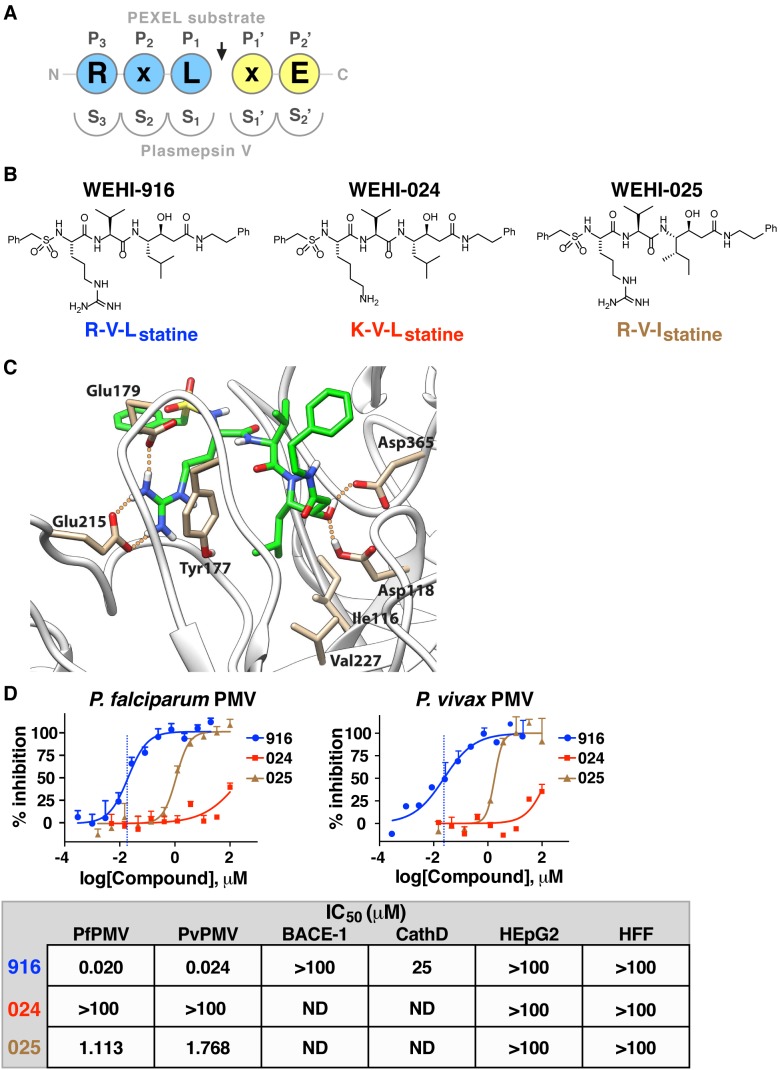
Figure 2. A PEXEL-mimetic inhibitor of PMV.
(A) Nomenclature for each residue in the PEXEL substrate (circles) and each pocket of the PMV active site (semicircles) with respect to the cleavage site (arrow). (B) Compound structures in this study include PMV inhibitor WEHI-916 and control analogs WEHI-024 and WEHI-025. (C) Structural model of PfPMV bound to WEHI-916. Residues forming the S3 site, Try177, Glu179, and Glu215, form interactions with the guanidine side chain of WEHI-916. The Leu side chain of WEHI-916 packs tightly against the side-chain groups of Ile116, Tyr177, and Val227 in the S1 site. The statine hydroxyl forms hydrogen bonds with the two catalytic aspartate residues Asp118 and Asp365. (D) Inhibition of PfPMV and PvPMV by WEHI-916 (blue) and weak activities of WEHI-024 (red) and WEHI-025 (brown). The grey box summarizes compound activity against BACE-1 and Cathepsin D (CathD) and lack of toxicity against human HEpG2 cells and Human Foreskin Fibroblasts (HFF).