAAACACTCGG-3′ Camk2b 5′-CGTATGATTTCCCATC CCCT-3′,5′-CCTTGAGCTTCCTCCTTGCA-3′ Gapdh 5′-TCGGTGTGAACGGAT TTGGC-3′,5′-CAAGTGGGCCCCGGCCTTCT-3′.
Transmission electron microscopy
Myocardial biopsy samples of mice 6 months of age were routinely fixed in 2.5% glutaraldehyde in 0.1 mol/L phosphate buffer (pH 7.4) and postfixed in buffered 1% osmium tetroxide for 1 h. Samples were then dehydrated using several changes of ethanol and embedded in Epon 812. The thin sections were stained with uranyl acetate and lead citrate and examined under a JEM-1230 Transmission Electronic Microscope (JEOL, Japan) equipped with a digital camera.
Western blot
Mice at the ages of 6 months were randomly chosen for protein extraction. Equal amounts of soluble protein were run in 10% SDS-PAGE and transferred onto a nitrocellulose membrane (Immobilon NC; Millipore, France). Immunoblotting was carried out with antibodies specific for myotilin (Santa Cruz, USA, 1:200) and E-cadherin (Santa Cruz, USA, 1:200). Primary antibodies were visualized with anti-goat and anti-rabbit HRP conjugate secondary antibodies (Santa Cruz), using a chemiluminescent detection system (Western blotting Luminal Reagent, Santa Cruz). Variations in sample loading were normalized relative to the GAPDH signal. Bands were quantified by the densitometry function of the Quantity One software.
Statistical analysis
The results are presented as means±SEM. Statistical analyses were performed by one-way ANOVA followed by Tukey's post-hoc test. A value of P<0.05 were considered to be statistically significant.
Results
TMPP decreased mortality of cTnTR141W transgenic mice with DCM
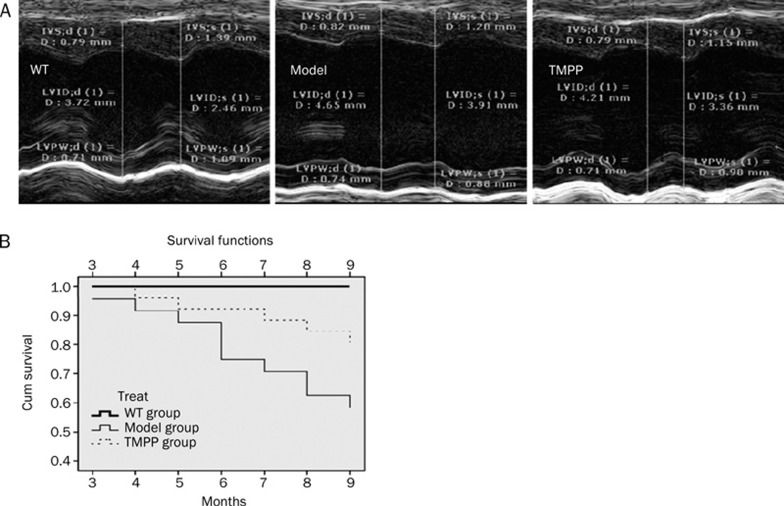
To evaluate the effect of long-term treatment with TMPP on the mortality due to DCM, survival analysis was assessed. Heart dilation and mural thrombi were observed on postmortem examination by the pathologist. Zero mortality was observed in the WT group between 2 and 9 months of age. At 9 months of age for the transgenic mice, there was 41% mortality in the model group, and 18% mortality in the TMPP group (Figure 1A). TMPP significantly decreased the mortality of cTnTR141W transgenic mice with DCM (P<0.05 by log-rank test).
Figure 1.
Effect of TMPP on survival rates and cardiac function in the cTnTR141W mice with DCM. (A) Representative M-mode echocardiographic images of parasternal long-axis of the LV from WT, Model and TMPP mice aged 6 months. They showed the LV chamber dimensions, the LV internal diameter, the thick and motion of LV posterior and septal wall during systole and diastole. (B) Kaplan-Meier survival curves. WT group (n=24), Model group (n=24) and TMPP group (n=24). Deaths of each group were recorded and cumulative every month in the mice from 2 months of age (the beginning of drug administration) to 9 months of age (treatment with TMPP for 7 months). Statistically significant difference was observed between Model group and TMPP group (P<0.05 by log-rank test).
TMPP prevented the cardiac dilation and dysfunction
The heart becomes enlarged and pumps blood less well in DCM. Echocardiography was performed once a month to assess left ventricular dimensions and function, and the data for mice aged 3 and 6 months were presented (Table 1, Figure 1B). The LVIDs of model group was increased by 65.8% (P<0.001) at 6 months, compared with the WT group, and the enlargement of the chamber was reduced with the treatment of TMPP by 41.3% (P<0.05) at 6 months. The increased LVIDd, LVESV and LVEDV in cTnTR141W transgenic mice were also decreased significantly (P<0.05) at 6 months after treatment with TMPP. The EF% of model group was decreased by 59.9% (P<0.001) at 6 months, compared with the WT group, and the pathological reduction of EF% was reversed by treatment with TMPP by 39.7% (P<0.05) at 6 months. The decreased FS% in cTnTR141W transgenic mice with DCM were also increased significantly (P<0.05) at 6 months after treatment with TMPP. The LVPWs and IVSs in the model group at 6 months were significantly decreased (P<0.05), compared with the WT group, but no statistically significant changes were found after treated with TMPP. There was no significant difference in the HR among three groups at 6 months.
Table 1. M-Mode echocardiographic analysis in 6-month-old mice. Values are expressed as the mean±SD. bP<0.05, cP<0.01 vs model group.
| 6 month (n=15) | |||
|---|---|---|---|
| Parameters | WT | Model | TMPP |
| BW, g | 31.80±1.03 | 28.56±3.11 | 29.58±3.69 |
| LVIDs, mm | 2.46±0.34b | 4.08±0.31 | 3.41±0.38b |
| LVIDd, mm | 3.85±0.26b | 4.95±0.29 | 4.45±0.31b |
| LVESV, μL | 21.97±5.89b | 105.51±14.12 | 86.42±12.43b |
| LVEDV, μL | 68.71±7.00c | 144.36±12.29 | 121.67±18.76b |
| IVSs, mm | 1.27±0.15b | 1.03±0.13 | 1.16±0.14 |
| IVSd, mm | 0.80±0.09 | 0.82±0.09 | 0.78±0.08 |
| LVPWs, mm | 1.03±0.13b | 0.71±0.11 | 0.85±0.09 |
| LVPWd, mm | 0.79±0.03 | 0.81±0.09 | 0.76±0.07 |
| EF, % | 66.30±7.18c | 26.56±5.92 | 42.33±6.85b |
| FS, % | 36.34±5.92c | 17.69±3.20 | 25.84±3.98b |
| HR | 455±28 | 470±21 | 465±37 |
LVIDs, left ventricular internal diameter at end-systole; LVIDd, left ventricular internal diameter at end-diastole; LVESV, left ventricular end-systolic volume; LVEDV, left ventricular end-diastolic volume; IVSs, interventricular septum during systole; IVSd, interventricular septum during diastole; LVPWs, posterior wall during systole; LVPWd, posterior wall during diastole; EF, ejection fraction; FS, fractional shortening; HR, heart rate.
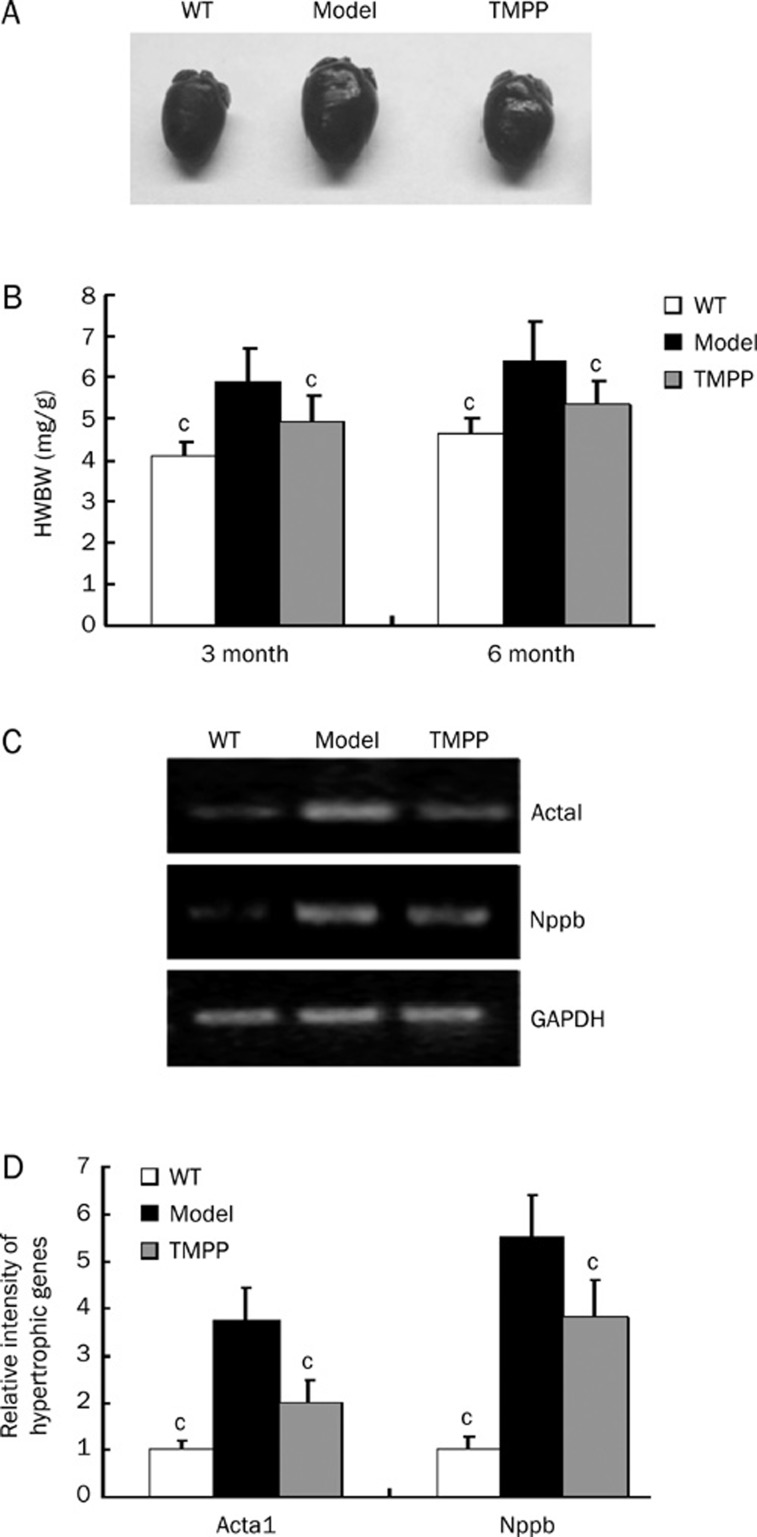
TMPP reduced cardiac hypertrophy
DCM hallmark is the combination of dystrophic and hypertrophic alterations of cardiomyocytes. There was an obvious increase in the size of heart in DCM (Figure 2A), and the HW/BW ratio of model group was increased by 43% (P<0.001) at 3 months and by 39% (P<0.001) at 6 months, and this change in heart weight due to DCM was reversed by treatment with TMPP by 55% (P<0.05) at 3 months and by 61% (P<0.05) at 6 months (Figure 2B). The expression levels of molecular markers of cardiac hypertrophy ACTA1 and NPPB were unregulated remarkably in the cTnTR141W mice aged 6 months, compared with WT mice, and decreased by 63% and 37%, respectively after TMPP treatment (Figure 2C, 2D).
Figure 2.
Effect of TMPP on cardiac hypertrophy in the cTnTR141W mice with DCM. (A) Whole-heart from WT, Model and TMPP mice aged 6 months. (B) HW/BW ratio in WT, Model and TMPP mice aged 3 months and 6 months. HW/BW, Heart weight/Body weight. n=7. cP<0.01 vs Model group. (C) Semiquantitative RT-PCR for expression levels of hypertrophic markers including ACTA1 and NPPB. The loading was normalized with GAPDH. (D) The intensities were quantified by densitometry using Bio-Rad Quantity One software. n=3. cP<0.01 vs Model group.
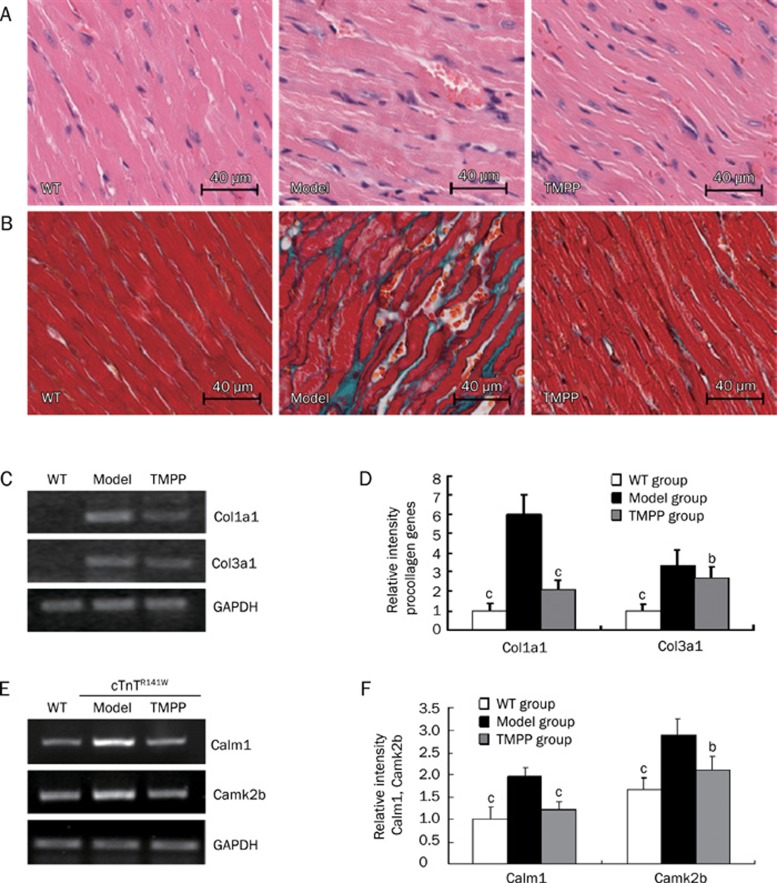
TMPP decreased myofibrillar disarray and interstitial fibrosis
Left ventricular systolic and diastolic dysfunction is related to the extent of myocardial disarray and interstitial fibrosis. HE staining showed that myofibrillar disarray was observed in the model group, but these pathological changes were ameliorated by the treatment with TMPP (Figure 3A). Masson staining showed that a large proportion of collagen was accumulated in interstitial space of the model heart, while the collagen deposition was decreased in TMPP-treated mice (Figure 3B). Expression levels of procollagens Col1a1 and Col3a1 in the model mice were decreased by 78% (P<0.01) and by 27% (P<0.05), respectively, after TMPP treatment, further substantiating the effect of TMPP in reducing collagen expression (Figure 3C, 3D).
Figure 3.
(A–D), Histological analysis of left ventricle. (A) HE staining of LV sections from WT, Model and TMPP mice aged 6 months. Original magnification, ×20. Scale bars=40 μm. (B) Masson's Trichrome staining of LV sections from WT, Model and TMPP mice aged 6 months. Myocytes stained red, collagenous tissue stained green. Original magnification, ×20. Scale bars=40 μm. (C) Semiquantitative RT-PCR for mRNA expression of Col1a1 and Col3a1 in the heart from WT, Model and TMPP mice aged 6 months. The loading was normalized with GAPDH. (D) The intensities were quantified by densitometry using Bio-Rad Quantity One software. n=3. bP<0.05, cP<0.01 vs Model group. (E−F), Effect of TMPP on the mRNA expression of Calm1 and Camk2b. (E) Semiquantitative RT-PCR for mRNA expression of Calm1, Camk2b in the heart from WT, Model and TMPP mice aged 6 months. The loading was normalized with GAPDH. n=3. (F) Relative gelatinolytic band Calm1 and Camk2b intensities were quantified by densitometry using Bio-Rad Quantity One software. bP<0.05, cP<0.01 vs Model group.
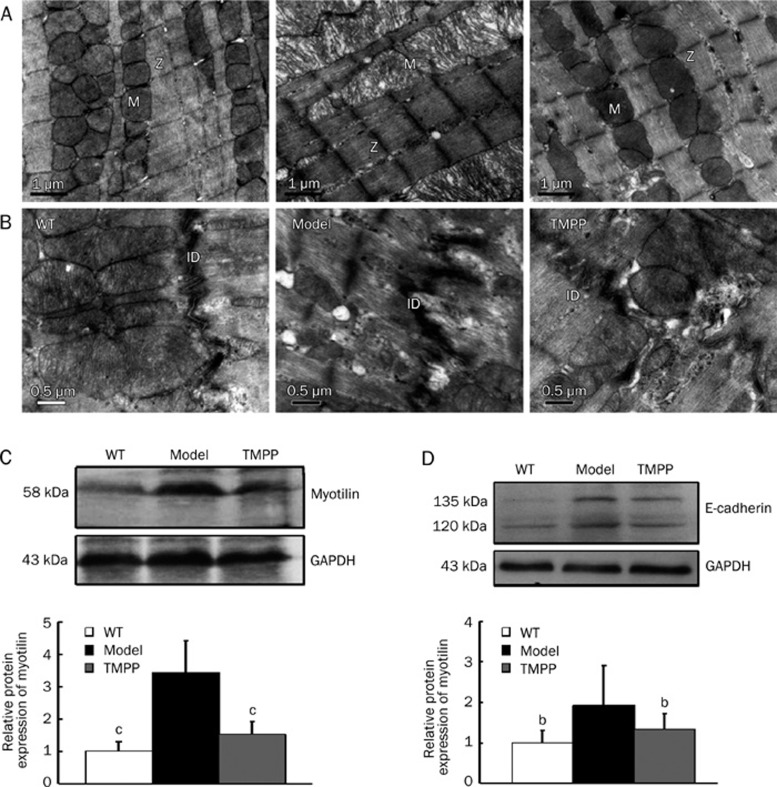
TMPP attenuated cardiac ultrastructural disruption
Transmission electron microscopy was used to examine the myocardial ultrastructure of hearts from 6-month-old mice (Figure 4A, 4B). In the model heart, lysed myofibrils were poorly organized with varying width and denser Z-lines compared with the WT heart, while the TMPP-treated heart showed regular arrangement of myofibrils with brighter dense Z-line (Figure 4A). Mitochondria were numerous between the parallel arrays of myofibrils in the WT and TMPP groups, while swollen mitochondria with loss of cristae and vacuolization were found in the model group (Figure 4A). Alteration of the specific cell contacts of cardiomyocytes, intercalated discs, was also observed. A higher degree of convolution of the membrane and even membrane detachment were quite reminiscent of intercalated disks in the model group, compared with the WT and TMPP groups (Figure 4B). Collectively, the disrupted myocardial ultrastructure in the cTnTR141W mice showed an evident alleviation upon treatment with TMPP.
Figure 4.
Representative transmission electron micrographs and the structural protein expression of LV in WT, Model and TMPP mice aged 6 months. (A) Sarcomere Z-disc (Z), mitochondria (M). Original magnification ×20 000. Scale bars=1 μm. (B) Intercalated discs (ID). Original magnification ×30 000. Scale bars=0.5 μm. (C) Western blot analysis for myotilin expression. GAPDH served as a loading control. The bands were quantified by densitometry using Bio-Rad Quantity One software. n=3. cP<0.01 vs Model group. (D) Western blot analysis for E-cadherin expression. GAPDH served as a loading control. The bands were quantified by densitometry using Bio-Rad Quantity One software. n=3. bP<0.05 vs Model group.
TMPP decreased the expression of myotilin and E-cadherin
Recent researches have proved that sarcomeric Z-disc proteins were related to the pathogenesis of dilated cardiomyopathy. Here we detected the expression of sarcomeric Z-line protein myotilin in the heart of 6-month-old mice. Western blot analysis indicated a significantly increased myotilin expression in the model heart compared with the WT heart, while decreased by 79% (P<0.01) in TMPP-treated mice (Figure 4C). Remodeling in intercalated discs also leads to dilated cardiomyopathy. We detected the expression of adherens junction structural component E-cadherin in the heart of 6-month-old mice. The expression of E-cadherin was increased remarkably in the model heart compared with the WT heart, and was decreased by 64% (P<0.05) after TMPP treatment (Figure 4D).
TMPP decreased the mRNA expression of Calm1 and Camk2b
CaM/CaMKII signaling pathway is always activated in patients and animal models of cardiomyopathy. Consistent with the previous investigations, CaM/CaMKII signaling pathway is also activated in the cTnTR141W transgenic mice of DCM, indicated by the significantly increased mRNA expression of Calm1 and Camk2b, which were respectively decreased by 77.9% and by 64.8% after TMPP treatment (Figure 3E, 3F).
Discussion
In the present study, using the cTnTR141W transgenic mouse model of FDCM, we examined the effects of TMPP on the progression of dilated cardiomyopathy. We first showed that long-term administration of TMPP to the cTnTR141W transgenic mice at 2 months of age, the early stage of DCM, prevented the development of DCM over a 7-months period of observation.
The chronic study clearly indicated that oral treatment with TMPP improved left ventricular function and survival in DCM. The missense mutation R141W in cTnT in mice leads to dilated cardiomyopathy, caused an increase in LVESV and LVEDV, resulting in a decrease in EF and FS, while TMPP administration produced decreases in both LVEDV and LVESV and thereby increased EF and FS. The IVSs and LVPWs in the Model group at 6 months decreased significantly, indicating a weak wall motion and systolic dysfunction, which was increased after TMPP treatment, but there was no statistical significance. These results proved that TMPP prevented the progressive LV dilation and systolic dysfunction in the development of DCM.
The pathologic characteristics of the heart in patients with DCM are severe interstitial fibrosis and myocyte hypertrophy with occasional disarray15, 16, 17. The extent of LV fibrosis is a strong predictor of cardiac death. Our study revealed that long-term TMPP treatment decreased interstitial fibrosis in the heart of cTnTR141W transgenic mouse, demonstrated by decreased total collagen deposition and reduced gene expression of procollagens COL1A1 and COL3A1. DCM hallmark is the combination of hypertrophic and dystrophic alterations of cardiomyocytes18. Cardiomyocyte hypertrophy involves the reactivation of embryonic gene expression, facilitating the maintenance of cardiac function in the short term, but gradually becomes maladaptive and leads to heart failure. TMPP ameliorated cardiomyocyte hypertrophy in the transgenic mouse of DCM, indicated by the decreased HW/BW ratio and the down-regulated expression of hypertrophic marker genes BNP and ACTA1 in the cTnTR141W transgenic mouse heart. In addition, histological analysis showed that TMPP attenuated myocyte disarray and ultrastructural disruption involving partially lysed myofibrils and swollen mitochondria in the model heart. The above results suggest that TMPP has beneficial effect on cardiac remodeling in cTnTR141W transgenic mouse, and all of these effects may contribute to improvement of cardiac function of the DCM model.
Sarcomeric Z-disc was related to the pathogenesis of dilated cardiomyopathy, supported by several transgenic animal models and the identification of mutations in various Z-disc proteins in patients19, 20, 21. We found the Z-lines of the sarcomere became denser in the cTnTR141W mouse, just like the change appeared in N-cadherin CKO mouse which also exhibit a DCM phenotype22. Then we detected the expression of Z-line protein myotilin, a candidate gene for DCM23, 24, and showed that its expression was significantly increased in our DCM model. Overexpression of wild-type myotilin in an limb-girdle muscular dystrophy type 1A (LGMD1A) mouse25, 26 showed more severe muscle degeneration compared with single-transgenic mutant mice27. Conversely, loss of myotilin does not cause alterations in the heart of newborn or adult myo−/− mice28. These findings suggested that strategies aimed at lowering total myotilin levels may be an effective therapeutic approach to the treatment of cardiomyopathy such as LGMD1A and FDCM carrying the cTnT (R141W) mutation. It is likely that the prevention of pathological development of DCM by TMPP was partially associated with depressing myotilin expression in cTnTR141W mice.
Remodeling in intercalated discs also leads to dilated cardiomyopathy29. Just like the pathological changes of intercalated discs found in the other two DCM models, MLP−/− (muscle LIM protein KO) and TOT (tropomodulin overexpression) mouse30, a higher degree of convolution of the membrane and even membrane detachment were quite reminiscent of intercalated discs in the cTnTR141 mouse. Components of the adherens junctions at the intercalated discs in MLP−/− and TOT mice are up-regulated. Cadherins are components of adherens junctions, N-cadherin is highly expressed by the developing and mature myocardium, while E-cadherin is distributed mainly in epithelia. A recent study showed that cardiac-specific expression of E-cadherin in adult transgenic mice led to DCM31, indicating that ectopic E-cadherin expression in the heart is not compatible with normal cardiac function. We detected significantly increased E-cadherin expression in the cTnTR141W mouse heart, which was reduced by TMPP treatment. Thus, the alleviation of cardiac dilation and dysfunction in the TMPP-treated mice was in part related to downregulation of E-cadherin expression in the heart.
Ca2+/CaM/CaMKII pathway is always activated in patients and animal models of cardiomyopathy32. Mice with cardiac overexpression of CaMKIIδc display DCM phenotype33, and CaMKII inhibition effectively protects against LV dilation and dysfunction caused by myocardial infarction and catecholamine toxicity34. Consistent with the previous investigations, the upregulated expression of calcium/calmodulin-regulated transcripts (Calm1 and Camk2b) is also observed in the cTnTR141W transgenic model. TMPP treatment inhibited the expression of Calm1 and Camk2b and may result in attenuation of cardiac remodeling and dysfunction in DCM.
In summary, TMPP significantly prevented the development of DCM due to R141W mutation in cTnT. The clinical significance of this finding is underscored by the lack of an effective pharmacological intervention to prevent FDCM in human patients. Present investigation raises the possibility for an early intervention, using a well-established pharmacological agent, in DCM mutation carriers to prevent the development of cardiac phenotype. Our results indicated the potential utility of TMPP in preventing FDCM, and it would be interesting to further test TMPP in other appropriate FDCM models.
Author contribution
Lian-feng ZHANG designed research; Hai-ping ZHAO performed research;, Dan LÜ analyzed data; Wei ZHANG, Li ZHANG and Shu-mei WANG breeded mice; Chuan QIN and Chun-mei MA contributed pathological analysis; Lian-feng ZHANG and Hai-ping ZHAO wrote the paper.
Acknowledgments
The authors thank Dr Harold JAMES for his patiently reading and correcting the English writing of this paper. The present work was supported in part by the National Commonwealth Institute Foundation (No DWS200704) and the Ministry of Health (No 200802036).
References
- Richardson P, Mckenna W, Bristow M, Maisch B, Mautner B, O'Connell J, et al. Report of the 1995 world health organization/international society and federation of cardiology task force on the definition and classification of cardiomyopathies. Circulation. 1996;93:841–2. doi: 10.1161/01.cir.93.5.841. [DOI] [PubMed] [Google Scholar]
- Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, et al. Contemporary definitions and classification of the cardiomyopathies. Circulation. 2006;113:1807–16. doi: 10.1161/CIRCULATIONAHA.106.174287. [DOI] [PubMed] [Google Scholar]
- Jessup M, Brozena S. Heart failure. N Engl J Med. 2003;348:2007–18. doi: 10.1056/NEJMra021498. [DOI] [PubMed] [Google Scholar]
- Kamisago M, Sharma SD, Depalma SR, Solomon S, Sharma P, McDonough B, et al. Mutations in sarcomere protein genes as a cause of dilated cardiomyopathy. N Engl J Med. 2000;343:1688–96. doi: 10.1056/NEJM200012073432304. [DOI] [PubMed] [Google Scholar]
- Towbin JA. The role of cytoskeletal proteins in cardiomyopathies. Curr Opin Cell Biol. 1998;10:131–9. doi: 10.1016/s0955-0674(98)80096-3. [DOI] [PubMed] [Google Scholar]
- Li D, Czernuszewicz GZ, Gonzalez O, Tapscott T, Karibe A, Durand JB, et al. Novel cardiac troponin T mutation as a cause of familial dilated cardiomyopathy. Circulation. 2001;104:2188–93. doi: 10.1161/hc4301.098285. [DOI] [PubMed] [Google Scholar]
- Juan F, Wei D, Xiongzhi Q, Ran D, Chunmei M, Lan H, et al. The changes of the cardiac structure and function in cTnTR141W transgenic mice. Int J Cardiol. 2008;128:83–90. doi: 10.1016/j.ijcard.2008.03.006. [DOI] [PubMed] [Google Scholar]
- Kwan CY, Daniel CE, Chen MC. Inhibition of vasoconstriction by tetramethylpyrazine: Does it act by blocking the voltage-dependent Ca2+ channel. J Cardiovasc Pharmacol. 1990;15:157–62. [PubMed] [Google Scholar]
- Sheu JR, Kan YC, Hung WC, Lin CH, Yen MH. The antiplatelet activity of tetramethylpyrazine is mediated through activation of NO synthase. Life Sci. 2000;67:937–47. doi: 10.1016/s0024-3205(00)00686-x. [DOI] [PubMed] [Google Scholar]
- Zhang Z, Wei T, Hou J, Li G, Yu S, Xin W. Tetramethylpyrazine scavenges superoxide anion and decreases nitric oxide production in human polymorphonuclear leukocytes. Life Sci. 2003;72:2465–72. doi: 10.1016/s0024-3205(03)00139-5. [DOI] [PubMed] [Google Scholar]
- Pang PK, Shan JJ, Chiu KW. Tetramethylpyrazine, a calcium antagonist. Planta Med. 1996;62:431–5. doi: 10.1055/s-2006-957933. [DOI] [PubMed] [Google Scholar]
- Hintz KK, Ren J. Tetramethylpyrazine elicits disparate responses in cardiac contraction and intracellular Ca2+ transients in isolated adult rat ventricular myocytes. Vasc Pharmacol. 2003;40:213–7. doi: 10.1016/j.vph.2003.08.002. [DOI] [PubMed] [Google Scholar]
- Chen KJ, Chen K. Ischemic stroke treated with Ligusticum chuanxiong. Chin Med J. 1992;105:870–3. [PubMed] [Google Scholar]
- Sutter MC, Wang YX. Recent cardiovascular drugs from Chinese medicinal plants. Cardiovasc Res. 1993;27:1891–901. doi: 10.1093/cvr/27.11.1891. [DOI] [PubMed] [Google Scholar]
- Aoki J, Ikari Y, Nakajima H, Mori M, Sugimoto T, Hatori M, et al. Clinical and pathologic characteristics of dilated cardiomyopathy in hemodialysis patients. Kidney International. 2005;67:333–40. doi: 10.1111/j.1523-1755.2005.00086.x. [DOI] [PubMed] [Google Scholar]
- Beltrami CA, Finato N, Rocco M, Feruglio GA, Puricelli C, Cigola E, et al. The cellular basis of dilated cardiomyopathy in humans. J Mol Cell Cardiol. 1995;27:291–305. doi: 10.1016/s0022-2828(08)80028-4. [DOI] [PubMed] [Google Scholar]
- Kajstura J, Zhang X, Liu Y, Szoke E, Cheng W, Olivetti G, et al. The cellular basis of pacing-induced dilated cardiomyopathy. Circulation. 1995;92:2306–17. doi: 10.1161/01.cir.92.8.2306. [DOI] [PubMed] [Google Scholar]
- Branishte TA, Dudnakova TV, Dergilev KV, Severin VV, Naumov VG, Shirinski VP, et al. Expression of contractile and cytoskeletal proteins in myocardium of patients with dilated cardiomyopathy. Kardiologiia. 2004;44:31–6. [PubMed] [Google Scholar]
- Arber S, Hunter JJ, Ross J Jr, Hongo M, Sansig G, Borg J, et al. MLP-deficient mice exhibit a disruption of cardiac cytoarchitectural organization, dilated cardiomyopathy, and heart failure. Cell. 1997;88:393–403. doi: 10.1016/s0092-8674(00)81878-4. [DOI] [PubMed] [Google Scholar]
- Hayashi T, Arimura T, Itoh-Satoh M, Ueda K, Hohda S, Inagaki N, et al. Tcap gene mutations in hypertrophic cardiomyopathy and dilated cardiomyopathy. J Am Coll Cardiol. 2004;44:2192–201. doi: 10.1016/j.jacc.2004.08.058. [DOI] [PubMed] [Google Scholar]
- Duboscq-Bidot L, Xu P, Charron P, Neyroud N, Dilanian G, Millaire A, et al. Mutations in the Z-band protein myopalladin gene and idiopathic dilated cardiomyopathy. Cardiovasc Res. 2008;77:118–25. doi: 10.1093/cvr/cvm015. [DOI] [PubMed] [Google Scholar]
- Kostetskii I, Li J, Xiong Y, Zhou R, Ferrari VA, Patel VV, et al. Induced deletion of the N-cadherin gene in the heart leads to dissolution of the intercalated disc structure. Circ Res. 2005;96:346–54. doi: 10.1161/01.RES.0000156274.72390.2c. [DOI] [PubMed] [Google Scholar]
- Salmikangas P, van der Ven PF, Lalowski M, Taivainen A, Zhao F, Suila H, et al. Myotilin, the limb-girdle muscular dystrophy 1A (LGMD1A) protein, cross-links actin filaments and controls sarcomere assembly. Hum Mol Genet. 2003;12:189–203. doi: 10.1093/hmg/ddg020. [DOI] [PubMed] [Google Scholar]
- Zeller R, Ivandic BT, Ehlermann P, Mucke O, Zugck C, Remppis A, et al. Large-scale mutation screening in patients with dilated or hypertrophic cardiomyopathy: a pilot study using DGGE. J Mol Med. 2006;84:682–91. doi: 10.1007/s00109-006-0056-2. [DOI] [PubMed] [Google Scholar]
- Hauser MA, Horrigan SK, Salmikangas P, Torian UM, Viles KD, Dancel R, et al. Myotilin is mutated in limb girdle muscular dystrophy 1A. Hum Mol Genet. 2000;9:2141–7. doi: 10.1093/hmg/9.14.2141. [DOI] [PubMed] [Google Scholar]
- Hauser MA, Conde CB, Kowaljow V, Zeppa G, Taratuto AL, Torian UM, et al. Myotilin mutation found in second pedigree with LGMD1A. Am J Hum Genet. 2002;71:1428–82. doi: 10.1086/344532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garvey SM, Liu Y, Miller SE, Hauser MA. Myotilin overexpression enhances myopathology in the LGMD1A mouse model. Muscle Nerve. 2008;37:663–7. doi: 10.1002/mus.20994. [DOI] [PubMed] [Google Scholar]
- Moza M, Mologni L, Trokovic R, Faulkner G, Partanen J, Carpén O. Targeted deletion of the muscular dystrophy gene myotilin does not perturb muscle structure or function in mice. Mol Cell Biol. 2007;27:244–52. doi: 10.1128/MCB.00561-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perriard JC, Hirschy A, Ehler E. Dilated cardiomyopathy: a disease of the intercalated disc. Trends Cardiovasc Med. 2003;13:30–8. doi: 10.1016/s1050-1738(02)00209-8. [DOI] [PubMed] [Google Scholar]
- Ehler E, Horowits R, Zuppinger C, Price RL, Perriard E, Leu M, et al. Alterations at the intercalated disk associated with the absence of muscle LIM protein. J Cell Biol. 2001;153:763–72. doi: 10.1083/jcb.153.4.763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferreira-Cornwell MC, Luo Y, Narula N, Lenox JM, Lieberman M, Radice GL. Remodeling the intercalated disc leads to cardiomyopathy in mice misexpressing cadherins in the heart. J Cell Sci. 2002;115:1623–34. doi: 10.1242/jcs.115.8.1623. [DOI] [PubMed] [Google Scholar]
- Zhang T, Brown JH. Role of Ca2+/calmodulin-dependent protein kinase II in cardiac hypertrophy and heart failure. Cardiovasc Res. 2004;63:476–86. doi: 10.1016/j.cardiores.2004.04.026. [DOI] [PubMed] [Google Scholar]
- Maier LS, Zhang T, Chen L, DeSantiago J, Brown JH, Bers DM. Transgenic CaMKIIδC overexpression uniquely alters cardiac myocyte Ca2+ handling: reduced SR Ca2+ load and activated SR Ca2+ release. Circ Res. 2003;92:904–11. doi: 10.1161/01.RES.0000069685.20258.F1. [DOI] [PubMed] [Google Scholar]
- Zhang R, Khoo MS, Wu Y, Yang Y, Grueter CE, Ni G, et al. Calmodulin kinase II inhibition protects against structural heart disease. Nat Med. 2005;11:409–17. doi: 10.1038/nm1215. [DOI] [PubMed] [Google Scholar]