Abstract
Treatment of type 1 diabetes with gene transfer–induced cellular reprogramming requires a pancreatic transcription factor such as Neurogenin-3 (Ngn3) and as of yet unknown component of the adenoviral particle. Despite intensive study, there are many unsolved processes related to the mechanisms and physiological parameters related to diabetes correction using this approach. While we confirm that systemic delivery of adenovirus (Ad)-Ngn3 provides long-lasting correction of streptozotocin (STZ)-induced hyperglycemia and restoration of growth curves, we found that insulin levels and glucose tolerance tests are not fully restored. By altering the innate and antigen-specific immune responses, we establish that the former likely plays some role in the reprogramming process. Interestingly, Ad-hNgn3 therapy in diabetic animals appeared to protect them from secondary STZ challenge. The resistance to secondary STZ response was more pronounced at later time points, indicating that a period of cell maturation and/or expansion may be required in order to promote lasting correction. More importantly, these results suggest that the long-term reprogrammed cells are not fully reprogrammed into β-cells, which in the case of autoimmune diabetes may be advantageous in a long-term treatment strategy. Finally, we show that the prophylactic administration of Ad-hNgn3 before diabetic induction protected mice from developing hyperglycemia, demonstrating the potential for reducing or eliminating disease progression should treatment be initiated early or before onset of symptoms.
Introduction
Type 1 diabetes or insulin-dependent diabetes mellitus results from the autoimmune destruction of the insulin-secreting pancreatic β-cells located within the islets of Langerhans. Changing the life style extensively in conjunction with monitoring and control of glycemia by subcutaneous insulin-replacement therapy is the current standard treatment option for patients, but requires a life-long commitment that may lead to noncompliance. Mismanagement of glycemic control raises the risk of complications, including diabetic ketoacidosis and hypoglycemic shock, as well as long-term complications, including impaired vision, heart disease, and renal damage. Therapeutic insulin replacement therapy fails to adequately recapitulate the tight control of blood glucose exhibited by endogenous insulin responses. As an alternative, the transplantation of isolated functional pancreatic islets derived from cadaveric donors (Rastellini et al., 1997; Shapiro et al., 2000) has been increasingly successful but is still considered experimental and is limited because of the availability of donor material, survival of the islet graft, and the need for lifelong immunosuppression. In light of these complications, there is a pressing need to develop new approaches for treatment of type 1 diabetes. Gene therapy strategies aimed toward stimulating β-cell regeneration or enhancing β-cell function, survival, and proliferation, as well as modulating the immune response in autoimmune models, all hold promise (Wideman et al., 2006; Shin et al., 2008; Riedel et al., 2010; Goudy et al., 2011). In addition, efforts toward creating renewable sources of high-quality β-cells in vitro through the differentiation of embryonic (Assady et al., 2001; D'Amour et al., 2006; Kroon et al., 2008), pluripotent (Alipio et al., 2010), and bone marrow-derived stem cells (Tang et al., 2004; Gao et al., 2008; Limbert et al., 2011) have been intensive and show future potential (Guo and Hebrok, 2009; Hori, 2009; Godfrey et al., 2011).
As an alternative to in vitro approaches, several groups are investigating the use of gene therapy to induce in vivo reprogramming of somatic cells, particularly those of the liver, pancreas, and/or gastrointestinal tract (Ferber et al., 2000; Ber et al., 2003; Kojima et al., 2003; Imai et al., 2005; Kaneto et al., 2005b, 2007; Wang et al., 2007; Zhou et al., 2008), into glucose-responsive insulin-expressing cells capable of correcting diabetes. In particular, adenoviral delivery of pancreatic and duodenal homeobox-1/insulin promoter factor (Pdx-1/Ipf-1) to the liver induced exocrine and endocrine gene expression, including insulin, resulting in long-term correction of hyperglycemia (Ferber et al., 2000; Ber et al., 2003). Kojima et al. (2003) subsequently demonstrated that helper-dependent adenovirus (Ad)-mediated delivery of Pdx-1 led to transient insulin expression and short-term correction of streptozotocin (STZ)-induced diabetes but that the mice suffered from viral hepatitis. Subsequently, islet neogenesis and correction of hyperglycemia in the absence of hepatitis after helper-dependent Ad-mediated delivery of a combination of NeuroD and the growth factor betacellulin was demonstrated. Adenoviral delivery of Pdx-1 and Neurogenin-3 (Ngn3) to the pancreas via the common bile duct (Taniguchi et al., 2003) led to islet ductal proliferation and islet neogenesis but failed to reverse STZ diabetes.
The initial small collection of transcription and growth factors was soon expanded to include MafA. Kaneto et al. (2005a) demonstrated the synergistic effect of including MafA in a therapeutic cocktail of Pdx-1 and NeuroD, leading to substantial expression of insulin mRNA and protein and ameliorating STZ diabetes in mice. In comparison, Pdx-1 and NeuroD administered without MafA were much less effective. Similarly, conversion of pancreatic acinar cells into functional β-cells after Pdx-1, Ngn3, and MafA transduction in the pancreas (Zhou et al., 2008) led to hyperglycemic correction and remodeling of the vasculature within the adult organ. In vivo reprogramming of exocrine to β-cells was found to be a direct conversion of cell types, bypassing an intermediate dedifferentiation step. Wang et al. (2007) found that hydrodynamic injection of plasmids carrying Pdx-1 and Ngn3 only reversed hyperglycemia when followed closely by injection of an Ad carrying an irrelevant transgene, or empty adenoviral capsids.
Repeat experiments in RAG-1−/− immunocompromised mice and a carbon tetrachloride–induced inflammation model suggested that the innate immunogenicity of the adenoviral capsid was required and that the effect was not caused simply by an increased nonspecific inflammation response. Ad transduction was shown to significantly upregulate several transcription factors, including signal transducer and activator of transcription 1 (Stat1) and the TATA-binding protein Abt1, either of which may contribute to reprogramming in general or be specifically involved in islet neogenesis in the liver. Subsequent work performed by Yechoor et al. (2009) demonstrated that Ngn3 expression in terminally differentiated hepatocytes led only to a short-term expression of insulin. Long-term correction of diabetes was found to be caused by the emergence of peri-portal islet-like cell clusters derived from hepatic oval stem cells (Yechoor et al., 2009).
Here, as a basis for further study, we provide a more detailed characterization of the physiological parameters associated with diabetes correction in an Ad-Ngn3-treated STZ diabetes mouse model and provide further evidence for the requirement of both an immune-based signaling mechanism and transfer of a pancreatic transcription factor Ngn3 for diabetes correction. Moreover, our studies suggest that the long-term correction of diabetes is based on an as-yet-unidentified cell that is not fully reprogrammed into a β-cell. Finally, we establish that pancreatic transcription factor-based reprogramming can be successful when initiated before the onset of disease.
Materials and Methods
Animals
C57BL/6J (000664) and B6.129S7-Rag1tm1Mom/J (002216) mice were purchased from Jackson Laboratories and were housed in a barrier facility at Stanford University. Mice were maintained on a 12 hr light/dark cycle and were fed a standard diet with unrestricted access to water. Mice were purchased at an age of 8–10 weeks. All animal protocols were approved by the Administrative Panel on Laboratory Animal Care at Stanford University.
First-generation Ad purification and injection
First-generation (E1/E3 deleted) Ads carrying the full human Ngn3 cDNA under control of the elongation factor 1-alpha promoter were amplified on HEK293 cells and purified by double caesium chloride density-gradient centrifugation. Total viral titers were calculated by spectrophotometry. The Rapid-X titration kit (Clontech) was used to determine functional infectious titer. Functional transgene expression was verified by Western blotting. Treatment injections consisted of 5×109 infectious viral particles delivered intravenously via the tail vein in 0.9% saline. Ad-hAAT and plasmid-encoded Ngn3 have been described in detail previously (Wang et al., 2007).
Plasmid administration by hydrodynamic tail vein injection
Plasmid encoding for Ngn3 under control of the EF1a promoter has been described previously (Wang et al., 2007). DNA was purified from large-scale bacterial cultures using Qiagen Maxi-prep columns. After ethanol precipitation, DNA was resuspended in TE to a final concentration of 1 mg/ml. Mice were injected with 50 μg of purified plasmid, in a final volume of 1 ml saline (0.9% NaCl in H2O) by rapid hydrodynamic injection.
Blood glucose monitoring and STZ induction of hyperglycemia
Mice were morning fasted (with access to water) for 6 hr and then weighed before blood collection from the retro-orbital sinus using a glass capillary. Blood glucose concentration was measured using a One-Touch Ultra II blood glucose meter (Lifescan Inc.) and test strips. Alternate eyes were used for blood collection on successive weekly sampling points to minimize potential damage. Hyperglycemia was induced by intraperitoneal injection of STZ (20 mg/ml in sodium citrate buffer, pH 4.5, prepared fresh) at a dose of 10 μl/g of body weight (final concentration of 200 mg/kg). Mice were considered diabetic after displaying a fasting blood glucose level of >300 mg/dl blood (16.6 mmol) on two consecutive testing days. Control mice were mock injected with an equivalent volume of citrate buffer. In most cases, severe diabetes led to profound morbidity and death within 21–28 days unless treated.
Intraperitoneal glucose tolerance testing and insulin ELISA
Insulin content of blood serum was determined using the Millipore ultra-sensitive mouse insulin kit as detailed in the manufacturer's instructions. Intraperitoneal glucose tolerance testing (IPGTT) was performed following the protocol detailed by the Animal Models of Diabetic Complications Consortium. Mice were fasted for 6 hr immediately after their dark cycle and then weighed. At this time, the basal blood glucose level was determined by sampling venous blood from a small tail clip. Mice were injected intraperitoneally with 10 μl/g body weight of 100 mg/ml d-glucose dissolved in saline. Venous blood samples were taken at the indicated times postinjection and assayed for glucose content.
Depletion of liver Kupffer cells with GdCl3
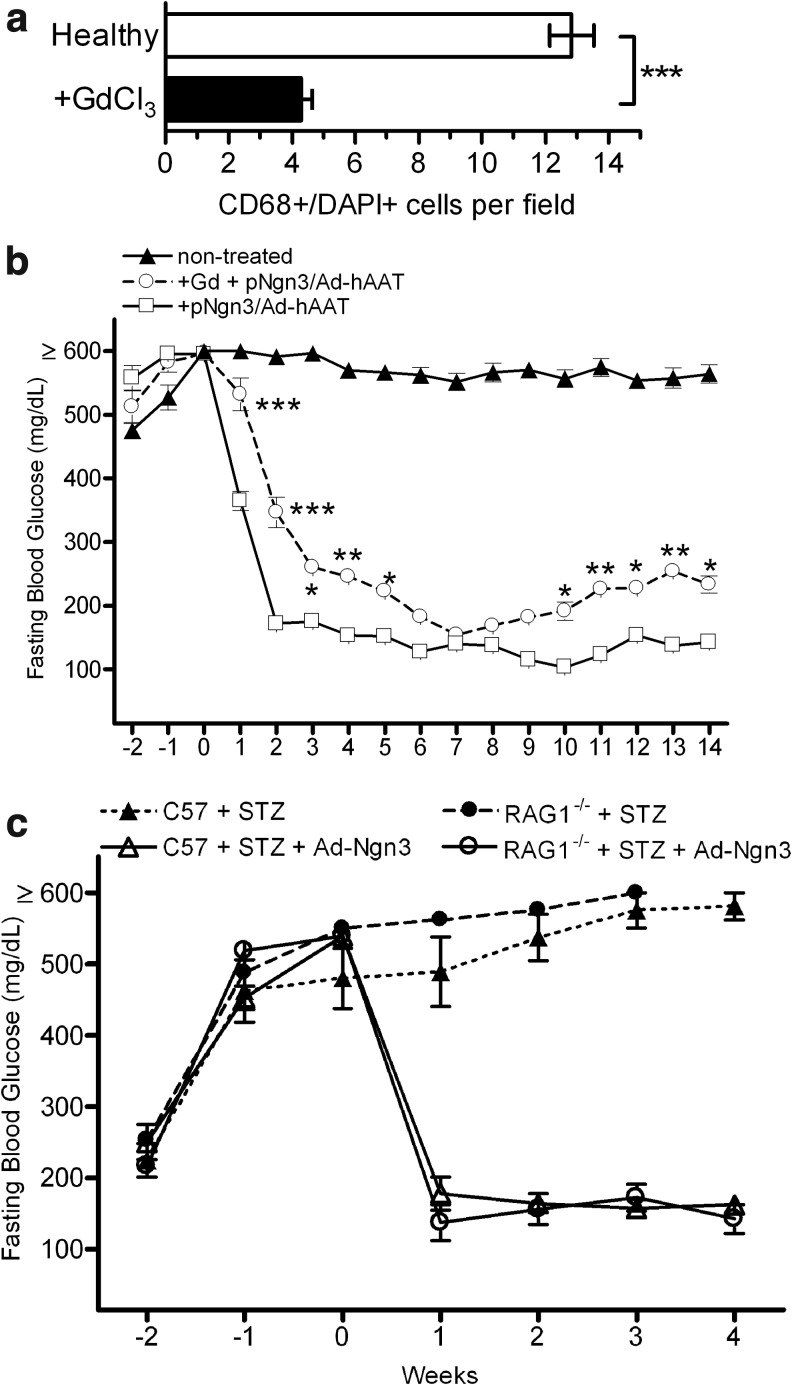
Transient depletion of liver Kupffer cells was achieved using a previously described protocol (Lieber et al., 1997). Briefly, gadolineum (III) chloride (GdCl3; Sigma G7532) was resuspended to 10 mg/ml in water, and then injected via the tail vein 30 and 6 hr before treatment at a final dose of 10 μg/g body weight adjusted to 200 μl with saline. Quantification of Kupffer cells was performed by staining liver sections with anti-CD68 primary antibody (Abcam Ab955, 1:200 dilution) followed by Texas Red-conjugated secondary antibody. Nuclei were stained using DAPI. Double-positive (CD68+DAPI+) cells were counted in randomly chosen fields of view using a Zeiss Axiovert fluorescence microscope.
Injection of dimethyldioctadecylammonium bromide and PolyIC adjuvants
Preparation of dimethyldioctadecylammonium bromide (DDA; Sigma D2779) and polyinosinic-polycytidylic acid (Poly(I:C); Sigma P1530) adjuvants was performed as detailed previously (Klinguer-Hamour et al., 2002). Briefly, 0.4 mg of DDA was resuspended in 1 ml of PBS, heated to 62°C for 30 min to dissolve, and then mixed for 4 hr at room temperature. Fifty microliters (20 μg) was injected subcutaneously at the base of the tail 24 hr before vector administration. Poly(I:C) was administered by intraperitoneal injection at a dose of 1 mg/ml in PBS (100 μg/0.1 ml per mouse) on three consecutive days.
Statistical analysis
Data were analyzed using Prism 4.0 software (Graph Pad Software). Data are expressed as mean±standard error of mean. Comparisons between two groups were performed using paired or unpaired Student's t-test, whereas comparisons among three or more groups were performed by one-way analysis of variance (ANOVA). Statistical significance was determined based on p<0.05. Significance is shown in figures as *p<0.05, **p<0.01, and ***p<0.001, unless otherwise indicated.
Results
Ad-hNgn3 treatment of STZ diabetic mice improves fasting blood glucose levels, body weight, insulin secretion, and glucose tolerance
STZ diabetic male C57BL/6J mice were injected with either a first-generation Ad carrying the human pancreatic transcription factor Ngn3 (Ad-hNgn3) or a control virus expressing human α-antitrypsin (Ad-hAAT). Human and murine Ngn3 genes are highly homologous on both the nucleotide (79% identity) and protein levels (76% identity, 82% similarity). Interestingly, the sequence of the DNA binding basic helix-loop-helix (bHLH) domain is 100% conserved between both species. After treatment, mice receiving Ad-hNgn3 (n=5) showed significant (p<0.001, all time points) improvement in fasting blood glucose levels (Fig. 1a) and increased body weight (Fig. 1b) compared with mice injected with control irrelevant Ad-hAAT (n=3). Both the improvement in blood glucose levels and weight gain were maintained for the duration of the experiment, terminated at week 8 because of development of cachexia in the control Ad-hAAT-injected mice. After Ad-hNgn3-mediated treatment of diabetes, blood was sampled from mice (n=3/group) in order to determine circulating insulin levels by ELISA (Fig. 1c). Treated mice had significantly elevated serum insulin levels compared with diabetic untreated controls (p=0.016; 0.85±0.13 ng/ml vs. 0.29±0.06 ng/ml). Despite being normoglycemic, treated mice had insulin levels twofold lower than healthy controls, demonstrating incomplete physiological restoration of hormone balance. Before and during the treatment schedule, Ad-hNgn3 mice were subjected to intraperitoneal glucose tolerance testing to determine glucose homeostasis (Fig. 1d). We modified the standard protocol slightly by reducing the time between successive blood collections in order to increase the temporal resolution of the assay. As expected, mice exhibited a pronounced defect in glucose homeostasis after STZ-induced diabetes when compared with their healthy baseline measurements. After Ad-hNgn3 treatment, mice displayed a marked improvement in glucose homeostasis, although they had higher peak glucose levels and a higher total excursion from the basal value compared with healthy measurements, revealing possible defects in both early (stage 1) and late (stage 2) insulin expression and release. These data suggest that treatment with an adenovirus encoding a single pancreatic transcription factor can reverse STZ-mediated hyperglycemia, increase serum insulin levels, and significantly improve glucose homeostasis.
FIG. 1.
Ad-hNgn3 treatment of STZ diabetic mice improves fasting blood glucose levels, body weight, serum insulin, and glucose tolerance. Fasting blood glucose (a) and body weights (b) of STZ diabetic mice treated with control Ad-hAAT (n=3) or Ad-hNgn3 (n=5). Male C57BL/6J mice were made diabetic via injection of 200 mg/kg STZ and then injected with virus 2 weeks later. Three weeks postinjection, serum insulin levels were measured by ELISA (c). Mice in the Ad-hNgn3 treatment group were subjected to intraperitoneal glucose tolerance testing (d) to determine glucose homeostasis before (Healthy) and after STZ induction (Diabetic) and after Ad-hNgn3 treatment (Treated). Mice were considered diabetic when fasting blood glucose exceeded 300 mg/dl. Data are shown as mean±SEM. ***p<0.001, *p=0.016. Ad, adenovirus; STZ, streptozotocin.
The role of reticulo-endothelial cells and antigen-dependent immune responses in diabetes correction
Previous studies have indicated that AAV-Ngn3 or plasmid-encoded Ngn3 is ineffective in treating STZ-induced hyperglycemia. However, administration of an irrelevant adenoviral vector immediately subsequent to hydrodynamic injection of pNgn3 leads to correction (Wang et al., 2007). Liver-resident Kupffer cells represent a first-line immune defense against infection by pathogens. We sought to determine the effect of Kupffer cell depletion on the outcome of pNgn3/AdhAAT treatment in diabetic mice. Kupffer cell depletion was performed as previously described (Lieber et al., 1997), initiated by administration of GdCl3 30 and 6 hr before liver harvest and sectioning. Depletion was verified by immunostaining of liver sections and counting of CD68+DAPI+ cells in randomly selected fields. Gadolinium treatment reduced the liver Kupffer cell population by 68% (p<0.001) (Fig. 2a). After confirmation that Kupffer cells could be efficiently depleted, diabetic mice were Kupffer-cell depleted immediately before pNgn3/Ad-hAAT treatment (n=5). Treated, Kupffer-depleted mice displayed significantly different blood glucose levels compared with nondepleted mice in the first 5 weeks after treatment and the last 4 weeks of the 14-week study period (p<0.001 weeks1 and 2; p<0.01, weeks 4, 11, and 13; p<0.05 weeks 3, 5, 10, 12, and 14) (Fig. 2b). Whereas correction in the nondepleted group was stable for the duration of the experiment (14 weeks posttreatment), normoglycemia in the depleted mice was temporary, with blood glucose levels gradually increasing from their lowest value at 7 weeks (154.6±11.7 mg/dl) over the next several weeks. Although mice displayed elevated glycemia 14 weeks after therapy, they remained below the diabetic threshold of 300 mg/dl and were very closely matched in blood glucose levels (233.8±31.0 mg/dl). These data would suggest that Kupffer cell-mediated immune responses may contribute in part to the pNgn3/Ad-hAAT-induced reprogramming events. As to how Kupffer cells play a role in this process, there are a couple of possibilities. First, reduced Kuppfer cell load in the liver could lower Ad-induced cytokine signaling, impacting the downstream reprogramming of cells transfected with the pNgn3 construct. Second, loss of Kuppfer cells could result in higher hepatocyte transduction leaving fewer adenoviral particles being free to interact with the relevant reprogramming signal-generating cells. Either of these scenarios would have the downstream effect of a lowered efficacy for reprogramming.
FIG. 2.
Kupffer cell depletion minimally influences pNgn3/Ad-hAAT therapy of STZ diabetes. Quantification of liver Kupffer cell load after GdCl3 treatment. Liver sections were stained with anti-CD68 primary antibody/Texas Red-conjugated secondary and DAPI, and double-stained cells were counted. GdCl3 treatment reduced Kupffer cell load within the liver by 68% (a). Fasting blood glucose levels of diabetic mice treated with pNgn3/Ad-hAAT with or without GdCl3-mediated Kupffer cell clearance. Diabetic C57BL/6J mice were injected with saline (n=5) or GdCl3 30 and 6 hr before (n=5) hydrodynamic administration of 50 μg pNgn3. Mice were injected 24 hr later with Ad-hAAT vector to complete treatment (b). Fasting blood glucose levels of diabetic C57BL/6J and Rag1−/− mice injected with Ad-hNgn3 (n=3 both groups) or saline (C57BL/6J n=3, Rag−/− n=2 because of a single mortality after STZ administration) 2 weeks after diabetic induction with STZ. Ad-hNgn3 administration led to correction in both groups of mice (c). Data are shown as mean±SEM. *p<0.05, **p<0.01, ***p<0.001.
A role for the immune response in pNgn3/Ad-hAAT treatment was previously hypothesized because of our inability to correct STZ diabetes with pNgn3/Ad-hAAT injection in Rag1−/− mice (Wang et al., 2007). We wanted to determine if this was also true when using Ad-hNgn3 for therapy. STZ diabetic C57BL/6J (n=3) and Rag1−/− mice (n=3) where treated with Ad-hNgn3. Both groups of mice were corrected of hyperglycemia with equal efficacy (Fig. 2c). No statistical differences were found between the Ad-hNgn3-treated groups at any stage of treatment (p>0.5 at 1 week posttreatment, n=3 for both groups), making it unlikely that an intact antigen-dependent immune system is an essential requirement for Ad-hNgn3-mediated hyperglycemic correction.
Immune function modulation by adjuvants facilitates correction of STZ diabetes after AAV-Ngn3 therapy
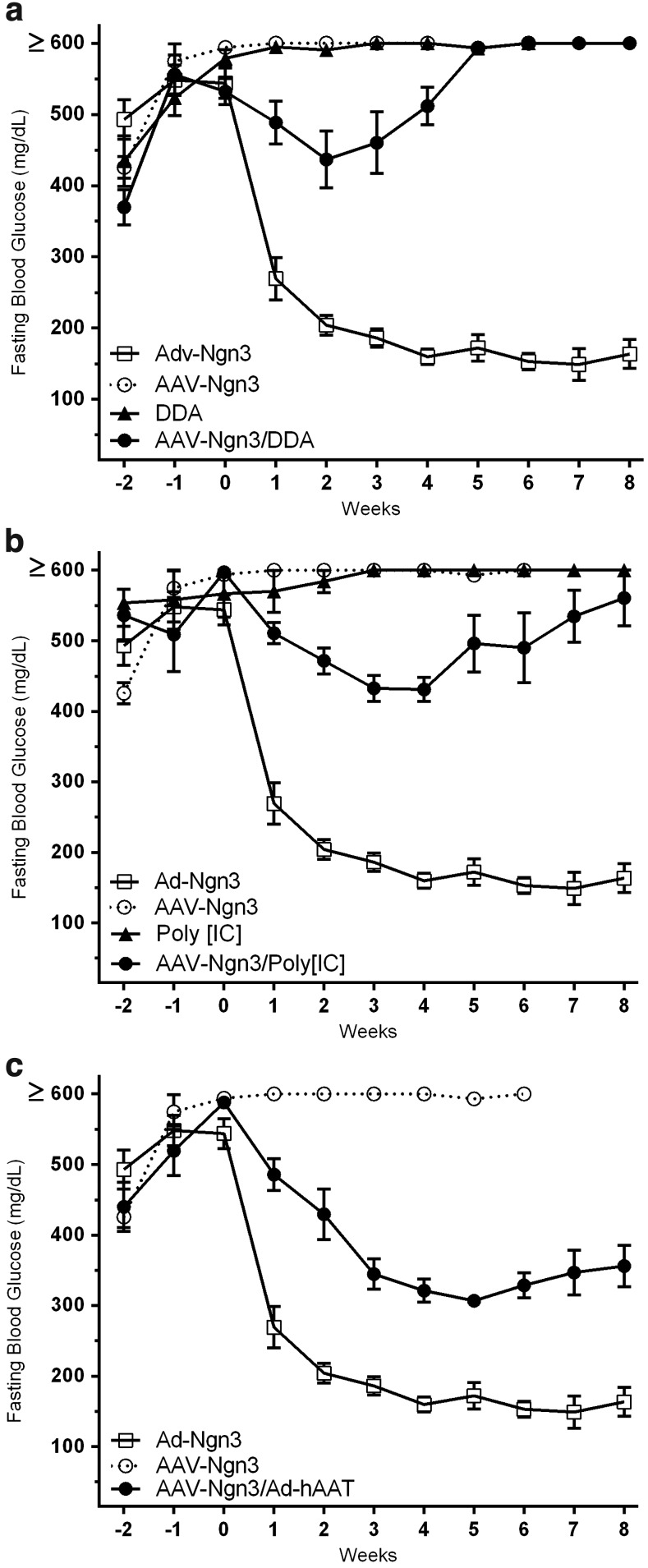
Previous studies have indicated that AAV-Ngn3 alone is ineffective in treating STZ-induced hyperglycemia (Wang et al., 2007). Two major possibilities for this exist: (1) there is a differential ability for the two vectors to transduce the cell types amenable to reprogramming; (2) there is a secondary signal induced by the Ad, but not AAV vector. Since it is known that Ad provides a much more potent innate immune response with the release of many cytokines (McCaffrey et al., 2008), we investigated the second possibility by using rAAV-Ngn3 in combination with two well-characterized adjuvants, DDA and polyinosinic-polycytidylicacid (poly IC), known to induce such a response. Adjuvant-AAV-Ngn3 administration resulted in a transient yet significant decrease in fasting blood glucose levels in hyperglycemic mice (Fig. 3). Mice treated with a combination of DDA and AAV-Ngn3 (n=5) displayed a 33% reduction in blood glucose 2 weeks after injection, falling to 391±14.1 mg/dl from >600 mg/dl (Fig. 3a). Mean blood glucose levels calculated over the entire treatment period were significantly different (**p=0.01) compared with controls treated with AAV-Ngn3 alone. However, fasting blood glucose levels then increased steadily, returning to pretreatment levels by 5 weeks posttreatment. Similarly, STZ diabetic mice injected with PolyIC:AAV-Ngn3 (n=4) displayed a similar reduction, with fasting blood glucose levels reaching a minimum level of 431±33.8 mg/dl 4 weeks after injection, increasing over the next 4 weeks to 560±68.1 mg/dl (Fig. 3b). Mean blood glucose levels calculated over the entire treatment period were significantly lower (*p=0.027) in polyIC:AAV-Ngn3 compared with AAV-Ngn3 alone controls. Neither DDA nor PolyIC administered alone had any effect on fasting blood glucose levels. The transient nature of the responses to adjuvant coadministration suggests that either a suboptimal number of cells were transduced and converted or that those cells were only partially corrected and failed to expand. We also tested the effect of adenoviral immune activation by coinjecting AAV-Ngn3 with Ad-hAAT, an adenoviral construct previously shown to be ineffective at correcting hyperglycemia. Although not as robust as treatment with Ad-hNgn3 itself, the AAV-Ngn3+ Ad-hAAT coinjection led to persistent correction (Fig. 3c). Five weeks postinjection, fasting blood glucose levels for these mice remained at 60% of diabetic levels, dropping to 306±10.2 mg/dl from 588±10.4 mg/dl. Mean blood glucose levels in this group were significantly different from both positive control Ad-hNgn3 (***p=0.0013) and negative control AAV-Ngn3 (***p=0.0046) treatment groups. These data suggest a requirement for an unidentified Ad-induced factor in the reprogramming signaling pathway that acts directly on the transduced cells during reprogramming and/or the result of Ad induced changes in AAV vector tropism and/or transduction efficiency. This suggests that by careful manipulation of the adenoviral responses induced during AAV-Ngn3 treatment, it may be possible to recapitulate the persistent correction seen using Ad based therapy.
FIG. 3.
Coadministration of AAV-Ngn3 and adjuvants can correct STZ diabetes. Fasting blood glucose levels of STZ diabetic mice treated with Ad-hNgn3 control (n=5) or AAV-Ngn3 (n=3) in the presence of immunostimulatory agents DDA (n=5) (a), Poly[IC] (n=4) (b), or Ad-hAAT (n=4) (c). Male C57BL/6J mice were made diabetic via injection of 200 mg/kg STZ and then treated 2 weeks later with AAV-Ngn3 alone or after immune stimulation with DDA or Poly[IC]. Contribution of Ad-mediated immunostimulation was determined by AAV-Ngn3/Ad-hAAT coinjection. Data are shown as mean±SEM. *p<0.05, **p<0.01, ***p<0.001. Control Ad-hNgn3 and AAV-Ngn3 curves are replicated in each panel for ease of comparison.
Ameliorative treatment with Ad-hNgn3 results in altered sensitivity to secondary STZ exposure
To be successful, any cell-based therapy for type 1 diabetes would have to be resistant to the underlying autoimmune processes that mediate the destruction of β-cells. As a first step to unraveling this issue, we reasoned that true β-cells may be susceptible to STZ-mediated ablation based on GLUT2 expression and that “transitional” or incompletely reprogrammed cells may show STZ resistance. To test this idea, a single large cohort of mice (n=27) was divided into two groups, one of which was made diabetic (n=15) using a single intraperitoneal dose of STZ. The remaining 12 healthy mice were to be used as age-matched controls undergoing naïve STZ exposure. Of the 15 diabetic mice, all but 3 were injected with Ad-hNgn3 as previously described, and treated mice quickly reattained normoglycemia and displayed improvements in body weight. At various times posttreatment, previously diabetic, Ad-hNgn3-treated mice (n=3 per time-point) were injected with a second dose of STZ, alongside age-matched control mice (n=3) taken from the STZ-naïve group. Fasting blood glucose levels (Fig. 4, left, and Supplementary Fig. S1; Supplementary Data are available online at www.liebertpub.com/hum) and body weights (Fig. 4, right) were measured weekly. Mice subjected to secondary STZ exposure after Ad-hNgn3 treatment have a delayed progression to hyperglycemia compared with their age-matched controls, with mice challenged at later times displaying lower blood glucose levels compared with those re-exposed earlier. Mice were re-exposed to STZ 5, 8, 18, and 25 weeks after the initial Ad-hNgn3 treatment injection. Although mice re-exposed to STZ 5 and 8 weeks postinjection (Fig. 4a and b) were sensitive to STZ and go on to develop severe hyperglycemia indistinguishable from control mice (n.s.; p=0.08 for 8 week group), the induction kinetics slightly lagged behind that of the age-matched controls. By 18 weeks posttreatment (Fig. 4c and Supplementary Fig. S1b), the delayed induction kinetics were evident, despite the differences in mean blood glucose lacking statistical significance overall. In the case of the 25 week cohort (Fig. 4d), delayed STZ induction kinetics were clearly evident and mean blood glucose levels were significantly different (p<0.001) compared with age-matched control mice. Additionally, these mice develop less severe hyperglycemia over the course of 6–8 weeks after secondary STZ injection compared with controls, which reach severe levels of blood glucose within 3 weeks. There were no statistically significant differences in STZ response displayed by untreated control mice and the 25-week control group (34 weeks old at time of STZ injection). Corresponding body weight measurements show that, in later cases, the rate of weight loss in re-exposed mice was equivalent to that of age-matched mice undergoing primary STZ exposure (Fig. 4, right, b–d). Mice in the 5-week re-exposure group that had a higher rate of weight loss than expected compared with age-matched controls were the exception. Weight loss in this group more closely paralleled that of the original diabetic untreated controls (Fig. 4, right, a), suggesting that the newly generated cells were more sensitive to STZ toxicity earlier in the reprogramming process, leading to more profound physiological disturbances during disease progression. This suggests the formation of an STZ-resistant population of cells that arises in response to treatment by Ad-Ngn3 and that this population may take several weeks to mature and confer a protective advantage.
FIG. 4.
Successful treatment of STZ diabetic mice with Ad-hNgn3 leads to development of STZ resistance. A single large cohort of mice (n=27) was divided into two groups, one of which was made diabetic (n=15) using a single intraperitoneal dose of STZ. The remaining 12 healthy mice were used as age-matched controls undergoing naïve STZ exposure. Of the 15 diabetic mice, all but 3 (non-Ad-Ngn3-injected controls) were injected with Ad-hNgn3 two weeks later and then exposed to a secondary dose of STZ alongside age-matched healthy controls (n=3 for each comparison group). Fasting blood glucose levels (left) and body weights (right) of mice treated with secondary STZ 5 weeks (a), 8 weeks (b), 18 weeks (c), and 25 weeks (d) after initial treatment. Data are shown as mean±SEM. Values for the untreated control group are shown replicated in each graph for ease of comparison.
Pretreatment with Adv-Ngn3 attenuates STZ-induced diabetic hyperglycemia
Finally, we sought to determine the therapeutic potential of administering Ad-hNgn3 before STZ-mediated hyperglycemia. We injected a cohort of mice with Adv-Ngn3 (n=15) and then, 4 weeks later, induced hyperglycemia using STZ. Subsequently, blood glucose levels were monitored weekly for 4 more weeks. Control mice not receiving Ad-hNgn3 pretreatment (n=6) became severely diabetic after STZ injection (Fig. 5a), with fasting blood glucose exceeding 550 mg/dl, whereas only 13% (2/15 mice) of pretreated mice had severe diabetes. On the whole the controls and pretreated mice had a significant difference in their fasting blood glucose (unpaired t-test p=0.0061). While the pretreated cohort of mice displayed a marked reduction in fasting blood glucose as a group, we found three distinct patterns based on a combination of fasting blood glucose levels and improvement in body weight. Of the 15 pretreated mice, 47% (7/15) developed moderate diabetes, defined as fasting blood glucose levels in the range of 300–550 mg/dl, and displayed delayed progression of disease (Supplementary Fig. S2). Fasting blood glucose levels in this group were significantly altered compared with both the untreated control group and the mice in the pretreated group that developed severe hyperglycemia (p=0.006). The remaining 40% (6/15) of mice in this group had blood glucose levels significantly reduced (p<0.001) compared with untreated/treated-severe (i.e., those mice pretreated with Ad-hNgn3 that developed severe hyperglycemia) and treated-moderate groups. Corresponding body weight measurements (Fig. 5b) indicate that, despite developing moderate hyperglycemia, the partially protected mice displayed only a small decrease in weight compared with the fully protected cohort and showed a small gain in weight as time progressed. Mice in this group had a slower rate of diabetic induction in response to STZ administration (Supplementary Fig. S2), a phenotype shared with the two pretreated mice that developed severe hyperglycemia. These two mice had higher body weight compared with the untreated control animals. We conclude that a prophylactic administration of Ad-hNgn3 can attenuate STZ-induced hyperglycemia in mice and can even prevent alterations in blood glucose levels entirely.
FIG. 5.
Pretreatment with Ad-hNgn3 protects against STZ-induced hyperglycemia. Fasting blood glucose levels and body weights of mice pretreated with Ad-hNgn3 before induction of STZ diabetes. Two groups of mice were injected with either saline (n=6) or Ad-hNgn3 (n=15) 3 weeks before STZ exposure. Graphs show fasting blood glucose levels (a) 4 weeks after STZ injection and body weights (b) for the duration of the experiment. Open symbols (□, ▵, ▿) show values for individual mice pretreated with Ad-hNgn3; closed circles (•) show values for untreated mice. When we compared the untreated versus pretreated cohorts using an unpaired t-test, the groups were significantly different (p=0.0061). The pretreated cohort could be grouped into the following categories: Full responders (▵), partial responders (□), and nonresponders (▿). In (a) horizontal bar represents mean of values in the group. Data in (b) are mean±SEM. **p<0.01, ***p<0.001.
Discussion
Mouse models of type 1 diabetes are limited and do not faithfully recapitulate the complex immunological mechanisms of human disease. Traditionally, the beta cell–selective toxins STZ or Alloxan have been used to induce hyperglycemia. These compounds are transported into the beta cell via the beta cell–restricted GLUT2 transporter, where they induce DNA damage or create free-radical-dependent oxidative stress and promote beta cell death. More recently, the use of transgenic mice expressing toxin receptors under control of the rat insulin 1 promoter has gained favor. The RIP-DTr (Thorel et al., 2010) and the doxycycline-inducible insulin-rtTA;TET-DTA (Nir et al., 2007) transgenic mice allow diphtheria-toxin-mediated beta cell ablation that results in hyperglycemia. However, these groups have shown that high-level, almost complete ablation of beta cells results in transdifferentiation of alpha cells into beta cells, or the expansion of residual beta cells that compensates and eventually repopulates beta cell mass. These “regenerative” pathways for beta cell compensation would have significantly affected our ability to directly monitor the response to Ngn3 gene therapy. Use of the autoimmune nonobese diabetic NOD mouse (Makino et al., 1980) would be similarly complicated by the fact that if reprogramming were to occur, its detection by glucose correction may not be long-lived or even detectable because of our inability to know if the newly reprogrammed cells would be subjected to destruction. Therefore, for these initial studies we elected to use the standard STZ-induced hyperglycemia model.
Although Ad-hNgn3-treated diabetic mice have physiologically normal levels of blood glucose, they had reduced serum insulin levels compared with healthy control animals. Despite appearing otherwise healthy and normoglycemic, a consequence of this are the defects in first- and second-stage insulin kinetics seen during intraperitoneal glucose tolerance testing. Delayed or decreased peak insulin secretion immediately after glucose administration would result in decreased glucose metabolism throughout testing, clearly demonstrated by increased glucose excursion compared with healthy animals. It is currently unknown if these defects persist throughout the remaining life of the animal or what the long-term complications of this might be. Defects in first-phase insulin response are well characterized in models of type II diabetes (Caumo et al., 2004). However, interpreting the results of acute glucose administration, as in IPGTT protocols, may be complicated by the fact that insulin secretion is not intrinsically biphasic in vivo during postprandial periods. Under physiological conditions, where blood glucose levels increase gradually after food ingestion, the first and second phases are not clearly distinguishable. As such, the long-term physiological relevance of the altered insulin/glucose kinetics is not known. Improving the efficiency of gene delivery into the yet-unidentified amenable cell population and/or the inclusion of additional pancreatic transcription factors or regulatory components may be required to properly recapitulate normal physiological responses.
Despite its demonstrated efficacy in this case, Ad-based systems are suboptimal for gene therapy. We attempted to translate our findings from the Ad study to an AAV-based therapy that is more relevant clinically. Normally, administration of an AAV-Ngn3 vector to STZ diabetic mice is ineffective at restoring normoglycemia. Coadministration of AAV-Ngn3 and Ad-hAAT can partially correct hyperglycemia; however, the treatment is not as robust as using Ad-hNgn3 and is associated with a high level of toxicity. We surmised that we could remove the Ad-hAAT vector component and modulate an immune response to AAV by coinjecting vaccine adjuvants known to induce a robust immune response. Poly[IC] activates toll-like receptor 3, mimicking a viral antigen (double-stranded RNA) and inducing a potent response similar to that displayed against viruses. DDA has been shown to induce a mixed Th1/Th2 response (Klinguer-Hamour et al., 2002) and has been used to augment immune responses to a number of different antigens, including viruses, bacteria, and plasmid DNA (Klinguer et al., 2001; van Rooij et al., 2002; Cai et al., 2004; Roh et al., 2006). Injection of AAV-Ngn3 in the presence of adjuvants led to a transient reduction in fasting blood glucose levels. In particular, the viral RNA mimic poly[IC] reduced hyperglycemia by approximately 30% over a period of 3–4 weeks. Similarly, fasting blood glucose in the cohort receiving combination DDA+ AAV-Ngn3 therapy was decreased compared with mice receiving only DDA. As such, we believe that this differential result shows that the drop in blood glucose was because of a synergistic effect of DDA and AAV-Ngn3 and was not caused solely by DDA administration alone. It is possible that, with further characterization of this response, for example, dose escalation or repeated administration studies, an effect comparable to that seen when using Ad-hNgn3 may be achieved. Interestingly, Poly[IC] has in the past been used to induce diabetes in mice when used as an adjuvant during administration of insulin peptides (Moriyama et al., 2002), hinting at the use of caution when performing adjuvant studies in this context. The ability to replace the Ad component of the combined treatment with immunostimulatory adjuvants indicates that an adenoviral-induced immune response may sensitize the host in some way, making it more responsive to cellular reprogramming or expansion. However, an alternative may be that Ad-induced changes in the tropism or uptake of AAV-Ngn3 result in increased cell transduction and gene expression, improving treatment efficacy compared with instances where AAV-Ngn3 is administered alone. Coinfection of Ad and AAV has been shown to increase AAV transduction and genome expression (Ferrari et al., 1996; Fisher et al., 1996), supporting this theory. It is unknown at this time if administration of adjuvants alongside AAV also results in increased transduction or can influence AAV tropism.
The development of STZ resistance in Ad-hNgn3-treated mice is an intriguing observation and perhaps because of the possibility that the reprogrammed cells, while able to produce insulin in response to glucose, are not fully differentiated into β-cells, making them resistant to STZ attack. Such a phenotype might include that these cells have insufficient GLUT2 expression (or downstream effectors) on the surface of the cells to diminish the effective STZ concentration required for cellular toxicity. Since GLUT2 receptor is important for glucose sensing and insulin release, the STZ resistance and phase 1–2 insulin deficiencies may stem from diminished GLUT2 expression.
Partial reprogramming may actually be advantageous because type 1 diabetes in humans results from autoimmune destruction of the β-cells. In the case of full reprogramming, it is possible that these cells would be subject to continued destruction. However, it becomes less clear if these cells would be subject to the same autoimmune destructive process.
We note that hyperglycemia resulting from STZ induction lacks many of the correlates of true autoimmune type 1 diabetes. A single high-dose exposure to STZ rapidly induces β-cell necrosis, leading to hyperglycemia. In contrast, multiple, low-dose exposure to STZ results in a delayed onset of hyperglycemia, which is kinetically independent of immediate STZ toxicity. MLD- STZ partially damages β-cells, triggering inflammatory processes that result in further β-cells ablation, insulin deficiency, and hyperglycemia, which more closely recapitulates type 1 diabetes mellitus. However, we found that multiple, low-dose administration of STZ was inconsistent in inducing a stable hyperglycemic state and mice would occasionally spontaneously revert to normoglycemia. Ultimately, if therapy is to be considered for human use, further work should be done using a model of type 1 diabetes that more fully reconstitutes the complexities of the autoimmune phenotype characteristic of human disease. Use of immunological models may represent a more useful approach, both for vector-induced reprogramming studies and for studies involving iPS conversion and transplantation. The presence of a robust diabetogenic immune response will allow better monitoring of the survival of reprogrammed and transplanted cells in context of continued autoimmune challenge. Prophylactic treatment of at-risk groups predisposed to developing type 1 diabetes is an attractive future direction for diabetes management. Using our therapeutic Ad-hNgn3 in a “pretreatment” context, we were able to reduce the severity of induced diabetes and, in some cases, completely prevent disease progression. Interestingly, those mice that did develop hyperglycemia had altered disease progression and displayed less severe weight loss compared with unprotected controls, which may provide a larger window of opportunity for further treatment efforts to control the hyperglycemia in these animals. However, we did not test the efficacy of a subsequent corrective injection of Ad-hNgn3 in the mice that were preinjected, but it is anticipated that complications because of vector immunity would be of relevant concern.
In conclusion, we have demonstrated that Ad-hNgn3 can be used to both prevent and ameliorate hyperglycemia in a mouse model of type 1 diabetes. Administration of Ad-hNgn3 at a well-tolerated dose to STZ diabetic mice resulted in a rapid and long-term reduction in blood glucose levels to physiologically healthy levels, with an associated improvement in both body weight and glucose clearance as determined by intraperitoneal glucose tolerance tests. Recently, a number of groups have demonstrated similar results using both Ad- and AAV-based vectors for delivering several pancreatic transcription factors. However, ours is the first demonstration of complete and long-term normoglycemia after only a single systemic administration of vector. Importantly, we found that pretreatment of mice with Ad-hNgn3 before STZ administration can ameliorate the severity of induced diabetes and, in some cases, can completely prevent induction of hyperglycemia.
Supplementary Material
Acknowledgments
We thank F. Zhang for performing tail vein injection of adenoviral constructs and hydrodynamic tail vein injection of DNA plasmids. We also thank A. Barzel and K. Jameson for critical review of the article. This work was supported by the Juvenile Diabetes Research Foundation Grant 46-2010-750 and NIH U01 DK089569.
Author Disclosure Statement
No competing financial interests exist for either author.
References
- Alipio Z., Liao W., Roemer E.J., et al. (2010). Reversal of hyperglycemia in diabetic mouse models using induced-pluripotent stem (iPS)-derived pancreatic beta-like cells. Proc. Natl. Acad. Sci. USA 107, 13426–13431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Assady S., Maor G., Amit M., et al. (2001). Insulin production by human embryonic stem cells. Diabetes 50, 1691–1697 [DOI] [PubMed] [Google Scholar]
- Ber I., Shternhall K., Perl S., et al. (2003). Functional, persistent, and extended liver to pancreas transdifferentiation. J. Biol. Chem. 278, 31950–31957 [DOI] [PubMed] [Google Scholar]
- Cai H., Tian X., Hu X.D., et al. (2004). Combined DNA vaccines formulated in DDA enhance protective immunity against tuberculosis. DNA Cell Biol. 23, 450–456 [DOI] [PubMed] [Google Scholar]
- Caumo A., and Luzi L. (2004). First-phase insulin secretion: does it exist in real life? Considerations on shape and function. Am. J. Physiol. Endocrinol. Metab. 287, E371–E385 [DOI] [PubMed] [Google Scholar]
- D'Amour K.A., Bang A.G., Eliazer S., et al. (2006). Production of pancreatic hormone-expressing endocrine cells from human embryonic stem cells. Nat. Biotechnol. 24, 1392–1401 [DOI] [PubMed] [Google Scholar]
- Ferber S., Halkin A., Cohen H., et al. (2000). Pancreatic and duodenal homeobox gene 1 induces expression of insulin genes in liver and ameliorates streptozotocin-induced hyperglycemia. Nat. Med. 6, 568–572 [DOI] [PubMed] [Google Scholar]
- Ferrari F.K., Samulski T., Shenk T., et al. (1996). Second-strand synthesis is a rate-limiting step for efficient transduction by recombinant adeno-associated virus vectors. J. Virol. 70, 3227–3234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher K.J., Gao G.P., Weitzman M.D., et al. (1996). Transduction with recombinant adeno-associated virus for gene therapy is limited by leading-strand synthesis. J. Virol. 70, 520–532 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao X., Song L., Shen K., et al. (2008). Transplantation of bone marrow derived cells promotes pancreatic islet repair in diabetic mice. Biochem. Biophys. Res. Commun. 371, 132–137 [DOI] [PubMed] [Google Scholar]
- Godfrey K.J., Mathew B., Bulman J.C., et al. (2011). Stem cell-based treatments for type 1 diabetes mellitus: bone marrow, embryonic, hepatic, pancreatic and induced pluripotent stem cells. Diabet. Med. 29, 14–23 [DOI] [PubMed] [Google Scholar]
- Goudy K.S., Johnson M.C., Garland A., et al. (2011). Inducible adeno-associated virus-mediated IL-2 gene therapy prevents autoimmune diabetes. J. Immunol. 186, 3779–3786 [DOI] [PubMed] [Google Scholar]
- Guo T., and Hebrok M. (2009). Stem cells to pancreatic beta-cells: new sources for diabetes cell therapy. Endocr. Rev. 30, 214–227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hori Y. (2009). Insulin-producing cells derived from stem/progenitor cells: therapeutic implications for diabetes mellitus. Med. Mol. Morphol. 42, 195–200 [DOI] [PubMed] [Google Scholar]
- Imai J., Katagiri H., Yamada T., et al. (2005). Constitutively active PDX1 induced efficient insulin production in adult murine liver. Biochem. Biophys. Res. Commun. 326, 402–409 [DOI] [PubMed] [Google Scholar]
- Kaneto H., Matsuoka T.A., Nakatani Y., et al. (2005a). A crucial role of MafA as a novel therapeutic target for diabetes. J. Biol. Chem. 280, 15047–15052 [DOI] [PubMed] [Google Scholar]
- Kaneto H., Nakatani Y., Miyatsuka T., et al. (2005b). PDX-1/VP16 fusion protein, together with NeuroD or Ngn3, markedly induces insulin gene transcription and ameliorates glucose tolerance. Diabetes 54, 1009–1022 [DOI] [PubMed] [Google Scholar]
- Kaneto H., Miyatsuka T., Fujitani Y., et al. (2007). Role of PDX-1 and MafA as a potential therapeutic target for diabetes. Diabetes Res. Clin. Pract. 77Suppl 1, S127–S137 [DOI] [PubMed] [Google Scholar]
- Klinguer C., Beck A., De-Lys P., et al. (2001). Lipophilic quaternary ammonium salt acts as a mucosal adjuvant when co-administered by the nasal route with vaccine antigens. Vaccine 19, 4236–4244 [DOI] [PubMed] [Google Scholar]
- Klinguer-Hamour C., Libon C., Plotnicky-Gilquin H., et al. (2002). DDA adjuvant induces a mixed Th1/Th2 immune response when associated with BBG2Na, a respiratory syncytial virus potential vaccine. Vaccine 20, 2743–2751 [DOI] [PubMed] [Google Scholar]
- Kojima H., Fujimiya M., Matsumura K., et al. (2003). NeuroD-betacellulin gene therapy induces islet neogenesis in the liver and reverses diabetes in mice. Nat. Med. 9, 596–603 [DOI] [PubMed] [Google Scholar]
- Kroon E., Martinson L.A., Kadoya K., et al. (2008). Pancreatic endoderm derived from human embryonic stem cells generates glucose-responsive insulin-secreting cells in vivo. Nat. Biotechnol. 26, 443–452 [DOI] [PubMed] [Google Scholar]
- Lieber A., He C.Y., Meuse L., et al. (1997). The role of Kupffer cell activation and viral gene expression in early liver toxicity after infusion of recombinant adenovirus vectors. J. Virol. 71, 8798–8807 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Limbert C., Path G., Ebert R., et al. (2011). PDX1- and NGN3-mediated in vitro reprogramming of human bone marrow-derived mesenchymal stromal cells into pancreatic endocrine lineages. Cytotherapy 13, 802–813 [DOI] [PubMed] [Google Scholar]
- Makino S., Kunimoto K., Muraoka Y., et al. (1980). Breeding of a non-obese, diabetic strain of mice. Jikken Dobutsu 29, 1–13 [DOI] [PubMed] [Google Scholar]
- McCaffrey A.P., Fawcett P., Nakai H., et al. (2008). The host response to adenovirus, helper-dependent adenovirus, and adeno-associated virus in mouse liver. Mol. Ther. 16, 931–941 [DOI] [PubMed] [Google Scholar]
- Moriyama H., Wen L., Abiru N., et al. (2002). Induction and acceleration of insulitis/diabetes in mice with a viral mimic (polyinosinic-polycytidylic acid) and an insulin self-peptide. Proc. Natl. Acad. Sci. USA 99, 5539–5544 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nir T., Melton D.A., and Dor Y. (2007) Recovery from diabetes in mice by beta cell regeneration. J. Clin. Invest. 117, 2553–2561 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rastellini C., Shapiro R., Corry R., et al. (1997). An attempt to reverse diabetes by delayed islet cell transplantation in humans. Transplant. Proc. 29, 2238–2239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riedel M.J., Gaddy D.F., Asadi A., et al. (2010). DsAAV8-mediated expression of glucagon-like peptide-1 in pancreatic beta-cells ameliorates streptozotocin-induced diabetes. Gene Ther. 17, 171–180 [DOI] [PubMed] [Google Scholar]
- Roh H.J., Sung H.W., and Kwon H.M. (2006). Effects of DDA, CpG-ODN, and plasmid-encoded chicken IFN-gamma on protective immunity by a DNA vaccine against IBDV in chickens. J. Vet. Sci. 7, 361–368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shapiro A.M., Lakey J.R., Ryan E.A., et al. (2000). Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N. Engl. J. Med. 343, 230–238 [DOI] [PubMed] [Google Scholar]
- Shin S., Li N., Kobayashi N., et al. (2008). Remission of diabetes by beta-cell regeneration in diabetic mice treated with a recombinant adenovirus expressing betacellulin. Mol. Ther. 16, 854–861 [DOI] [PubMed] [Google Scholar]
- Tang D.Q., Cao L.Z., Burkhardt B.R., et al. (2004). In vivo and in vitro characterization of insulin-producing cells obtained from murine bone marrow. Diabetes 53, 1721–1732 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taniguchi H., Yamato E., Tashiro F., et al. (2003). beta-cell neogenesis induced by adenovirus-mediated gene delivery of transcription factor pdx-1 into mouse pancreas. Gene Ther. 10, 15–23 [DOI] [PubMed] [Google Scholar]
- Thorel F., Nepote V., Avril I., et al. (2010) Conversion of adult pancreatic alpha-cells to beta-cells after extreme beta-cell loss. Nature 464, 1149–1154 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Rooij E.M., Glansbeek H.L., Hilgers L.A., et al. (2002). Protective antiviral immune responses to pseudorabies virus induced by DNA vaccination using dimethyldioctadecylammonium bromide as an adjuvant. J. Virol. 76, 10540–10545 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang A.Y., Ehrhardt A., Xu H., et al. (2007). Adenovirus transduction is required for the correction of diabetes using Pdx-1 or Neurogenin-3 in the liver. Mol. Ther. 15, 255–263 [DOI] [PubMed] [Google Scholar]
- Wideman R.D., Yu I.L., Webber T.D., et al. (2006). Improving function and survival of pancreatic islets by endogenous production of glucagon-like peptide 1 (GLP-1). Proc. Natl. Acad. Sci. USA 103, 13468–13473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yechoor V., Liu V., Espiritu C., et al. (2009). Neurogenin3 is sufficient for transdetermination of hepatic progenitor cells into neo-islets in vivo but not transdifferentiation of hepatocytes. Dev. Cell 16, 358–373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Q., Brown J., Kanarek A., et al. (2008). In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature 455, 627–632 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.