Figure 6.
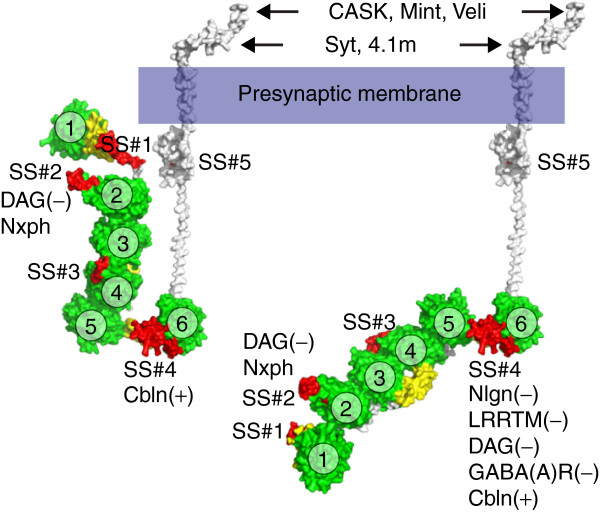
Structural models of α-neurexin. The diagram visualizes conformations that the extracellular domain of α-neurexin can assume. In the U-form (modeled from PDB ID: 3R05; left) only cerebellin (Cbln), neurexophilin (Nxph) and dystroglycan (DAG) might bind to LNS6 and LNS2, respectively. After rotation of about 180° in the αLNS5-αLNS6 hinge (modeled using PDB ID: 3ASI and 3R05; right), the core structure and αLNS6 become elongated and accessible to additional ligands, including neuroligins (Nlgn) and leucine-rich repeat molecules (LRRTM). The parentheses indicate the required presence (+) or absence (−) of the splice inserts in αLNS6 (SS#4) or αLNS2 (SS#2). Coordinates for αLNS1 have been modeled by sequence homology to other LNS domains because its electron density map was not resolved in the crystal structure [60]. Intracellularly, cytosolic proteins such as synaptotagmin (Syt), protein 4.1 from brain (4.1 m), CASK, Mint and Veli bind to the disordered carboxy-terminal domain of neurexins. LNS domains, green (numbered 1 to 6); EGF-like domains, yellow; splice inserts at splice sites #1 to #5, red. EGF, epidermal growth factor-like; LNS, laminin-neurexin-sex hormone binding globulin.
