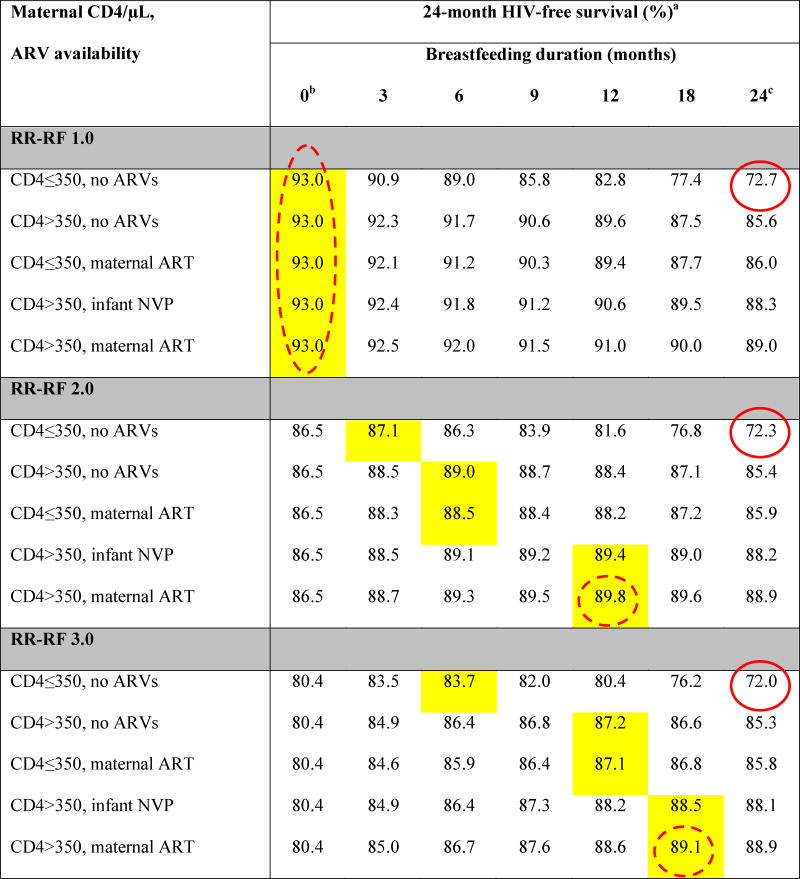
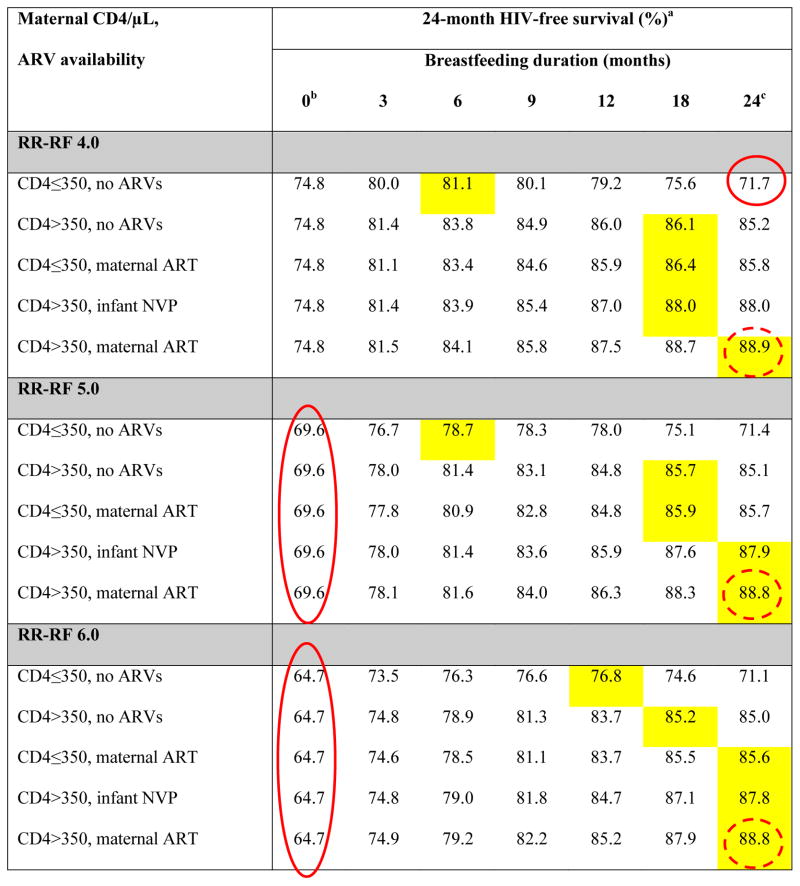
Table 2.
Base-case results: Projected 24-month HIV-free survival among HIV-exposed, uninfected infants at birth
|
|
ARVs: antiretroviral drugs; ART: 3-drug antiretroviral therapy; NVP: nevirapine; RR-RF: relative risk of mortality among replacement-fed compared to breastfed infants (see Methods).
Yellow shading indicates the maximum value of HIV-free survival (HFS) for each unique combination of RR-RF value, maternal CD4 count and ARV availability. Circled values are the high HFS (solid circles) and low HFS (dashed circles) for each value of RR-RF.
When breastfeeding duration is 0 months, HFS is equivalent for all CD4/ARV categories for each value of RR-RF. Because we assume no MTCT risk with replacement feeding from birth, HFS is determined solely by mortality, which depends on RR-RF, but not on maternal CD4 or ARV availability.
In all scenarios, the RR-RF is not applied to infants who are breastfed for 24 months (weaning occurs at the end of the simulation). For each combination of maternal CD4 and ARV availability, HFS at breastfeeding durations of 24 months decreases slightly as the RR-RF increases (for example, with CD4>350 and maternal ART, HFS decreases from 89.0 at RR-RF=1.0 to 88.8 at RR-RF=6.0). This occurs because infants necessarily wean if maternal mortality occurs, and the RR-RF then takes effect for the small proportion of infants whose mothers have died.
