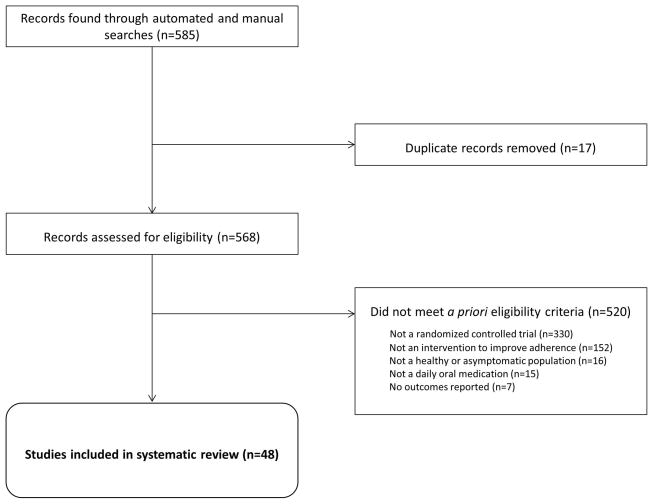
Figure 1. Identified studies of interventions to support adherence to daily oral medications prescribed in healthy or asymptomatic populations.
Eligibility criteria were as follows: 1) randomized controlled trial; 2) at least one adherence outcome; 3) designed to support adherence to a daily oral medication in healthy or asymptomatic populations; 4) conducted in the following clinical fields: hypertension, latent tuberculosis infection, hyperlipidemia, oral contraceptives to prevent pregnancy, osteoporosis, malaria prophylaxis, and post-exposure prophylaxis for HIV infection.