As one of the facultatively intracellular bacteria, Salmonella enterica serovar Typhimurium (S. Typhimurium) replicates even in macrophages and induces their death—a key strategy of this virulent pathogen in evading the innate immune response.1 The underlying mechanisms are well known but complex, because many pathways are involved, such as apoptosis,2 necrosis,3 pyroptosis4 and autophagy.5 In a recent issue of Nature Immunology, Robinson et al. reveal a novel pathway for killing macrophages during infection: type 1 interferon- (IFN-α and IFN-β) associated necroptosis.6
To understand the novel pathway better, it is important to clarify the major differences between necrosis, apoptosis, autophagy, necroptosis and pyroptosis, and to recognize the various roles of IFN-α or IFN-β. The four pathways of programmed cell death (apoptosis, autophagy, necroptosis and pyroptosis) are compared in Table 1.
Table 1. Comparison of four major pathways of programmed cell death.
| Apoptosis | Autophagy | Necroptosis | Pyroptosis | |
|---|---|---|---|---|
| Meaning in Greek | apo-: off- ptosis: falling | auto-: self- phagy: eating | nekro-: corpse | pyro-: fire/fever |
| Year first introduced | 197221 | 19637 | 20059 | 200116 |
| Morphological feature | ||||
| Cell and organelle swelling | No | No | Yes | Yes |
| Membrane blebbing | Yes | Yes | No | No |
| Rapid membrane rupture | No | No | Yes | Yes |
| Nuclear condensation | Yes | Yes | No | No |
| DNA laddering | Yes | ? | Random degradation | Random degradation |
| Apoptotic bodies | Yes | No | No | No |
| Autophagic vesicles | No | Yes | No | No |
| Biochemical feature | ||||
| Caspase-1 dependence | No | No | No | Yes |
| Caspase-3 activation | Yes | No | No | No |
| RIP-1 and -3 kinase activation | No | No | Yes | No |
| Cytochome-c release | Yes | No | No | No |
| MAP1LC3 lipidation | No | Yes | No | No |
| SQSTM1 degradation | No | Yes | No | No |
| Target for genetic inhibition | Caspases-3, -8, -9 | Atg-5, -6, -7 etc. | RIP-1 and RIP-3 | Caspase-1 |
| Specific inhibitor | Z-VAD-fmk | VPS34 inhibitors | Necrostatin-1 | Ac-YVAD-CHO |
| Inflammation | No | No | Yes | Yes |
Abbreviations: Ac-YVAD-CHO, Acetyl-Tyr-Val-Ala-Asp-aldehyde; Atg, autophagy (autophagy-related genes); MAP1LC3, microtubule-associated protein 1 light chain 3; RIP-1, receptor-interacting protein 1 kinase; SQSTM1, sequestosome 1; VPS34 inhibitors, inhibitors of class III phosphoinositide 3-kinase (PI3K), e.g., wortmannin and 3-methyladenine; Z-VAD-fmk, N-benzyloxycarbonyl-Val-Ala-Asp-fluoromethylketone.
For decades, necrosis and apoptosis have been the central paradigms for cell death. Apoptosis is programmed cell death characterized by activation of caspases in an extrinsic or intrinsic pathway and by the quiet elimination of unwanted cells without causing an inflammatory response. Apoptotic cells show cell membrane blebbing, cell shrinkage, chromatin condensation, ordered DNA fragmentation and formation of apoptotic bodies. On the other hand, necrosis represents accidental and uncontrolled passive cell death induced by, for example, severe heat shock, ischemia or mechanical rupture. Necrotic cells show an increase in cell volume, swelling of organelles, plasma membrane rupture and eventual release of intracellular contents with an inflammatory response as a consequence.
Despite the widespread use of apoptosis as a synonym for programmed cell death, accumulated evidence clearly shows that there are other types of programmed cell death as well. Non-apoptotic pathways include, at least, autophagy, necroptosis and pyroptosis.
The term autophagy was invented by Christian de Duve in 1963 to describe the ability of cells to digest their own cellular components.7 Today, autophagy represents a self-digesting mechanism in which cytoplasmic components are engulfed by autophagosomes and degraded in lysosomes.8 Autophagy has become increasingly recognized as a pathway of programmed cell death. However, autophagy can serve both pro-survival and pro-death functions. Autophagic cells are morphologically characterized by massive vacuolization due to the formation of many autophagic vesicles. As in apoptosis, autophagic cells can ultimately be removed by phagocytosis without causing inflammation.
The term necroptosis was proposed by Degterev et al.9 to distinguish programmed necrosis of ischemic cells from accidental, uncontrolled necrosis, and from apoptosis. Cells undergoing necroptosis have morphological features of necrosis. However, necroptosis requires a unique signaling pathway associated with the activation of receptor-interacting protein (RIP) kinases.10 RIP-1 and RIP-3 form a phosphorylation complex to activate necroptosis,11 and RIP-1 kinase can be specifically inhibited by necrostatin-1. Necroptosis can be initiated by tumor-necrosis factor α (TNF-α), Fas ligand and TNF-related apoptosis-inducing ligand, which are also well known to activate apoptosis. Inhibition of apoptotic caspases, particularly caspase-8, moves the cells towards necroptosis in response to TNF-α.12,13 RIP-1-deficient mice display extensive apoptosis, dying soon after birth.14 Necroptosis is now considered a major cell death pathway that is as important as apoptosis in regulating development and immunity.15 However, the biological role of necroptosis during infection requires further study.
The term ‘pyroptosis' was introduced by Cookson and Brennan to describe the atypical death of macrophages infected with S. Typhimurium.16 Pyroptosis is also referred to as pro-inflammatory programmed cell death, because caspase-1 and IL-1β cause fever and inflammation.4,16 Pyroptotic cell death is mechanistically distinct from apoptosis and necroptosis, and may be classified as programmed necrosis.
Caspases are cysteine proteases that play essential roles in apoptosis, necrosis and inflammation. They can be roughly divided into apoptotic caspases and inflammatory caspases, of which the former includes apoptosis initiator caspases (-2, -8, -9 and -10) and executioner caspases (-3, -6 and -7). Activation of this cascade is a biochemical hallmark of apoptosis. The latter includes caspase-1, -4, -5, -13 and -14. In contrast to apoptosis, pyroptosis is triggered by caspase-1, which is activated in a large supramolecular complex termed the inflammasome. Caspase-1 is also an IL-1β-converting enzyme (ICE), hydrolyzing pro-IL-1β and pro-IL-18 into active IL-1β and IL-18. Unlike necroptosis, RIP-1 and -3 kinases are not involved in pytoptosis. Pyroptotic cells release not only intracellular contents, but also pro-inflammatory cytokines IL-1β and IL-18.
Type 1 IFNs play various roles in bacterial infections, e.g., protection from infections by extracellular bacteria17 such as group B streptococci, pneumococci and Escherichia coli, and exacerbation of infections by intracellular bacteria such as Listeria monocytogenes.18,19 The involvement of the IFN-α/IFN-β cascade in response to intracellular bacteria in phagocytes has been well established; however, the mechanism(s) is still largely unknown.
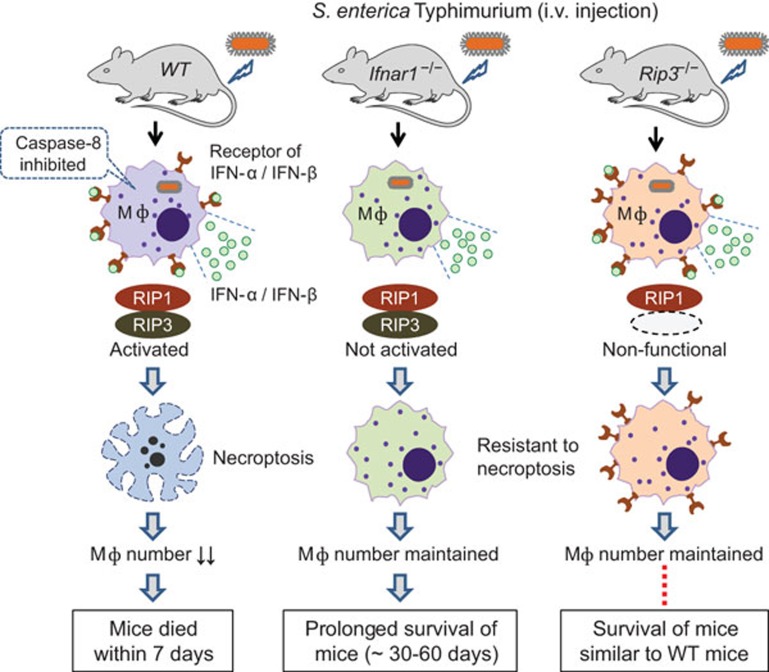
Robinson et al.'s paper adds IFN-α/IFN-β-associated necroptosis to the list of mechanisms that S. Typhimurium uses to kill macrophages.6 By comparing the survival and bacterial burden in S. Typhimurium-infected wild-type C57BL/6J mice, Ifnar1−/− mice (deficient in the receptor for IFN-α and IFN-β) and Rip3−/− mice (deficient in RIP-3 kinase), Robinson et al. have demonstrated for the first time that S. Typhimurium induces autocrine secretion of IFN-β, which drives necroptosis of macrophages. As a result, mice die within 7 days owing to the impairment of innate immune defense. Stimulation of uninfected macrophages with IFN-α or IFN-β alone neither activates RIP-1/RPI-3 nor elicits necroptosis. Consistent with the previous report that necroptosis is triggered only when caspase-8 activity is blocked,13 S. Typhimurium infection in macrophages downregulates caspase-8. This down-regulation allows IFN-α/IFN-β to induce the formation of the RIP-1/RPI-3 complex, which then promotes necroptosis. This mechanism is confirmed by knockdown of the gene encoding RIP-3, by adding neutralizing antibody against IFN-β, and by adding IFN-β in combination with Z-VAD-fmk (caspase-8 inhibitor) and/or necrostatin-1 (RIP-1 kinase inhibitor). The possibilities of impaired cytokine expression or inflammasome activation in Ifnar1−/− and RIP-3−/− macrophages have been convincingly excluded by a series of experiments. Addition of the caspase-1 inhibitor YVAD-CHO partially improved survival of infected wild-type macrophages, suggesting a coexistence of necroptosis and caspase-1-associated pyroptosis.
The main approaches of this study are outlined in Figure 1. There is an obvious discrepancy in the in vivo and in vitro data from Rip3−/− mice, which did not show substantial differences in survival relative to wild-type mice, but Ifnar1−/− mice survived an extra 30–60 days. However, macrophages from Rip3−/− mice behaved like those from Ifnar1−/− mice: they resisted necroptosis in vitro, and enhanced control of S. Typhimurium in vivo when transferred into wild-type hosts. It may be important to address this discrepancy and identify the underlying mechanisms. It would be interesting to know whether other intracellular bacteria, such as Mycobacterium tuberculosis and Legionella pneumophila, also use the necroptosis pathway to kill macrophages, and thus escape innate immune defenses.
Figure 1.
Type 1 interferon-dependent induction of macrophage necroptosis is a major mechanism by which the pathogen deactivates the innate immune responses in mice infected with S. Typhimurium. IFN, interferon; Ifnar1−/− mouse, deficient in the receptor for IFN-α and IFN-β Mφ, macrophage; Rip3−/− mouse, deficient in RIP-3 kinase; WT mouse, wild-type C57BL/6J.
Taken together, the above findings add a novel pathway, necroptosis, to the list of pathways by which S. Typhimurium kills macrophages. Multiple pathways of cell death exist simultaneously in macrophages exposed to S. Typhimurium, where a complex interplay between apoptosis, autophagy, necroptosis and pyroptosis has been observed. Inhibition of a dominant molecule for a pathway may lead to a different type of cell death. As described above, for example, IFN-α/IFN-β-induced necroptosis depends on inhibition of apoptotic caspase-8, either by intracellular infection with S. Typhimurium, or by adding the specific caspase inhibitor Z-VAD-fmk. The death receptors related to the TNF family can trigger apoptosis, but when apoptosis is blocked, they trigger necroptosis.12,13 Salmonella infection of caspase-1-deficient macrophages bypasses pyroptosis, but results in an autophagic cell death.3,5 In L929 cells, knockdown of autophagy-related genes such as Atg-6 (beclin-1) and Atg-7 also inhibits necroptosis.20
During infection, apoptotic and autophagic cells are removed quietly so that other tissues are less impaired by inflammation. Necroptotic and pytoptotic cells can trigger an inflammatory response that can help the organism to eliminate the pathogen. A range of cell death pathways is needed to orchestrate the protective and homeostatic functions of the immune system, with important questions remaining concerning which pathway will predominate and what the switch is. In infections caused by intracellular bacteria, further research is needed to define the biological and immunological significance of these different pathways, and their contribution to pathogenesis.
References
- Lindgren SW, Stojiljkovic I, Heffron F. Macrophage killing is an essential virulence mechanism of Salmonella typhimurium. . Proc Natl Acad Sci USA. 1996;93:4197–4201. doi: 10.1073/pnas.93.9.4197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hersh D, Monack DM, Smith MR, Ghori N, Falkow S, Zychlinsky A. The Salmonella invasin SipB induces macrophage apoptosis by binding to caspase-1. Proc Natl Acad Sci USA. 1999;96:2396–2401. doi: 10.1073/pnas.96.5.2396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hueffer K, Galan JE. Salmonella-induced macrophage death: multiple mechanisms, different outcomes. Cell Microbiol. 2004;6:1019–1025. doi: 10.1111/j.1462-5822.2004.00451.x. [DOI] [PubMed] [Google Scholar]
- Brennan MA, Cookson BT. Salmonella induces macrophage death by caspase-1-dependent necrosis. Mol Microbiol. 2000;38:31–40. doi: 10.1046/j.1365-2958.2000.02103.x. [DOI] [PubMed] [Google Scholar]
- Hernandez LD, Pypaert M, Flavell RA, Galan JE. A Salmonella protein causes macrophage cell death by inducing autophagy. J Cell Biol. 2003;163:1123–1131. doi: 10.1083/jcb.200309161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson N, McComb S, Mulligan R, Dudani R, Krishnan L, Sad S. Type I interferon induces necroptosis in macrophages during infection with Salmonella enterica serovar Typhimurium. Nat Immunol. 2012;13:954–962. doi: 10.1038/ni.2397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klionsky DJ. Autophagy revisited: a conversation with Christian de Duve. Autophagy. 2008;4:740–743. doi: 10.4161/auto.6398. [DOI] [PubMed] [Google Scholar]
- Klionsky DJ, Emr SD. Autophagy as a regulated pathway of cellular degradation. Science. 2000;290:1717–1721. doi: 10.1126/science.290.5497.1717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Degterev A, Huang Z, Boyce M, Li Y, Jagtap P, Mizushima N, et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat Chem Biol. 2005;1:112–119. doi: 10.1038/nchembio711. [DOI] [PubMed] [Google Scholar]
- Holler N, Zaru R, Micheau O, Thome M, Attinger A, Valitutti S, et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat Immunol. 2000;1:489–495. doi: 10.1038/82732. [DOI] [PubMed] [Google Scholar]
- He S, Wang L, Miao L, Wang T, Du F, Zhao L, et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell. 2009;137:1100–1111. doi: 10.1016/j.cell.2009.05.021. [DOI] [PubMed] [Google Scholar]
- Vercammen D, Beyaert R, Denecker G, Goossens V, van Loo G, Declercq W, et al. Inhibition of caspases increases the sensitivity of L929 cells to necrosis mediated by tumor necrosis factor. J Exp Med. 1998;187:1477–1485. doi: 10.1084/jem.187.9.1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vandenabeele P, Declercq W, van Herreweghe F, Vanden Berghe T. The role of the kinases RIP1 and RIP3 in TNF-induced necrosis. Sci Signal. 2010;3:re4. doi: 10.1126/scisignal.3115re4. [DOI] [PubMed] [Google Scholar]
- Kelliher MA, Grimm S, Ishida Y, Kuo F, Stanger BZ, Leder P. The death domain kinase RIP mediates the TNF-induced NF-kappaB signal. Immunity. 1998;8:297–303. doi: 10.1016/s1074-7613(00)80535-x. [DOI] [PubMed] [Google Scholar]
- Lu JV, Walsh CM. Programmed necrosis and autophagy in immune function. Immunol Rev. 2012;249:205–217. doi: 10.1111/j.1600-065X.2012.01147.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cookson BT, Brennan MA. Pro-inflammatory programmed cell death. Trends Microbiol. 2001;9:113–114. doi: 10.1016/s0966-842x(00)01936-3. [DOI] [PubMed] [Google Scholar]
- Mancuso G, Midiri A, Biondo C, Beninati C, Zummo S, Galbo R, et al. Type I IFN signaling is crucial for host resistance against different species of pathogenic bacteria. J Immunol. 2007;178:3126–3133. doi: 10.4049/jimmunol.178.5.3126. [DOI] [PubMed] [Google Scholar]
- Auerbuch V, Brockstedt DG, Meyer-Morse N, O'Riordan M, Portnoy DA. Mice lacking the type I interferon receptor are resistant to Listeria monocytogenes. . J Exp Med. 2004;200:527–533. doi: 10.1084/jem.20040976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Connell RM, Saha SK, Vaidya SA, Bruhn KW, Miranda GA, Zarnegar B, et al. Type I interferon production enhances susceptibility to Listeria monocytogenes infection. J Exp Med. 2004;200:437–445. doi: 10.1084/jem.20040712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu L, Alva A, Su H, Dutt P, Freundt E, Welsh S, et al. Regulation of an ATG7-beclin 1 program of autophagic cell death by caspase-8. Science. 2004;304:1500–1502. doi: 10.1126/science.1096645. [DOI] [PubMed] [Google Scholar]
- Kerr JF, Wyllie AH, Currie AR. Apoptosis: a basic biological phenomenon with wide-ranging implications in tissue kinetics. Br J Cancer. 1972;26:239–257. doi: 10.1038/bjc.1972.33. [DOI] [PMC free article] [PubMed] [Google Scholar]