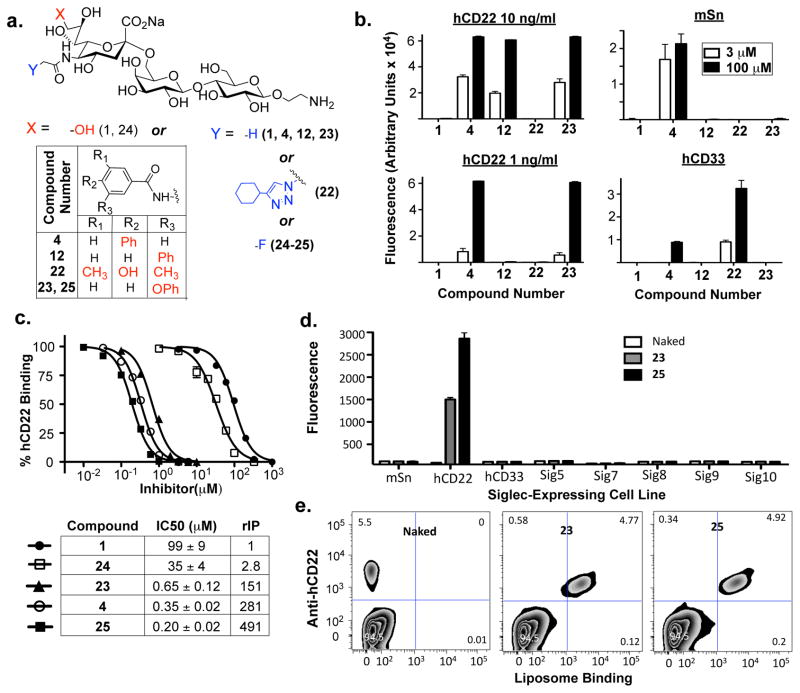
Fig. 3.
CD22-Counterscreening Leads to Insight for Subsequent Generation of a Selective, High-Affinity hCD22 Ligand. (a) Structures of sialoside analogues synthesized and tested. (b) Microarray screening of a small library of sialosides, including 12 and 23, show that bulky substituents at the meta-position of the ring gives selective increases in affinity for hCD22 over other siglecs. (c) A solution-phase inhibitor assay was used to validate and quantify the affinity gains identified in (b), as well as for newly generated analogues 24 and 25 containing a 5-N-Fluoroacetamide substituent known previously to give selective increases in affinity for hCD22 over mSn. (d) The mono- and disubstituted CD22 ligands 23 and 25 were coupled to lipids and formulated into fluorescent ligand-displaying liposomes. These were then assessed for binding to a panel of recombinant siglec-expressing cell lines. (e) Binding of these liposomes to white blood cells isolated from peripheral human blood demonstrate the ability of 23 and 25 to selectively target hCD22-expressing cells in this complex cellular mixture. Results shown in all experiments are representative of three independent experiments carried out in (b) quintuplicate, (c) duplicate, or triplicate (d–e). The IC50 values in (c) are the average of these three independent trials.