Figure 3.
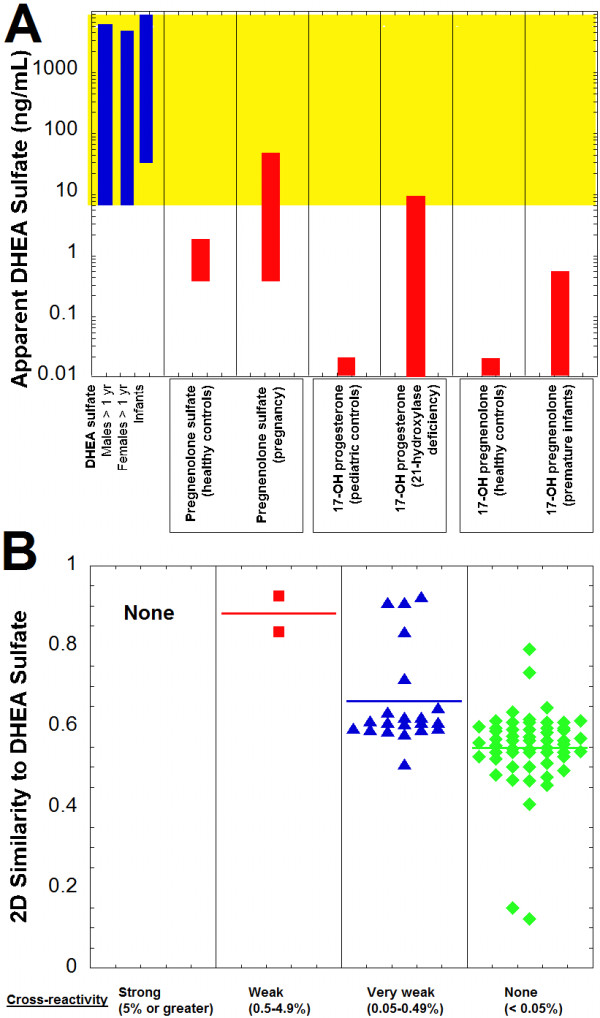
DHEA sulfate immunoassay cross-reactivity and similarity predictions. A. The plot shows the DHEA sulfate reference range for males greater than 1 year old, females greater than 1 year old, and infants in comparison to the predicted apparent DHEA sulfate concentrations produced on the Roche Elecsys DHEA sulfate assay by pregnenolone sulfate (healthy controls and in pregnancy), 17-hydroxyprogesterone (pediatric controls and patients with 21-hydroxylase deficiency), and 17-hydroxypregnenolone (healthy controls and premature infants). Table 2 contains the concentration ranges and percent cross-reactivity values from which the estimated apparent DHEA sulfate concentrations are derived. B. Two-dimensional similarity of compounds to DHEA sulfate is shown, sorted by degree of cross-reactivity in the Roche DHEA sulfate assay (horizontal line in each column indicates average similarity within that group). Similarity values vary from 0 to 1, with 1 being maximally similar. The compounds are subdivided into categories of strong cross-reactivity (5% or greater, black circles), weak cross-reactivity (0.5-4.9%, red squares), very weak cross-reactivity (0.05-0.49%, blue triangles), and no cross-reactivity (<0.05%, green diamonds) to the Roche DHEA sulfate assay (complete list of compounds and associated cross-reactivities and 2D similarities is in Additional file 1).
