Abstract
Leukotrienes (LTs) are lipid mediators derived from the 5-lipoxygenase (5-LO) pathway of arachidonic acid metabolism and are markers and mediators of pulmonary inflammation. Research over the past two decades has established that LTs modulate inflammation in pulmonary arterial hypertension (PAH). The purpose of this review was to summarize the current knowledge of LTs in the pathophysiology of PAH and to highlight a recent study that advances our understanding of how leukotriene B4 (LTB4) specifically contributes to pulmonary vascular remodeling. The results of these studies suggest that pharmacological inhibition of LT pathways, especially LTB4, has high potential for the treatment of PAH.
Keywords: Leukotriene, Pulmonary arterial hypertension, Vascular remodeling, Inflammation, 5-Lipoxygenase
Introduction
Pulmonary arterial hypertension (PAH) is a life-threatening disease characterized by vasoconstriction and vascular remodeling that result in increased pulmonary vascular resistance and pulmonary arterial pressure [1–3]. Advances in the understanding of the pathobiology of PAH have led to the development of a number of effective vasodilating therapies, such as endothelin receptor antagonists, phosphodiesterase type 5 inhibitors, and prostacyclin or analogs. However, even in the current treatment era, the average life expectancy of patients with PAH remains poor and is estimated to be 5–7 years after diagnosis [4]; new approaches are clearly needed. Perivascular inflammation is common in PAH and is characterized by the presence of various immune cells, including T cells, B cells, plasma cells, mast cells, dendritic cells, and macrophages, as well as inflammatory molecules, such as cytokines, chemokines, growth factors, eicosanoids, reactive oxygen, and nitrogen species [5, 6]. Recent preclinical studies demonstrate that abnormal regulatory T cell (Treg) activity exacerbates inflammation associated with pulmonary vascular injury and facilitates disease development [7, 8]. In PAH arising in conditions associated with immune dysregulation, the role of leukotrienes (LTs) appears to be an exciting new target for disease intervention that could complement conventional vasodilator therapy.
LTs are lipid mediators derived from the polyunsaturated fatty acid and arachidonic acid. Their function is in initiating and amplifying both the innate and adaptive immune responses by regulating the recruitment and activation of leukocytes in inflamed tissues [9, 10]. In this review, we will discuss the current understanding of how LTs are involved in several aspects of the pathogenesis of PAH.
Overview of LT synthesis and actions
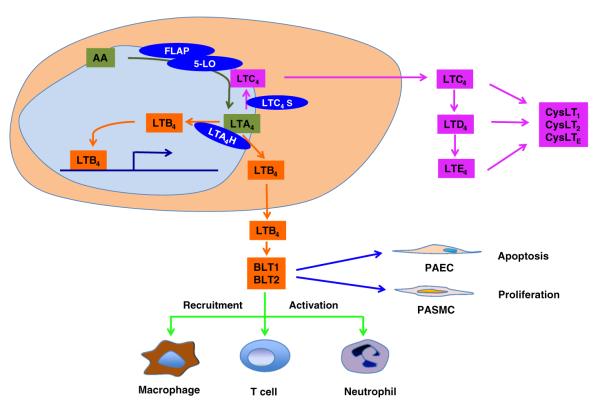
LTs are synthesized primarily in leukocytes at the Golgi apparatus and endoplasmic reticulum (ER)/nuclear membrane. At these sites, activated phospholipase A2 (PLA2), especially cytosolic PLA2, hydrolyzes membrane phospholipids and liberates arachidonic acid from the membrane bilayer. 5-LO in the cytosol or nucleus is subsequently activated and translocates to the inner and outer nuclear membrane initiating the synthesis of LTs by converting arachidonic acid to 5-hydroperoxyeicosatetraenoic acid (5-HPETE) and LTA4. This reaction requires the integral nuclear envelope protein 5-lipoxygenase-activating protein (FLAP) [11–13]. LTA4 has a half-life of less than 3 s at physiological pH [14] and is quickly either conjugated to glutathione by LTC4 synthase (LTC4S) to form LTC4 or hydrolyzed by LTA4 hydrolase (LTA4H) to generate LTB4 [15–19]. Both LTC4 and LTB4 can be transported out of the source cell into the extracellular milieu. LTB4 may also potentially act in the nucleus as a modulator of transcription. LTC4 undergoes sequential peptide cleavage of the glutathione moiety to form LTD4 or LTE4 [20–23]. LTC4, LTD4, and LTE4, as a group, are named the cysteinyl leukotrienes (CysLTs) (Fig. 1).
Fig. 1.
LT pathways and functions in PAH. LTs are synthesized from the arachidonic acid (AA) pathway, where 5-LO works together with FLAP on the perinuclear membrane that converts AA to LTA4. LTA4 is quickly metabolized to LTB4 by LTA4H, or is converted to LTC4 by LTC4S. LTC4 undergoes sequential peptide cleavage of the glutathione moiety to form LTD4 or LTE4. LTB4 may function as a transcriptional regulator in the nucleus or is transported out from the source cell and binding to its cognate receptors (BLT1 and BLT2) to initiate the downstream signaling. In PAH, elevated LTB4 signaling around the disease arteriole results in the recruitment of the leukocytes. Recent data demonstrate that LTB4 may also cause vascular remodeling by inducing the PAEC apoptosis and PASMC proliferation
Different leukocytes generate different LT profiles: neutrophils synthesize exclusively LTB4 [24, 25], eosinophils, and mast cells predominantly LTC4 [26–30], while macrophages generate both [31–33]. The actions of LTs are mediated through a series of G-protein-coupled receptors. These cell-surface receptors are classified into three groups: receptors for LTB4 (BLT1, BLT2), receptors for CysLTs (CysLT1, CysLT2) [34], and a newly discovered receptor specific for LTE4 known as CysLTE [35, 36]. The proinflammatory effects of LTB4 in leukocytes are mainly transduced through the high-affinity receptor, BLT1, while the role of the lower-affinity BLT2 receptor remains unknown. The CysLTs are known to cause long-lived bronchoconstriction mediated by the CysLT1 receptor. This receptor recognizes ligands with the following rank order of the affinity: LTD4 > LTC4 = LTE4. CysLT2 recognizes its ligands with following order of affinity: LTC4 = LTD4 < LTE4 [37–40].
The role of the 5-LO/LT pathway in PAH
5-LO and FLAP expression in animal models and in humans with PAH
The correlation between 5-LO and PAH was first noted by Voelkel et al. in 1996; they found that both alveolar macrophages and vascular endothelial cells from rats exposed to chronic hypoxia had increased expression of 5-LO [41]. The lungs of chronically hypoxic rats showed increased translocation of 5-LO from the cytosol to the perinuclear membrane, indicating a higher level of 5-LO activation. Treatment with the FLAP inhibitor, MK-886, an antagonist for 5-LO and FLAP interaction, inhibited hypoxia-induced pulmonary vasoconstriction and prevented the development of chronic hypoxic pulmonary hypertension (PH) in rats (By convention, animal models of PAH are referred to as ‘PH’). Additionally, compared to controls, 5-LO knockout mice exposed to chronic hypoxia had attenuated right ventricle hypertrophy, as measured by the right ventricle (RV) over left ventricle (LV) plus septum (S) ratio (RV/LV + S). This was the first study to suggest that 5-LO is involved in the regulation of pulmonary vascular tone and plays a role in the pathogenesis of PH in chronic hypoxia rodent models.
The Denver group then went on to examine the expression of 5-LO and FLAP in the lungs of patients with PAH. They found that compared to healthy controls, patients with PAH had elevated protein levels of both 5-LO and FLAP demonstrated by immunohistochemistry and increased 5-LO mRNA levels measured by in situ hybridization [42]. 5-LO and FLAP were prominent in alveolar macrophages clustered around plexiform lesions. Additionally, in the lungs of PAH patients, the endothelial cells in plexiform and concentric lesions expressed both 5-LO and FLAP.
While 5-LO overexpression in rat lungs using a recombinant adenovirus expressing 5-LO (Ad5-LO) did not cause PH in normal rats, it markedly accelerated the progression of PH in rats treated with monocrotaline (MCT) [43]. An increase in pulmonary artery pressure occurred earlier in the rats treated with MCT + Ad5-LO (7–10 days) compared with those treated with control vector, or MCT alone (15–18 days). Lung tissue histological sections from MCT + Ad5-LO rats exhibited severe inflammation and pulmonary vascular muscularization. Treatment of 5-LO inhibitors, zileuton or MK-886, prevented either MCT- or MCT + Ad5-LO induced PH. These data suggested that 5-LO plays a critical role in the progression of PH in the setting of pulmonary vascular endothelial cell dysfunction [43].
BMPR2 and 5-LO in PH
Heterozygous germline mutations in the bone morphogenetic protein type 2 receptor (BMPR2) account for approximately 80 % of patients with familial PAH [44, 45]. The disease is transmitted in an autosomal dominant fashion, but has only a 20 % penetrance, which suggests that additional triggers are needed to develop the disease. Additionally, it is estimated that 15–40 % of patients with idiopathic PAH may also have mutations in BMPR2 [45–47]. The identification of this genetic mutation in PAH has provided a focus for studying the complex pathobiology of this disease.
Under unstressed conditions, BMPR2+/− mice were found to have the same life span, right ventricular systolic pressure (RVSP), and lung histology as those of wild-type mice. However, intratracheal instillation of Ad5-LO significantly increased the RVSP of BMPR2+/− mice compared to the wild-type mice. Also, modest but significant muscularization of distal pulmonary arterioles was discovered 4 weeks after Ad5-LO treatment [48]. This study showed that under the inflammatory insult triggered by 5-LO, BMPR2+/− mice were more susceptible to an increase in RVSP and vascular remodeling than wild-type mice, suggesting a critical role for 5-LO in BMPR2 haploinsufficiency and the development of PAH.
LTB4 in PAH
Several prior observations suggested that LTB4 may contribute to the pathogenesis of PAH. First, LTB4 has long been recognized as a chemoattractant for many inflammatory cell types observed in PAH, such as T and B lymphocytes, macrophages, and neutrophils. Next, it is known that the phosphorylation of 5-LO (on the Ser271 residue by p38 MAPK) significantly increases its enzyme activity in vitro [49, 50] and, thus, facilitates 5-LO nuclear retention which favors LTB4 biosynthesis [51, 52]. Indeed, increased LTB4 and myeloperoxidase (MPO) level was observed within 3 days after MCT injection in rats. Administration of ONO4057, a BLT1 antagonist, reduced right ventricular hypertrophy induced by MCT and prevented these animals from developing PH [53].
Recently, our group showed that in both the SU5416 (SU; a VEGFR2 inhibitor)-treated athymic rat model of severe PH associated with an absence of T cell-mediated regulation and in the lungs of PAH patients, the expression of biosynthetic enzymes for LTB4 production, specifically phospho-Ser271 5-LO (p5-LO) and LTA4H, were elevated compared to controls [54]. p5-LO and LTA4H expression were prominent in macrophages around partially or fully occluded small-tomid-sized pulmonary arterioles. Upregulation of these enzymes correlated with high levels of LTB4 in the lung bronchoalveoloar fluid as in the systemic circulation. A detailed morphometric assessment of these macrophages demonstrated that macrophages closest to diseased arterioles had the highest expression of p5-LO, suggesting that LTB4-producing cells were in close proximity to the site of disease, possibly playing a role in the observed vascular remodeling.
Because pulmonary arterial endothelial cell (PAEC) injury is regarded as an important early event in PH pathogenesis, we established a macrophage-PAEC co-culture system to determine whether macrophages, specifically p5-LO positive, LTB4-producing macrophages, could injure adjacent endothelial cells. We found that macrophages isolated from the lungs of SU-treated athymic rats with PH induced significant endothelial cell apoptosis. Further, macrophages from healthy rats that were transfected with S271E 5-LO, a plasmid that produces a 5-LO phosphorylation mimic mutant, caused marked PAEC death to a similar degree. Furthermore, LTB4 cultured with PAEC alone demonstrated a capacity to injure the cells in a dose-dependent fashion. Blocking BLT1, the major receptor for LTB4 on endothelial cells, prevented LTB4-mediated apoptosis [54].
To explain our findings of LTB4-induced PAEC apoptosis, we hypothesized that LTB4 may be inhibiting a signaling cascade vital to endothelial cell survival, such as the sphingosine 1 phosphate (S1P)—endothelial nitric oxide synthase (eNOS) pathway. S1P is a class of vasoprotective signaling lipids produced by the phosphorylation of sphingosine by sphingosine kinase 1 (Sphk1) and downstream, and activates eNOS [55, 56]. Recent studies of S1P have revealed that this bioactive sphingolipid decreases vascular permeability and promotes endothelial survival through eNOS and prostacyclin [57–59]. In this recent study [54], we showed that LTB4 induced PAEC apoptosis by inhibiting the expression and activation of both Sphk1 and eNOS.
We further demonstrated that LTB4 induced the proliferation and hypertrophy of human pulmonary arterioles smooth muscle cells (PASMC) in a concentration- and BLT1-dependent manner [54]. These results are consistent with the established role of LTB4 on other types of vascular smooth muscle cells. For example, LTB4 has been strongly implicated in the process of atherogenesis, and BLT1 expression is increased in atherosclerotic plaques and on smooth muscle cells under inflammatory stress [60]. Thus, LTB4, liberated by activated macrophages, was produced in sufficient concentrations (as gauged by the bronchoalveolar fluid levels) to induce both PAEC apoptosis as well as PASMC proliferation, two hallmark pathologic events in PAH vasculopathy.
Because LTB4-secreting macrophages were contiguous with diseased arterioles, we hypothesized that inhibiting LTB4 signaling might be an effective treatment for PH. Bestatin [(2S, 3R)-3-amino-2-hydroxy-4-phenylbutanoyl-L-leucine] is a well-tolerated LTA4H inhibitor that blocks LTB4 formation [61, 62]. In our rat models, bestatin treatment started as late as 3 weeks after SU administration (when PH is severe and animals are near death from their cardiopulmonary disease), subsequently reduced serum LTB4 levels, prevented PAEC injury, restored Sphk1-eNOS signaling, and rescued the animals from death. Since bestatin is known to exert pharmacological actions other than LTA4H inhibition [63, 64], we also tested a different LTA4H inhibitor (JNJ-26993135) [65, 66] and a BLT1 antagonist (LY293111) [67, 68]. Treatment with both of these agents also reversed PH and prevented PH-related death. While bestatin was also effective with reversing MCT-induced PH, it was ineffective in the SU-chronic hypoxia model which is consistent with the notion that different PH pathophysiologies can emerge from different disease triggers, and results can vary in hosts with different genetic backgrounds.
To determine the clinical relevance of LTB4 in human PAH, we examined the expression of LTA4H in the lung tissue of PAH patients. In five out of six samples, LTA4H expression was notably elevated in macrophages that were clustered around occluded vessels and in the endothelial cells lining the lumen of the plexiform lesions. The fact that LTA4H was seen in both macrophages and endothelial cells in advanced disease (as opposed to being principally localized in macrophages early in disease) upregulation of both direct and transcellular synthetic pathways; it is possible that in addition to LTB4 being directly secreted by macrophages, LTA4 was also secreted by these cells, and this LT was taken up in PAECs (which are poor LTA4-producers) and converted in these increasingly abnormal occlusive intimal cells into LTB4 [69]. Also, the serum concentration of LTB4 was significantly elevated in PAH patients, especially in those with connective tissue disease-associated PAH (CTD-PAH). By contrast, six of eight patients with idiopathic PAH appeared to have normal LTB4 levels. One explanation for these heterogeneous findings is that CTDs, such as systemic lupus erythematosus and systemic sclerosis, exhibit significant defects in Treg function and number (more so than, perhaps, idiopathic PAH patients) that may facilitate the inappropriate activation of macrophages including increased LTB4 production [8, 70–72]. Thus, these new findings again lend support to the idea that different forms of PAH likely have unique pathogenic mechanisms [73, 74].
CysLTs in PAH
CysLTs are known to cause hypoxic pulmonary vasoconstriction [75], bronchoconstriction [76, 77], decreased lung compliance, and pulmonary edema [77]. The role of Cys-LTs has been well studied in pulmonary airways disease, but little is known about their role in PAH. Elevated levels of CysLTs were first reported in neonates with hypoxemia and pulmonary hypertension in 1983 [78]. The concentrations of eicosanoids, specifically TxB2, 6-keto-PGF1α, PGD2, PGE2, LTB4, LTC4, and LTE4, in bronchoalveolar lavage fluid (BALF) and blood of infants with persistent PH were also reported to be markedly increased. Furthermore, increased eicosanoids concentration correlated with poor clinical outcome of persistent pulmonary hypertension [79]. The continuing study of CysLTs in the airways may lend further insights into their role in the development of persistent PH as well as PAH.
Conclusions
This article has highlighted the growing information concerning how LTs may be participating in the development of certain forms of PAH. PAH remains an incurable and progressive disease. The last few decades have yielded a significantly deeper understanding of the pathobiology of PAH and have led to pharmacological advances, but there are challenges associated with these treatments, including the delivery systems, toxicity, frequent dosing schedules, variable efficacy, and cost [80]. Importantly, targeted immunotherapy as an adjuvant approach to current standard-of-care vasodilators has yet to be implemented. Anti-LT treatments may prove an effective complementary therapy for a subset of these patients.
Footnotes
Conflict of interest WT and MRN and Stanford University (OTL #S11-438) have a patent pending concerning the use of LTB4 antagonists for the treatment of PAH.
Contributor Information
Wen Tian, VA Palo Alto Health Care System, Stanford University, Med111P, 3801 Miranda Ave., Palo Alto, CA 94304, USA.
Xinguo Jiang, VA Palo Alto Health Care System, Stanford University, Med111P, 3801 Miranda Ave., Palo Alto, CA 94304, USA.
Yon K. Sung, VA Palo Alto Health Care System, Stanford University, Med111P, 3801 Miranda Ave., Palo Alto, CA 94304, USA
Jin Qian, VA Palo Alto Health Care System, Stanford University, Med111P, 3801 Miranda Ave., Palo Alto, CA 94304, USA.
Ke Yuan, Stanford University School of Medicine, Stanford, CA 94305, USA.
Mark R. Nicolls, VA Palo Alto Health Care System, Stanford University, Med111P, 3801 Miranda Ave., Palo Alto, CA 94304, USA
References
- 1.Benza RL, Miller DP, Gomberg-Maitland M, Frantz RP, Foreman AJ, Coffey CS, Frost A, Barst RJ, Badesch DB, Elliott CG, et al. Predicting survival in pulmonary arterial hypertension: insights from the registry to evaluate early and long-term pulmonary arterial hypertension disease management (REVEAL) Circulation. 2010;122(2):164–72. doi: 10.1161/CIRCULATIONAHA.109.898122. [DOI] [PubMed] [Google Scholar]
- 2.Rubin LJ. Primary pulmonary hypertension. N Engl J Med. 1997;336(2):111–7. doi: 10.1056/NEJM199701093360207. [DOI] [PubMed] [Google Scholar]
- 3.Rubin LJ. Pathology and pathophysiology of primary pulmonary hypertension. Am J Cardiol. 1995;75(3):51A–4A. doi: 10.1016/s0002-9149(99)80383-x. [DOI] [PubMed] [Google Scholar]
- 4.Barst RJ, McGoon M, Torbicki A, Sitbon O, Krowka MJ, Olschewski H, Gaine S. Diagnosis and differential assessment of pulmonary arterial hypertension. J Am Coll Cardiol. 2004;43(12 Suppl S):40S–7S. doi: 10.1016/j.jacc.2004.02.032. [DOI] [PubMed] [Google Scholar]
- 5.Schermuly RT, Ghofrani HA, Wilkins MR, Grimminger F. Mechanisms of disease: pulmonary arterial hypertension. Nat Rev Cardiol. 2011;8(8):443–55. doi: 10.1038/nrcardio.2011.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hassoun PM, Mouthon L, Barbera JA, Eddahibi S, Flores SC, Grimminger F, Jones PL, Maitland ML, Michelakis ED, Morrell NW, et al. Inflammation, growth factors, and pulmonary vascular remodeling. J Am Coll Cardiol. 2009;54(1 Suppl):S10–9. doi: 10.1016/j.jacc.2009.04.006. [DOI] [PubMed] [Google Scholar]
- 7.Nicolls MR, Taraseviciene-Stewart L, Rai PR, Badesch DB, Voelkel NF. Autoimmunity and pulmonary hypertension: a perspective. Eur Respir J. 2005;26(6):1110–8. doi: 10.1183/09031936.05.00045705. [DOI] [PubMed] [Google Scholar]
- 8.Taraseviciene-Stewart L, Nicolls MR, Kraskauskas D, Scerbavicius R, Burns N, Cool C, Wood K, Parr JE, Boackle SA, Voelkel NF. Absence of T cells confers increased pulmonary arterial hypertension and vascular remodeling. Am J Respir Crit Care Med. 2007;175(12):1280–9. doi: 10.1164/rccm.200608-1189OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Peters-Golden M, Henderson WR., Jr Leukotrienes. New Engl J Med. 2007;357(18):1841–54. doi: 10.1056/NEJMra071371. [DOI] [PubMed] [Google Scholar]
- 10.Peters-Golden M, Canetti C, Mancuso P, Coffey MJ. Leukotrienes: underappreciated mediators of innate immune responses. J Immunol. 2005;174(2):589–94. doi: 10.4049/jimmunol.174.2.589. [DOI] [PubMed] [Google Scholar]
- 11.Peters-Golden M, Brock TG. 5-lipoxygenase and FLAP. Prostaglandins Leukot Essent Fatty Acids. 2003;69(2–3):99–109. doi: 10.1016/s0952-3278(03)00070-x. [DOI] [PubMed] [Google Scholar]
- 12.Peters-Golden M, Brock TG. Intracellular compartmentalization of leukotrienebiosynthesis. AmJRespirCritCareMed. 2000;161(2Pt2):S36–40. doi: 10.1164/ajrccm.161.supplement_1.ltta-8. [DOI] [PubMed] [Google Scholar]
- 13.Samuelsson B, Funk CD. Enzymes involved in the biosynthesis of leukotriene B4. J Biol Chem. 1989;264(33):19469–72. [PubMed] [Google Scholar]
- 14.Fitzpatrick FA, Liggett WF, Wynalda MA. Albumin-eicosanoid interactions. A model system to determine their attributes and inhibition. J Biol Chem. 1984;259(5):2722–7. [PubMed] [Google Scholar]
- 15.Yokomizo T, Uozumi N, Takahashi T, Kume K, Izumi T, Shimizu T. Leukotriene A4 hydrolase and leukotriene B4 metabolism. J Lipid Mediat Cell Signal. 1995;12(2–3):321–32. doi: 10.1016/0929-7855(95)00041-n. [DOI] [PubMed] [Google Scholar]
- 16.Shimizu T, Izumi T, Seyama Y, Tadokoro K, Radmark O, Samuelsson B. Characterization of leukotriene A4 synthase from murine mast cells: evidence for its identity to arachidonate 5-lipoxygenase. Proc Natl Acad Sci USA. 1986;83(12):4175–9. doi: 10.1073/pnas.83.12.4175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Izumi T, Minami M, Ohishi N, Bito H, Shimizu T. Site-directed mutagenesis of leukotriene A4 hydrolase: distinction of leukotriene A4 hydrolase and aminopeptidase activities. J Lipid Mediat. 1993;6(1–3):53–8. [PubMed] [Google Scholar]
- 18.Minami M, Ohno S, Kawasaki H, Radmark O, Samuelsson B, Jornvall H, Shimizu T, Seyama Y, Suzuki K. Molecular cloning of a cDNA coding for human leukotriene A4 hydrolase. Complete primary structure of an enzyme involved in eicosanoid synthesis. J Biol Chem. 1987;262(29):13873–6. [PubMed] [Google Scholar]
- 19.Shimizu T, Izumi T, Ohishi N, Seyama Y, Kitamura S. Biosynthesis and further transformations of leukotriene A4. Adv Prostaglandin Thromboxane Leukot Res. 1987;17A:64–8. [PubMed] [Google Scholar]
- 20.Hammerstrom S, Samuelsson B. Detection of leukotriene A4 as an intermediate in the biosynthesis of leukotrienes C4 and D4. FEBS Lett. 1980;122(1):83–6. doi: 10.1016/0014-5793(80)80407-8. [DOI] [PubMed] [Google Scholar]
- 21.Funk CD. Prostaglandins and leukotrienes: advances in eicosanoid biology. Science. 2001;294(5548):1871–5. doi: 10.1126/science.294.5548.1871. [DOI] [PubMed] [Google Scholar]
- 22.Radmark O, Malmsten C, Samuelsson B. Leukotriene A4: enzymatic conversion to leukotriene C4. Biochem Biophys Res Commun. 1980;96(4):1679–87. doi: 10.1016/0006-291x(80)91367-4. [DOI] [PubMed] [Google Scholar]
- 23.Orning L, Hammarstrom S. Inhibition of leukotriene C and leukotriene D biosynthesis. J Biol Chem. 1980;255(17):8023–6. [PubMed] [Google Scholar]
- 24.Malmsten CL, Palmblad J, Uden AM, Radmark O, Engstedt L, Samuelsson B. Leukotriene B4: a highly potent and stereospecific factor stimulating migration of polymorphonuclear leukocytes. Acta Physiol Scand. 1980;110(4):449–51. doi: 10.1111/j.1748-1716.1980.tb06696.x. [DOI] [PubMed] [Google Scholar]
- 25.Sha’afi RI, Naccache PH, Molski TF, Borgeat P, Goetzl EJ. Cellular regulatory role of leukotriene B4: its effects on cation homeostasis in rabbit neutrophils. J Cell Physiol. 1981;108(3):401–8. doi: 10.1002/jcp.1041080314. [DOI] [PubMed] [Google Scholar]
- 26.Henderson WR, Jorg A, Klebanoff SJ. Eosinophil peroxidase-mediated inactivation of leukotrienes B4, C4, and D4. J Immunol. 1982;128(6):2609–13. [PubMed] [Google Scholar]
- 27.Czarnetzki BM. Is eosinophil chemotactic factor identical with leukotriene B? Int Arch Allergy Appl Immunol. 1982;67(2):181–3. doi: 10.1159/000233011. [DOI] [PubMed] [Google Scholar]
- 28.Jorg A, Henderson WR, Murphy RC, Klebanoff SJ. Leukotriene generation by eosinophils. J Exp Med. 1982;155(2):390–402. doi: 10.1084/jem.155.2.390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Murphy RC, Mathews WR. Purification and characterization of leukotrienes from mastocytoma cells. Methods Enzymol. 1982;86:409–16. doi: 10.1016/0076-6879(82)86213-7. [DOI] [PubMed] [Google Scholar]
- 30.Holgate ST, Church MK. Control of mediator release from mast cells. Clin Allergy. 1982;12(Suppl):5–13. doi: 10.1111/j.1365-2222.1982.tb03293.x. [DOI] [PubMed] [Google Scholar]
- 31.Hsueh W, Sun FF. Leukotriene B4 biosynthesis by alveolar macrophages. Biochem Biophys Res Commun. 1982;106(4):1085–91. doi: 10.1016/0006-291x(82)91223-2. [DOI] [PubMed] [Google Scholar]
- 32.Doig MV, Ford-Hutchinson AW. The production and characterisation of products of the lipoxygenase enzyme system released by rat peritoneal macrophages. Prostaglandins. 1980;20(6):1007–19. doi: 10.1016/0090-6980(80)90055-6. [DOI] [PubMed] [Google Scholar]
- 33.Fels AO, Pawlowski NA, Cramer EB, King TK, Cohn ZA, Scott WA. Human alveolar macrophages produce leukotriene B4. Proc Natl Acad Sci USA. 1982;79(24):7866–70. doi: 10.1073/pnas.79.24.7866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Hui Y, Funk CD. Cysteinyl leukotriene receptors. Biochem Pharmacol. 2002;64(11):1549–57. doi: 10.1016/s0006-2952(02)01357-6. [DOI] [PubMed] [Google Scholar]
- 35.Yokomizo T, Masuda K, Kato K, Toda A, Izumi T, Shimizu T. Leukotriene B4 receptor. Cloning and intracellular signaling. Am J Respir Crit Care Med. 2000;161(2 Pt 2):S51–5. doi: 10.1164/ajrccm.161.supplement_1.ltta-11. [DOI] [PubMed] [Google Scholar]
- 36.Izumi T, Yokomizo T, Igarashi T, Shimizu T. Molecular cloning and characterization of leukotriene B4 receptor. Adv Exp Med Biol. 1999;469:237–44. doi: 10.1007/978-1-4615-4793-8_35. [DOI] [PubMed] [Google Scholar]
- 37.Crooke ST, Mattern M, Sarau HM, Winkler JD, Balcarek J, Wong A, Bennett CF. The signal transduction system of the leukotriene D4 receptor. Trends Pharmacol Sci. 1989;10(3):103–7. doi: 10.1016/0165-6147(89)90206-x. [DOI] [PubMed] [Google Scholar]
- 38.Cristol JP, Provencal B, Sirois P. Leukotriene receptors. J Recept Res. 1989;9(4–5):341–67. doi: 10.3109/10799898909066063. [DOI] [PubMed] [Google Scholar]
- 39.Metters KM. Leukotriene receptors. J Lipid Mediat Cell Signal. 1995;12(2–3):413–27. doi: 10.1016/0929-7855(95)00027-n. [DOI] [PubMed] [Google Scholar]
- 40.Lipworth BJ. Leukotriene-receptor antagonists. Lancet. 1999;353(9146):57–62. doi: 10.1016/S0140-6736(98)09019-9. [DOI] [PubMed] [Google Scholar]
- 41.Voelkel NF, Tuder RM, Wade K, Hoper M, Lepley RA, Goulet JL, Koller BH, Fitzpatrick F. Inhibition of 5-lipoxygenase-activating protein (FLAP) reduces pulmonary vascular reactivity and pulmonary hypertension in hypoxic rats. J Clin Invest. 1996;97(11):2491–8. doi: 10.1172/JCI118696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wright L, Tuder RM, Wang J, Cool CD, Lepley RA, Voelkel NF. 5-Lipoxygenase and 5-lipoxygenase activating protein (FLAP) immunoreactivity in lungs from patients with primary pulmonary hypertension. Am J Respir Crit Care Med. 1998;157(1):219–29. doi: 10.1164/ajrccm.157.1.9704003. [DOI] [PubMed] [Google Scholar]
- 43.Jones JE, Walker JL, Song Y, Weiss N, Cardoso WV, Tuder RM, Loscalzo J, Zhang YY. Effect of 5-lipoxygenase on the development of pulmonary hypertension in rats. Am J Physiol Heart Circ Physiol. 2004;286(5):H1775–84. doi: 10.1152/ajpheart.00281.2003. [DOI] [PubMed] [Google Scholar]
- 44.Runo JR, Vnencak-Jones CL, Prince M, Loyd JE, Wheeler L, Robbins IM, Lane KB, Newman JH, Johnson J, Nichols WC, et al. Pulmonary veno-occlusive disease caused by an inherited mutation in bone morphogenetic protein receptor II. Am J Respir Crit Care Med. 2003;167(6):889–94. doi: 10.1164/rccm.200208-861OC. [DOI] [PubMed] [Google Scholar]
- 45.Aldred MA, Vijayakrishnan J, James V, Soubrier F, Gomez-Sanchez MA, Martensson G, Galie N, Manes A, Corris P, Simonneau G, et al. BMPR2 gene rearrangements account for a significant proportion of mutations in familial and idiopathic pulmonary arterial hypertension. Hum Mutat. 2006;27(2):212–3. doi: 10.1002/humu.9398. [DOI] [PubMed] [Google Scholar]
- 46.Newman JH, Trembath RC, Morse JA, Grunig E, Loyd JE, Adnot S, Coccolo F, Ventura C, Phillips JA, 3rd, Knowles JA, et al. Genetic basis of pulmonary arterial hypertension: current understanding and future directions. J Am Coll Cardiol. 2004;43(12 Suppl S):33S–9S. doi: 10.1016/j.jacc.2004.02.028. [DOI] [PubMed] [Google Scholar]
- 47.Takahashi H, Goto N, Kojima Y, Tsuda Y, Morio Y, Muramatsu M, Fukuchi Y. Downregulation of type II bone morphogenetic protein receptor in hypoxic pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol. 2006;290(3):L450–8. doi: 10.1152/ajplung.00206.2005. [DOI] [PubMed] [Google Scholar]
- 48.Song Y, Jones JE, Beppu H, Keaney JF, Jr, Loscalzo J, Zhang YY. Increased susceptibility to pulmonary hypertension in heterozygous BMPR2-mutant mice. Circulation. 2005;112(4):553–62. doi: 10.1161/CIRCULATIONAHA.104.492488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Radmark O, Samuelsson B. Regulation of the activity of 5-lipoxygenase, a key enzyme in leukotriene biosynthesis. Biochem Biophys Res Commun. 2010;396(1):105–10. doi: 10.1016/j.bbrc.2010.02.173. [DOI] [PubMed] [Google Scholar]
- 50.Werz O, Klemm J, Samuelsson B, Radmark O. 5-lipoxygenase is phosphorylated by p38 kinase-dependent MAPKAP kinases. Proc Natl Acad Sci USA. 2000;97(10):5261–6. doi: 10.1073/pnas.050588997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Radmark O, Samuelsson B. 5-Lipoxygenase: mechanisms of regulation. J Lipid Res. 2009;50(Suppl):S40–5. doi: 10.1194/jlr.R800062-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Werz O, Szellas D, Steinhilber D, Radmark O. Arachidonic acid promotes phosphorylation of 5-lipoxygenase at Ser-271 by MAPK-activated protein kinase 2 (MK2) J Biol Chem. 2002;277(17):14793–800. doi: 10.1074/jbc.M111945200. [DOI] [PubMed] [Google Scholar]
- 53.Tabata T, Ono S, Song C, Noda M, Suzuki S, Tanita T, Fujimura S. Role of leukotriene B4 in monocrotaline-induced pulmonary hypertension. Nihon Kyobu Shikkan Gakkai Zasshi. 1997;35(2):160–6. [PubMed] [Google Scholar]
- 54.Tian W, Jiang X, Tamosiuniene R, Sung YK, Qian J, Dhillon G, Gera L, Farkas L, Rabinovitch M, Zamanian RT, et al. Blocking macrophage leukotriene b4 prevents endothelial injury and reverses pulmonary hypertension. Sci Transl Med. 2013;5(200):200ra117. doi: 10.1126/scitranslmed.3006674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Chalfant CE, Spiegel S. Sphingosine 1-phosphate and ceramide 1-phosphate: expanding roles in cell signaling. J Cell Sci. 2005;118(Pt 20):4605–12. doi: 10.1242/jcs.02637. [DOI] [PubMed] [Google Scholar]
- 56.Wang L, Dudek SM. Regulation of vascular permeability by sphingosine 1-phosphate. Microvasc Res. 2009;77(1):39–45. doi: 10.1016/j.mvr.2008.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rodriguez C, Gonzalez-Diez M, Badimon L, Martinez-Gonzalez J. Sphingosine-1-phosphate: a bioactive lipid that confers high-density lipoprotein with vasculoprotection mediated by nitric oxide and prostacyclin. Thromb Haemost. 2009;101(4):665–73. [PubMed] [Google Scholar]
- 58.Gonzalez-Diez M, Rodriguez C, Badimon L, Martinez-Gonzalez J. Prostacyclin induction by high-density lipoprotein (HDL) in vascular smooth muscle cells depends on sphingosine 1-phosphate receptors: effect of simvastatin. Thromb Haemost. 2008;100(1):119–26. doi: 10.1160/TH07-11-0675. [DOI] [PubMed] [Google Scholar]
- 59.Gude DR, Alvarez SE, Paugh SW, Mitra P, Yu J, Griffiths R, Barbour SE, Milstien S, Spiegel S. Apoptosis induces expression of sphingosine kinase 1 to release sphingosine-1-phosphate as a “come-and-get-me” signal. FASEB J. 2008;22(8):2629–38. doi: 10.1096/fj.08-107169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Heller EA, Liu E, Tager AM, Sinha S, Roberts JD, Koehn SL, Libby P, Aikawa ER, Chen JQ, Huang P, et al. Inhibition of atherogenesis in BLT1-deficient mice reveals a role for LTB4 and BLT1 in smooth muscle cell recruitment. Circulation. 2005;112(4):578–86. doi: 10.1161/CIRCULATIONAHA.105.545616. [DOI] [PubMed] [Google Scholar]
- 61.Orning L, Krivi G, Fitzpatrick FA. Leukotriene A4 hydrolase. Inhibition by bestatin and intrinsic aminopeptidase activity establish its functional resemblance to metallohydrolase enzymes. J Biol Chem. 1991;266(3):1375–8. [PubMed] [Google Scholar]
- 62.Muskardin DT, Voelkel NF, Fitzpatrick FA. Modulation of pulmonary leukotriene formation and perfusion pressure by bestatin, an inhibitor of leukotriene A4 hydrolase. Biochem Pharmacol. 1994;48(1):131–7. doi: 10.1016/0006-2952(94)90232-1. [DOI] [PubMed] [Google Scholar]
- 63.Ota K, Kurita S, Yamada K, Masaoka T, Uzuka Y, Ogawa N. Immunotherapy with bestatin for acute nonlymphocytic leukemia in adults. Cancer Immunol Immunother. 1986;23(1):5–10. doi: 10.1007/BF00205548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ota K, Uzuka Y. Clinical trials of bestatin for leukemia and solid tumors. Biotherapy. 1992;4(3):205–14. doi: 10.1007/BF02174207. [DOI] [PubMed] [Google Scholar]
- 65.Rao NL, Dunford PJ, Xue X, Jiang X, Lundeen KA, Coles F, Riley JP, Williams KN, Grice CA, Edwards JP, et al. Anti-inflammatory activity of a potent, selective leukotriene A4 hydrolase inhibitor in comparison with the 5-lipoxygenase inhibitor zileuton. J Pharmacol Exp Ther. 2007;321(3):1154–60. doi: 10.1124/jpet.106.115436. [DOI] [PubMed] [Google Scholar]
- 66.Whittle BJ, Varga C, Berko A, Horvath K, Posa A, Riley JP, Lundeen KA, Fourie AM, Dunford PJ. Attenuation of inflammation and cytokine production in rat colitis by a novel selective inhibitor of leukotriene A4 hydrolase. Br J Pharmacol. 2008;153(5):983–91. doi: 10.1038/sj.bjp.0707645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Marder P, Spaethe SM, Froelich LL, Cerimele BJ, Petersen BH, Tanner T, Lucas RA. Inhibition of ex vivo neutrophil activation by oral LY293111, a novel leukotriene B4 receptor antagonist. Br J Clin Pharmacol. 1996;42(4):457–64. doi: 10.1046/j.1365-2125.1996.43210.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Marder P, Sawyer JS, Froelich LL, Mann LL, Spaethe SM. Blockade of human neutrophil activation by 2-[2-propyl-3-[3-[2-ethyl-4-(4-fluorophenyl)-5- hydroxyphenoxy]propoxy]phenoxy] benzoic acid (LY293111), a novel leukotriene B4 receptor antagonist. Biochem Pharmacol. 1995;49(11):1683–90. doi: 10.1016/0006-2952(95)00078-e. [DOI] [PubMed] [Google Scholar]
- 69.Feinmark SJ, Cannon PJ. Endothelial cell leukotriene C4 synthesis results from intercellular transfer of leukotriene A4 synthesized by polymorphonuclear leukocytes. J Biol Chem. 1986;261(35):16466–72. [PubMed] [Google Scholar]
- 70.Tamosiuniene R, Nicolls MR. Regulatory T cells and pulmonary hypertension. Trends Cardiovasc Med. 2011;21(6):166–71. doi: 10.1016/j.tcm.2012.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tamosiuniene R, Tian W, Dhillon G, Wang L, Sung YK, Gera L, Patterson AJ, Agrawal R, Rabinovitch M, Ambler K, et al. Regulatory T cells limit vascular endothelial injury and prevent pulmonary hypertension. Circ Res. 2011;109(8):867–79. doi: 10.1161/CIRCRESAHA.110.236927. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Buckner JH. Mechanisms of impaired regulation by CD4(+)CD25(+)FOXP3(+) regulatory T cells in human auto-immune diseases. Nat Rev Immunol. 2010;10(12):849–59. doi: 10.1038/nri2889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Rubin LJ. Treatment of pulmonary arterial hypertension due to scleroderma: challenges for the future. Rheum Dis Clin North Am. 2008;34(1):191–7. viii. doi: 10.1016/j.rdc.2007.11.003. [DOI] [PubMed] [Google Scholar]
- 74.Chin KM, Rubin LJ. Pulmonary arterial hypertension. J Am Coll Cardiol. 2008;51(16):1527–38. doi: 10.1016/j.jacc.2008.01.024. [DOI] [PubMed] [Google Scholar]
- 75.Smedegard G, Hedqvist P, Dahlen SE, Revenas B, Hammarstrom S, Samuelsson B. Leukotriene C4 affects pulmonary and cardiovascular dynamics in monkey. Nature. 1982;295(5847):327–9. doi: 10.1038/295327a0. [DOI] [PubMed] [Google Scholar]
- 76.Weiss JW, Drazen JM, Coles N, McFadden ER, Jr, Weller PF, Corey EJ, Lewis RA, Austen KF. Bronchoconstrictor effects of leukotriene C in humans. Science. 1982;216(4542):196–8. doi: 10.1126/science.7063880. [DOI] [PubMed] [Google Scholar]
- 77.Hanna CJ, Bach MK, Pare PD, Schellenberg RR. Slow-reacting substances (leukotrienes) contract human airway and pulmonary vascular smooth muscle in vitro. Nature. 1981;290(5804):343–4. doi: 10.1038/290343a0. [DOI] [PubMed] [Google Scholar]
- 78.Stenmark KR, James SL, Voelkel NF, Toews WH, Reeves JT, Murphy RC. Leukotriene C4 and D4 in neonates with hypoxemia and pulmonary hypertension. New Engl J Med. 1983;309(2):77–80. doi: 10.1056/NEJM198307143090204. [DOI] [PubMed] [Google Scholar]
- 79.Dobyns EL, Wescott JY, Kennaugh JM, Ross MN, Stenmark KR. Eicosanoids decrease with successful extracorporeal membrane oxygenation therapy in neonatal pulmonary hypertension. Am J Respir Crit Care Med. 1994;149(4 Pt 1):873–80. doi: 10.1164/ajrccm.149.4.8143049. [DOI] [PubMed] [Google Scholar]
- 80.Dingemanse J, Sidharta PN, Maddrey WC, Rubin LJ, Mickail H. Efficacy, safety and clinical pharmacology of macitentan in comparison to other endothelin receptor antagonists in the treatment of pulmonary arterial hypertension. Expert Opin Drug Saf. 2013 doi: 10.1517/14740338.2014.859674. doi:10.1517/14740338.2014.859674. [DOI] [PubMed] [Google Scholar]