Figure 7. Sumoylation and ubiquitination regulate CDK6 degradation.
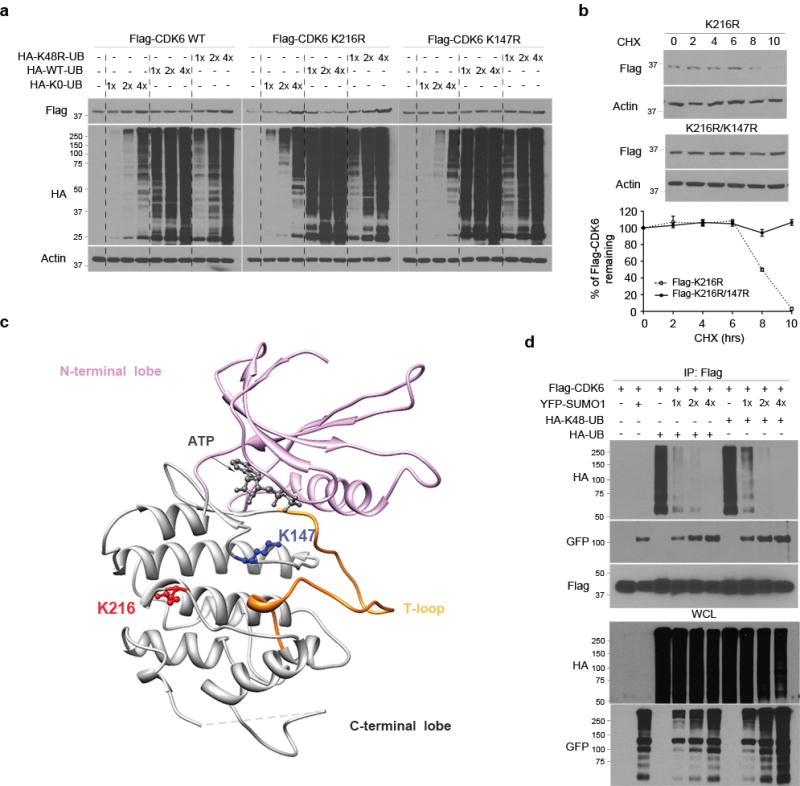
(a) Flag-CDK6 wt, K216R and K147R mt were transfected in LN229 cells with HA-K48R polyubiquitin (HA-48-UB), HA-ubiquitin (HA-UB) and/or non-conjugated ubiquitin mt (K0-UB) in the concentrations as indicated (top). Flag-CDK6 and HA-ubiquitin were detected by western blotting using Flag and HA antibodies. (b) The half-life of Flag-CDK6 K216R and K147R mt were evaluated by western blotting (top) and quantified against the control β-actin in SUMO1 and control shRNA expressing cells (bottom) (points: means; bar: SD; n = 6 from two independent experiments). (d) The CDK6 protein structure, generated at http://www.rcsb.org (PDB 2EUF), shows the kinase active site with the ATP binding side chains in the deep cleft between N-terminal and C-terminal lobe from which T-loop rises. K216 of the C-terminal is close to K147 near the kinase active site. (d) LN229 cells were transfected with Flag-CDK6, HA-K48-UB, HA-UB and YFP-SUMO1. Flag immunoprecipitates (IP) and whole cell lysate (WCL) were tested by western blotting using the indicated antibodies (left).
