Abstract
This review summarizes and integrates our current understanding of how sea stars make gametes. Although little is known of the mechanism of germ line formation in these animals, recent results point to specific cells and to cohorts of molecules in the embryos and larvae that may lay the ground work for future research efforts. A coelomic outpocketing forms in the posterior of the gut in larvae, referred to as the posterior enterocoel (PE), that when removed, significantly reduces the number of germ cell later in larval growth. This same PE structure also selectively accumulates several germ-line associated factors – vasa, nanos, piwi – and excludes factors involved in somatic cell fate. Since its formation is relatively late in development, these germ cells may form by inductive mechanisms. When integrated into the morphological observations of germ cells and gonad development in larvae, juveniles, and adults, the field of germ line determination appears to have a good model system to study inductive germ line determination to complement the recent work on the molecular mechanisms in mice. We hope this review will also guide investigators interested in germ line determination and regulation of the germ line in how these animals can help in this research field. The review is not intended to be comprehensive – sea star reproduction has been studied over 100 years and many reviews are comprehensive in their coverage of, for example, seasonal growth of the gonads in response to light, nutrient, and temperature. Rather the intent of this review is to help the reader focus on new experimental results attached to the historical underpinnings of how the germ cell functions in sea stars with particular emphasis to clarify the important areas of priority for future research.
Keywords: Sea star, starfish, posterior enterocoel, germ line, primordial germ cell
1) Introduction
Sea stars are some of the more recognizable animals for the general public. From Patrick on “Spongebob Square Pants”1, to decorative icons on dishware and shower curtains, sea stars hold a special place in the world. The generic body plan linked to this animal, that is, of a central body disc with five symmetric arms emanating from the disk is in fact a prevalent body plan for this popular order of Echinoderms. But morphology in the order is greatly diverse; some adults have upwards of dozens of arms (e.g. the sun-star, Heliaster helianthus), arms that are fused (e.g. the batstar, Pateria miniata), arms that are barbed (e.g. the crown of thorns sea star, of the Acanthaster genus), and arms of a myriad of colors (the reader is referred to the excellent website of echinoderms Echinoblog [www.echinoblog.com] and the World Asteroidea database (http://www.marinespecies.org/asteroidea/) for a view of this diversity].
Sea stars2 also have a special place in the ecosystem. They are common predators in a variety of marine habitats. They usually occupy the top level of the food chain in the benthic ecosystem and are in some cases keystone species (e.g. Pisaster ochraceus; defined as consumers having a large effect on communities relative to their abundance e.g.(Menge and Sanford, 2013). Sea stars have several characteristics that underlie this importance in the ecosystem. They are relatively large compared to most prey of the ecosystem, they have indeterminate growth and are thereby “rewarded” for high prey consumption, their digestive systems can handle a variety of foods, they have relatively rapid reproduction and development, high fecundity, and their mode of locomotion enables their stability in a variety of physical challenges in the environment of currents, intertidal motions, and wave actions (Menge and Sanford, 2013). Many animals are prevalent in varied habitats but seldom are they as successful as sea stars.
Echinoderms are an ancient taxon, first appearing in the fossil record approximately 550 Mya (Porter, 1977) (Greenstein, 1993) (Kroh and Smith, 2010) (Kroh, 2007) (Erwin et al., 2011) (Smith et al., 2013). The ancient echinoderms appeared to diversify rapidly, with four distinct body plans appearing within the first 10–15 million years following their first appearance (Smith et al., 2013). This rapid diversification was then amplified over the subsequent 500 million years, making it difficult to accurately determine the phylogentic relationship between extant echinoderms, especially within sea stars.
In the midst of this wonderful diversity are many common characters for which sea stars are used for basic research in cellular, molecular, and developmental biology. The broadcast spawning of large numbers of oocytes is valuable for easy laboratory culturing of single to millions of synchronous embryos (Figure 1). Many of the species yield large eggs (>200 microns) of optical clarity enabling micromanipulations/microinjections and in vivo analysis (see for example (Foltz et al., 2004) and (Jaffe and Terasaki, 2004) for more details on these procedures). Uniquely though in the sea stars are the experimental controls possible over oocyte maturation. Adult females store gametes as germinal vesicle staged oocytes (prophase of meiosis I) which can be stimulated to re-initiate meiosis by the natural inducer, 1-methyl-adenine (commercially available). This means that oocytes can be studied effectively for mechanisms of meiotic progression, or the oocytes can be preloaded with biological compounds to examine developmental mechanisms in the resulting embryo. More recently it was shown that the abundance of specific proteins in the oocyte can reduced (knocked down) by conventional morpholino antisense oligonucleotides (MASO) injection into the oocyte followed by culturing for 12–48 hrs (Oulhen et al., 2014). In this way, sea star oocytes can be studied more effectively than even sea urchins for meiosis or early embryogenesis, and coupled with the ability to reactivate meiotic progression with high synchrony, enables a special research tool into basic biological features in the oocyte-to-embryo transition. Another feature that is special in these echinoderms is regeneration; the embryos, larvae, and adults are remarkably capable of replacing missing pieces and developing compensatory cellular arrangements and fates into a normal structure.
Figure 1.

Adult sea star (Asterina miniata) shedding several million eggs. These cells are spawned having begun the process of meiosis and are fertilizable for the next few hours. The gonads line the sides of each arm and the gonopores (site of gamete release, asterisk) from each gonad is at the base of the body. Thus, the gonopores from adjacent arms are close to each other but shed oocytes from distinct gonads (double arrow pointing to the sites of the two adjacent gonopores). From (Wessel et al., 2010)
2) Sea stars as an important model of regeneration
Sea stars are renowned for their strong regenerative abilities. Most, if not all sea stars are able to replace body parts following their injury or removal (Figure 2). Many sea stars are capable of autonomously casting off or detaching an arm following stress or injury to the limb and then growing the structure back with correct proportionality. In some species, such as the sea star from the Linckia genus, the self-detached (autotomized) arms may even regenerate to form a completely new adult (Cuenot, 1948). Most species, however, require an intact central disk (body) for their complete regeneration. Then, they are capable of regenerating most of the normal functional appendage including its spines, tube feet, and pedicellariae as well as its internal organs such as the gut, nervous system, and even gonad, complete with germ cells (Emson and Wilkie, 1980; Hyman, 1955)..
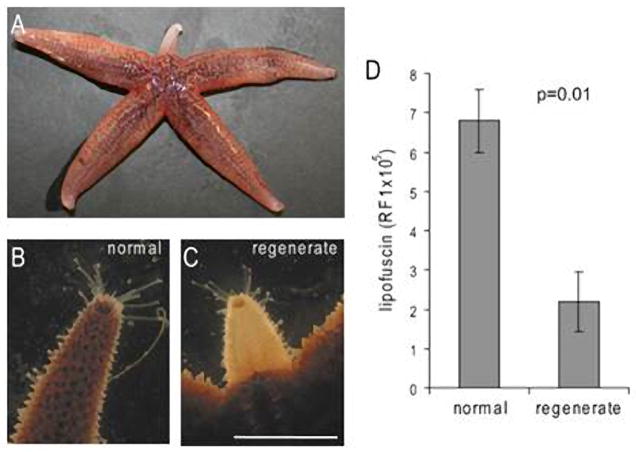
Figure 2.
Asterias rubens with an arm regenerating from the base (A–C). The early regenerate (C) is less pigmented than an intact arm of the same animal (B). Fluorometrical analysis of extracted lipofuscin content shows lower levels of lipofuscin in regenerating arms than in the tips of corresponding intact arms of the same animals (D). Lipofuscin is expressed as mean+/− s.e.m. Relative fluorescence intensities (RFI). R, regenerate; N, normal/intact arm. Light micrographs and data were obtained from animals with approximately 1 cm regenerates. Data obtained from animals with 1.5–2 cm regenerates. Scale bar: 1 cm (B, C). From (Hernroth et al., 2010).
Sea star larvae also have regenerative properties and were the first larvae reported in deuterostomes to completely regenerate all missing body structures, tissues, and organs at the occasion of amputation (Vickery and McClintock, 1998) (Figure 3). This remarkable regeneration process appears to be also supported by an intrinsic mechanism, and the sea star larvae were also first reported for its cloning capability (asexual budding) in natural conditions (Jaeckle, 1994) (Vickery and McClintock, 2000) (Knott et al., 2003). Followed by those findings in sea star larvae, all echinoderm larvae were identified to have remarkable abilities of asexual reproduction through cloning (Evaes and Palmer, 2003). Despite the contributions of starfish larvae initially in regeneration research, knowledge of the cellular and molecular mechanisms of regeneration/cloning is very limited. Several genes have been identified that are differentially expressed during early larval sea star regeneration (Vickery et al., 2001), yet functional analysis and the contributions of each gene during the regeneration process has not been tested. Further, it is not known whether regenerated larvae recover any germ cell function.
Figure 3.
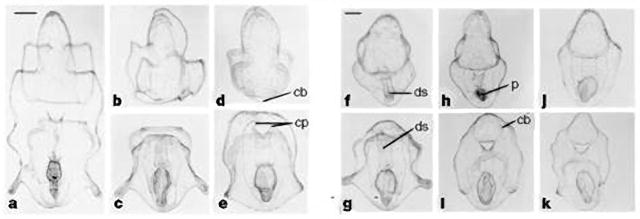
Bipinnaria larvae of P. ochraceus at various stages of regeneration after surgical bisection. The anterior larva is shown in the top row, and the posterior larva in the bottom row. a, Bipinnaria before surgical treatment. b, c, Day 1. Anterior (b) and posterior (c) larvae after surgical division. The gut of the posterior larva is intact enough for feeding. d, Day 3. e, Day 4. Elongation along the larval axis has taken place. f, g, Day 7. The anterior larva (f) has regenerated a functional digestive system. h, Day 9. i, Day 10. Complete regeneration of ciliary bands has occurred in both anterior and posterior larvae. j, k, Day 12. Both larvae have regenerated and are similar to control larvae. cb, ciliary band; cp, coelomic pouch; ds, digestive system; p, phytoplankton. a–e, Scale bar (in a), 200 μm. f–k, Scale bar (in f), 200 μm. From (Vickery and McClintock, 1998)
3) Environmental impact on reproduction
Development of gametes (gametogenesis) in sea stars is highly dependent on the environmental conditions of the animal. These conditions include temperature, light, food availability, age of the animal, salinity of the media, and parasite load. While many of these conditions have been replicated and tested under laboratory conditions, overall the influences of each as they contribute to gametogenesis and gonad growth are only beginning to be resolved. The consensus model is that these conditions likely coordinate the adult physiology and behavior through hormonal regulation. Some of these hormones and mechanisms have been identified (see e.g. (Mercier and Hamel, 2013)). It is clear that the genetic makeup of the animal also has a significant impact on this process. Different species of sea stars may exhibit markedly different reproductive cycles even when living in the same environment, and different populations of the same species may also exhibit different reproductive cyclicities within the same environment. Sea stars can be shifted experimentally in their gondal cyclicity under laboratory conditions by altering the light, temperature, and nutritional conditions (Chia and Walker, 1991). This approach has been successfully applied in adults to understand and to prioritize the environmental conditions that impact the animal. Not surprisingly, the hierarchy of environmental conditions is different for various species and for different times of the year. From these studies it is also obvious that both physiology and behavior are influenced by the environmental conditions. Of particular interest is the fluctuation of steroidal hormones during the seasonality changes (Hines et al., 1992; Lawrence, 2013). Several reviews on environmental impact of reproduction in asteroids are available that very effectively document this biological – environmental relationship (reviewed in e.g. (Chia and Walker, 1991; Mercier and Hamel, 2013)). The results of these studies also suggest the important role of germ line stem cells within the adult. As a broadcast spawner, many sea stars are capable of releasing millions of oocytes (Figure 1), and countless sperm, during their reproductive life. This robust fecund capability implies a very large and constantly renewing source of germ line stem cells, although such cells have not been identified. These (anticipated) cells are likely a target of the hormonal fluctuations impacted by the environment and, were they identified and isolated, would be an excellent cell-type to study regulatory mechanisms of germ line stem cells and the hormonal influences on their proliferation.
4) Origin and development of the germ line in adults
The germ cells appear to accumulate in adult sea stars within the aboral aspect of the haemal system (Figure 4). Prior to development of this structure, and prior to the development of the gonad, the presumptive germ cells are present within the haemal tissue, in a structure called the genital bud (Bouland and Jangoux, 1990), which is located in the most aboral part in the haemal system (in P. pectinifera, Figure 4). The genital bud is a candidate of the source of germ-line stem cells in the adult because they appear in this structure throughout the year, even in post-spawning stages. Since complete regression of the gonad occurs after spawning in many species, annual neo-formation of the gonad must occur from cells within the arms of the adult prior to gametogenesis (Bouland and Jangoux, 1990). During regression and then subsequent proliferation, the gonads remain connected to the haemal sinuses, which are not lost after spawning (in Asterias pectinifera, now Patiria pectinifera, (O’Loughlin and Waters, 2004). This connection is a viable source of germ-line stem cells for the next breeding season, in addition to any remaining gonia within the spent gonads (Inoue and Shirai, 1991). Thus, the replenishment of stem cells for each round of gametogenesis likely includes germ cell migration into the regenerating gonads from the stem of the haemal system, and proliferation of remaining gonia within the gonadal remnant.
Figure 4.
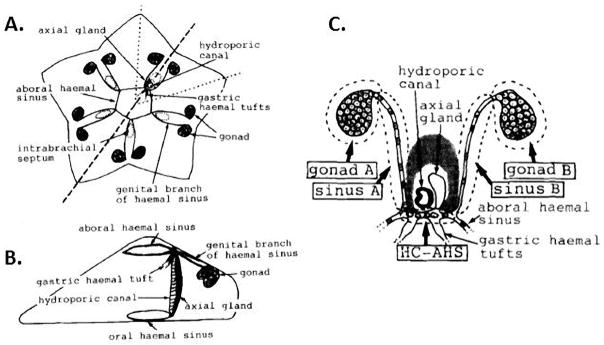
Schematic diagrams of the relationships between the gonad and various haemal sinuses in A. pectinifera. A. Oral view. The gonads are connected to each other by genital branches of the aboral haemal sinus. Gastric haemal tufts connect with pyloric caeca. The broken line shows the position of thte semi-cross section for a lateral view. The dotted line shows the regions shown in Part C. B. Lateral view. The aboral haemal sinus connects with the oral haemal sinus through the axial gland. C. Schematic diagram of the genital region. Aboral view. The arrangement of the areas in dotted lines show many cells left out of this diagram for clarity. HC-AHS; aboral haemal sinus near the hydroporic canal, sinus A and B, genital branches of haemal sinus. Shaded area surrounding HC and axial gland indicates the intrabrachial septum. (From (Inoue and Shirai, 1991))
Inoue and Shirai examined the appearance of germ cells during ontogeny of the sea star, P. pectinifera, by electron microscopic histological approaches (Inoue and Shirai, 1991). Germ-line stem cells (gonia) were first identified in gonads following spawning: the gonia are larger than somatic cells, have large nuclei with electron-lucent nucleoplasm, and contain mitochondrial aggregates associated with electron-dense bodies (much like nuage). Similar cells were detected in young sea stars before gonad formation (6 mm) or even in the final stage of larval development (4 weeks) and in metamorphosed juveniles (10 weeks). They could detect germ cells as a cellular cluster in the coelomic epithelium near the hydroporic canal (see Figure 4). Quantitation of the germ cells in each region indicated that the migratory path of these putative PGCs is through the haemal system to the gonads during normal development (hence also a model for populating regenerating/cycling gonads with gonia). The central aspect of the haemal system is an oral and aboral ring connecting the axial sinus running along the hydroporic canal. Ten genital branches (two for each arm) extend from the aboral ring to the gonads. Germ cells migrate in a cluster from the aboral haemal sinus near the hydroporic canal (a part of aboral haemal ring), through the genital branches, and to the gonads. In other starfish (e.g. Asterina gibbosa), gonia were also observed in the vicinity of the hydropholic canal a short time before metamorphosis (Delavault, 1966). Thus, the germ line stem cells of the organism appear to under seasonal migration from a distant site into the growing gonad. The genital bud appears to be formed from the accumulation of these cells in the juvenile and the germ cell cluster observed in the late larvae of P. pectinifera is thought to correspond to this genital bud. In Leptasterias hexactis, which has large eggs (a mean of diameter 0.8 mm) and exhibits modified development through a non-feeding larva compared to indirect developing species, germ cells were observed first above the dorsal horn of the left posterior coelom near the aboral portion of the stone canal before metamorphosis, then they subsequently invade the haemal sinus (Chia, 1968). It is still not clear what the origin is of these germ cells but the classical morphological appearance of germ cells in late larvae is consistent in asteroids.
5) Origin of the germ line during embryogenesis – the posterior enterocoel (PE)
The origin of the germ line in many sea stars appears to be within the posterior coelom (Inoue, 1992). In sea star embryos, coelomic pouch formation begins during late gastrulation. Chia and Walker (Chia and Walker, 1991) distill the diverse developmental patterns used by asteroids to develop their various coeloms into three general systems (Figure 5). In Type 1 (our terminology to facilitate this discussion) two coelomic pouches arise from the anterior of the archenteron and grow posteriorly, one on the left side and one on the right side of the gut (Figure 5a). The left coelom grows much faster and opens to the outside via a hydroporic canal, the hydrocoel, which gives rise to the water-vascular system during larva development. This is the most familiar type of coelom where popular research species such as Luidia, Astropecten, Asterias, and Pisaster are found. In Type 2 coelom development, coeloms arise from the anterior of the archenteron and grow posteriorly on both sides as a U-shaped coelom. Later in development, two septa form on each side of the “U” to form the anterior coelom and two lateral posterior coeloms (left and right). The left lateral coelom grows much faster and a part of it will become the hydrocoel (Figure 5b). Species as Parvulastra exigua (formerly Patiriella exigua), Asterina gibbosa, and Leptasterias hexactis belong to this group. In the Type 3 system, the coeloms arise from both the anterior and posterior ends of the archenteron (Chia and Walker, 1991). This mode of coelomogenesis is used in development of Patiria miniata, Patiriella regularis, Pateria pectinifera, Pteraster tesselatus, and A. glacialis (Marthasterias glacialis), some of the more intensively studied species for molecular mechanisms of germ-line determination. In these animals the left posterior coelomic pouch (Posterior enterocoel or PE) arises first from the dorsal side of the archenteron and then shifts to the left side and buds off the left endodermal epithelium (Figure 7c), in an manner independent and physically separated from the anterior coeloms (Bury, 1889; David et al., 1994; Gemmill, 1914; Heath, 1917; Hinman et al., 2003; Kuraishi and Osanai, 1992; McEdward, 1995; Newman, 1925). Observations made by Newman described PE formation as a thick-walled evagination of the wall of the archenteron, which did not expand into a thin-walled vesicle, but merely constricted off a small solid ball of cells that later expanded and formed a lumen (Newman, 1925). Deviations from this mode of coelomogenesis were noted in earlier descriptions of other Asteroidea larval development. For example, in 1914 Gemmill noticed that the PE in the sea star Asterias rubens also arose periodically from the right side of the gut and also medially (Gemmill, 1914). These observations were in contrast to observations in Porania pulvillus, where the PE arose dorsally on both sides of the median line, but sometimes in the median line, or on one side only, occurring rather more often on the right than on the left side (Gemmill, 1914). Lineage analysis of each of the blastomeres of an eight cell embryo, documented the formation of the PE from the mid-dorsal surface of the archenteron in Patiria pectinifera. These cells originated from two different eight cell blastomeres and shifted in concert to the left side of the archenteron (Kuraishi and Osanai, 1992). The expression pattern resulting from an endoderm (or archenteron) marker shows that the PE never expressed this marker suggesting that the mechanism of commitment of the PE is different than that of the endoderm (Kuraishi and Osanai, 1994). On the other hand, the direct action of toxic contaminant agents affected coelomic formation in P. miniata (Child, 1941). The effect of temporary exposure to moderate concentration of these chemical agents (such as lithium chloride, nile blue sulphate, acidified sea water, hypotonic sea water) was development of several posterior coeloms (from two to six), at the same level as the original PE and equally spaced. A longer exposure of these agents inhibited posterior coelom development completely (Child, 1941). These results suggest that formation of the PE is dependent on inductive cellular interactions and suggests that distinct from the sea urchin, the germ line may originate from cellular communication during gastrulation.
Figure 5.
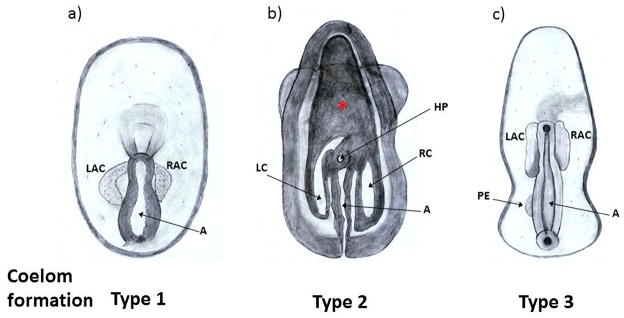
Different patterns for coelomic development in Asteroids. Schematic representation of the three different types of coelomic pouch formation. Dorsal view of larvae. a) 4-Day bipinaria larva from Pisaster ochraceus shows two lateral bulges derived from the anterior part of the archenteron which become the right and left enterocoels. b) In 4-Day brachiolaria larva of Parvulastra exigua, coeloms are conformed by three parts, an anterior coelom which extends from the archenteron, and two septa formed from the anterior coelom which become the left lateral coelom and the right lateral coelom. The anterior coelom and the archenteron are formed by a continuous cavity which can be seen in a ventral view of the larva. The left lateral coelom is larger than the right lateral coelom at the level of their junction with the archenteron. The hydropore is close to where the left lateral coelom joins the archenteron cavity. A red asterisk represents the localization of the anterior coelom in a ventral view of the larva (Morris et al., 2009). c) In 2.5-Day bipinaria larva from Patiriella regularis, right and left enteroels form from the expanded tip of the archenteron and a small group of cells forms the posterior enterocoel (Byrne and Barker, 1991). A: archenteron; LAC: left anterior enterocoel; RAC: right anterior enterocoel; LC: left lateral coelom; RC: right lateral coelom; HP: Hydropore; PE: Posterior enterocoel.
6) Evidence that the PE is a source of germ cells in the sea star
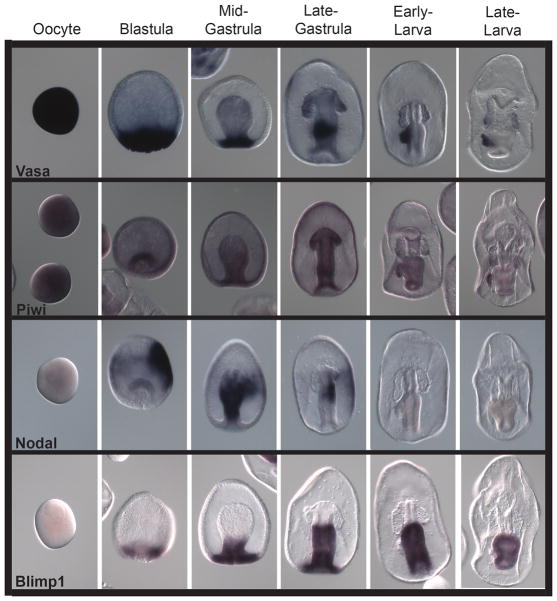
Several complementary lines of evidence suggest that the PE contributes to the germ line of the sea star. These include: 1) PE removal experiments that result in larvae with significantly fewer germ cells (Inoue, 1992), 2) Vasa immunolabeling experiments show the PE is the first restricted site of Vasa protein localization (Juliano and Wessel, 2009), 3) conserved germ line factors accumulate selectively in the PE e.g. nanos and piwi in addition to vasa (Figure 6) (Fresques et al., 2013), and 4) the PE exhibits rapid depletion of mRNAs encoding factors involved in somatic cell fates e.g. Blimp1 (Fresques et al., 2013). None of this evidence on its own is sufficient to conclude that the PE is the germ line, or that only primordial germ cells are in the PE, but in concert, the evidence is sufficiently compelling to conclude that the PE contributes to the germ line.
Figure 6.
Genes associated with germ-line determination. Vasa and piwi are factors often associated with germ cell development, and these markers become enriched in the PE in early larval stages. Nodal is a TGF-beta signaling model and is present in the right side of the embryo, and gut, opposite of the PE. We hypothesize that nodal signaling may direct vasa and piwi localization, and perhaps the asymmetry of the PE. Blimp1 is a transcription factor important for somatic endomesoderm formation and is markedly enriched in the gut during gastrulation. Importantly, it is excluded from the PE when it is formed, at a time when vasa and piwi are enriched in the PE, suggesting that clearance of somatic determinants is important in PE formation. Oocytes of this sea star (P. miniata) are ~200 microns in diameter. From (Fresques et al., 2013)
Critical analysis of each aspect of this evidence helps identify shortcomings in this conclusion. In Inoue et al., (Inoue, 1992) the investigators removed cells of the PE from the larvae (at 48 hrs) and analyzed the resultant larvae several weeks later. Previous evidence (see above) suggested that the germ cells are present in the coelomic epithelium near the hydroporic canal and contained characteristic features of germ cells in the transmission electron microscope: large nuclei, mitochondria aggregates, distinctive chromatin condensation along the nuclear envelope, less cytoplasmic organelles than nearby somatic cells. Older morphological studies that identified PGC’s with classical germ cell characteristics are consistent with this recent data because the origin of the germ line was described to be in the “dorsal horn of the left somatocoel of the bipinnaria larvae”, which is interpreted to be derived from cells in the PE (Chia and Walker, 1991; Gemmill, 1914). In larvae resulting from PE removal, the number of these phenotypic cells in the hydroporic region was significantly reduced, and retained only about 35–40% the value of the control larvae. While the loss did not reach 100 % of the cells, several qualifiers should be considered: 1) The assay for detection was microscopic (TEM) analysis of cellular morphology. This assay is highly detailed, but less quantitative than current approaches of detecting germ line markers by immunolabeling, by in situ RNA hybridization, by GFP-recordings, or by qPCR analysis. Further, the tissue sections needed for TEM may have missed displaced cells as a result of the surgical operation for PE removal. 2) The effectiveness of PE removal – PGCs have strong proliferative abilities especially later in development and were the investigators to have missed removing a single PE cell, it may have had the ability to proliferate and even compensate the lack of sibling cells. 3) The PE is not the sole source of PGCs and may continually accumulate over larval development. Mice have (at least) two phases of primordial germ cell determination and although much closer in developmental time, it does suggest that PGC may be fated more broadly than at a distinct point in time (Hayashi et al., 2007). 4) Compensatory PGCs. The sea urchin has an ability to make new germ cells following removal of the precursor cells of the PGC (Ransick et al., 1996; Voronina et al., 2008; Yajima and Wessel, 2011). Perhaps trans-fating of multipotent cells within the larvae to a PGC is one way that the larvae maximizes its germ line potential, and/or retains multipotency beyond the initial period of specification. This explanation is particularly relevant in sea stars, an animal with remarkable regenerative capacities (see section #2 above).
The PE is the site of enriched germ line markers. This too is suggestive of the germ line originating in the PE. In the past decade though, many non-model organisms have been shown to have germ line markers in many non-germ line cells. Moreover, these markers are often in multipotent cells than can (or do) give rise to the germ line. Therefore, although the results support the contention that the PE is the site of germ cells, the PE may instead be a highly multipotent cell that has the developmental capabilities to give rise to many cell types. A fine lineage analysis is important to accomplish on the PE cells, all ~25 of them, in order to understand what diversity of fates these cells may contribute. The point of understanding the fates of all the cells (not necessarily each individual cell) is because recent evidence suggests the cell in the PE are heterogeneous, at least by SoxE accumulation (Fresques et al., 2013). Thus, the fate(s) of the PE in general should be tested.
Finally, the rapid turnover of mRNAs of genes involved in somatic differentiation in the PE is consistent with the PE fate of germ cells. Blimp is an important germ line factor in mice (Ancelin et al., 2006) but is a somatic factor in sea urchins and sea stars (Hinman and Davidson, 2003; Livi and Davidson, 2006) for endomesodermal cell fates, not germ line fates. As recently shown in mice, an organism relying strongly on inductive mechanisms of germ line determination, the cells that become the germ line first have to exclude mRNAs and gene activity responsible for somatic cell fates. Were the cells to not receive the critical germ line signals, these cells in the mouse instead would develop into mesoderm. Much of this transition in the future germ cell is in changes in the epigenetic landscape, making areas of the genome encoding somatic cell fates heterochromatic and shifting the balance to inductive-based gene expression important for these germ cells. Currently, we do not know how Blimp is excluded from the PE so rapidly, but it is hypothesized that it includes a change in the epigenetics of the PE cells, and a selective turnover of its mRNA (Fresques et al., 2013). Blimp is not the only somatic factor excluded from the PE but it is a robust marker making this analysis more tractable.
Most animals reveal one or another of two broad and overlapping mechanisms of germ line determination, inductive or inherited (also referred to as acquired) (Ewen-Campen et al., 2010; Extavour and Akam, 2003; Juliano and Wessel, 2010; Seervai and Wessel, 2013). Some animal embryos acquire partitioned information stored in the egg that directs the fate of the cells along the path of the germ line, the inherited mechanism. The fruit fly, Drosophila, and the roundworm C. elegans are good examples of this mechanism in play. Although only a few molecular candidates are known to be important in the acquired material, the structures within which they reside are apparent. These are referred to as nuage, perinuclear granules, or germ plasm, depending on the animal. The mechanism in the opposite extreme is where cells signal amongst themselves and germ cells are somehow diverted from somatic fate to become the germ line. This is usually referred to as inductive germ line determination, and the sea star appears to utilize this mechanism in the cells of the PE. We hypothesize that the PE forms by inductive mechanisms based on the timing of PE formation - it forms relatively late in development, during late gastrulation. It would be hard to imagine how maternal factors could be so tightly partitioned during this prolonged development (from 1-to- thousands of cells at the time of PE formation) to be a consideration in an acquired germ line mechanism. This conclusion of inductive germ line behavior would be important experimentally since currently very few animals are amenable to study that use inductive mechanisms, the mouse being the most prominent. Yet in mice, the embryo is embedded in the uterus during this important time of PGC generation, making embryonic, cellular, and molecular analysis more challenging. The genetic applications in mice, along with stem cell analysis in vitro (e.g. iPS, GSC) do offer remarkable benefits, each of which may be complemented by another organism tractable for more direct cellular, molecular, and visual analysis. The sea star may be important for more direct visualization, biochemical analysis of isolated germ cells, and molecular manipulations in germ cells whose fate is determined largely by inductive mechansims.
7) Molecular mechanism of PE formation in the sea star and evolutionary transitions from the sea urchin
In P. pectinifera and P. miniata, the two most intensively used species for studies in PE formation, the PE first arises as a bump on the dorsal gut which then appears to transition to the left side of the gut ((Kuraishi and Osanai, 1992); Fresques “unpublished observations” ). The cellular dynamics of PE formation, however, remain poorly understood. Do these cells undergo an Epithelial to Mesenchymal Transition (EMT) to break away from the dorsal side of the gut and undergo active migration as a cohort to the left side of the gut? Or do they first outpocket as a sheet that migrates leftward and pinches off from the dorsal gut? Detailed microscopic and lineage studies are necessary to understand the molecular mechanisms of PE formation and morphogenesis and a live marker of PE cells would be valuable to answer these questions using live-fluorescence microscopy.
Although the mechanism of the transition of the PE is poorly understood, conserved molecules involved in Left/Right asymmetry may be involved. For example, at the same time that Vasa mRNA is restricted to the left side of the gut, Nodal mRNA is restricted to the right side of the gut (Fresques et al., 2013). This is significant because Nodal signaling is required for the correct specification of Left/Right asymmetries in many animals (e.g.(Vandenberg and Levin, 2013)). How Nodal signaling on the right side of the gut inhibits the accumulation of Vasa positive cells in the right remains unclear. Nodal signaling may cause the PE cells in the right 1) to migrate to the left, 2) to lose Vasa mRNA, or 3) to undergo programmed cell death. These hypotheses may not be mutually exclusive as is the case with the impact of L/R asymmetry on the accumulation of putative PGC’s in sea urchins.
In sea urchins, Nodal signaling appears to influence the sMic’s (putative PGC’s), as to whether they accumulate in either the left or right coelomic pouch. Sea urchins do not have posterior coelomic pouches and only have two anterior coelomic pouches. During gastrulation the sMics translocate from the vegetal pole, to the tip of the archenteron, and finally accumulate in the coelomic pouches (Luo and Su, 2012; Pehrson and Cohen, 1986; Yajima and Wessel, 2012). Normally they exhibit a Left/Right asymmetry in regards to how many germ cells first migrate to each coelomic pouch and normally an average of 5 cells migrate to the left coelomic pouch and 3 cells migrate to the right coelomic pouch (Campanale and Hamdoun, 2012; Pehrson and Cohen, 1986; Tanaka and Dan, 1991). In addition, by the pluteus stage all of the sMics that have translocated to the right coelomic pouch undergo apoptosis while many (all?) of the sMics that have ended up in the left coelomic pouch survive (Campanale and Hamdoun, 2012; Luo and Su, 2012). This Left/Right asymmetry is dependent on Nodal signaling. Nodal is active in the right coelomic pouch of the sea urchin embryo and is required for the apoptosis of sMic cells in the right (Luo and Su, 2012).
Were Nodal to have similar effects in both sea urchins and sea stars we hypothesize that Nodal first inhibits germ cells from accumulating in the dorsal-right by influencing their directional translocation and then clears any remaining right-sided germ cells via programmed cell death. However, since Vasa accumulates in an entire structure (the PE) in the sea star and only 8 cells (the sMics) in the sea urchin it is likely that Nodal has an extra role in sea stars in regards to posterior coelomic pouch (PE) formation and translocation that was lost in sea urchins with the loss of the posterior coelomic pouches.
It is important to note that disruption of Left/Right asymmetry signals in sea urchins does not affect the accumulation of Nanos, Vasa, or Piwi in sMics (Luo and Su, 2012), a testament that these cells have already been committed to a germ lineage. It will be important to test if Left/Right asymmetry signaling molecules have a similar effect in sea stars. If these signals are involved in acquisition or loss of the germ cell fate and not just migration then we can use this system to understand how signals actively promote or inhibit the germ cell specification network.
8) Conclusions and future directions
The utility of studies on reproduction in the sea star is broad. From regeneration of their gonads and germ cells, to the environmental impact on the germ line growth, to its formation in larvae and replenishment in cycling adults, to the more recent molecular revelations in the PE formation, the sea star has many unique and complementary biological features that make them valuable for studies in reproduction. The diversity of these animals is also a distinct asset – especially in the reproductive mechanisms in which a comparative approach has many revelations for extracting consensus mechanisms that can be used for prioritization of more detailed studies. With the rapid expansion of genomic resources in this group of animals, coupled with the advanced imaging capabilities for embryos with optical clarity and the new methods in transcriptional and post-transcriptional gene disruption, sea stars throughout the world have a great deal of excitement for future work in development and reproduction.
Abbreviations
- PE
posterior enterocoel
- sMics
small micromeres
- hpf
hours post-fertilization
- dpf
days post-fertilization
- MASO
morpholino antisense oligonucleotides
Footnotes
The terms sea stars and starfish refer to the same group of organisms. Since starfish are not actual fish with a backbone, the community is more often referring to them as sea stars, a nomenclature parallel to other some of the other organisms in this phylum, e.g. sea urchins, sea cucumbers, sea lilies, and yet still distinguishable from others, e.g. brittle stars.
References
- Ancelin K, Lange UC, Hajkova P, Schneider R, Bannister AJ, Kouzarides T, Surani MA. Blimp1 associates with Prmt5 and directs histone arginine methylation in mouse germ cells. Nature cell biology. 2006;8:623–630. doi: 10.1038/ncb1413. [DOI] [PubMed] [Google Scholar]
- Bouland C, Jangoux M. Origin of germinal cells and the reproductive cycle of the asteroid Asterias fubens L. (Echinodermata) Invertebr Reprod Dev. 1990;17:97–102. [Google Scholar]
- Bury H. Studies in the Embryology of the Echinoderms. Qrc Sci. 1889:409–449. [Google Scholar]
- Byrne M, Barker M. Embryogenesis and larval development of the asteroid Patiriella regularis viewed by light and scanning electron microscopy. Biological Bulletin. 1991;180:332–345. doi: 10.2307/1542335. [DOI] [PubMed] [Google Scholar]
- Campanale JP, Hamdoun A. Programmed reduction of ABC transporter activity in sea urchin germline progenitors. Development. 2012;139:783–792. doi: 10.1242/dev.076752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chia F. The embryology of a brooding starfish, Leptasterias hexactis (Stimpson) Acta Zool. 1968;49:321–364. [Google Scholar]
- Chia F, Walker C. Echinodermata: Asteroidea. In: Giese J, Pearse J, Pearse V, editors. Reproduction of Marine Invertebrates. The Boxwood Press; Pacific Grove CA USA: 1991. pp. 301–355. [Google Scholar]
- Child C. Differential Modifications of coelom development in the starfish, Patiria miniata. Physiological Zoology. 1941;14:449–460. [Google Scholar]
- Cuenot L. Les Echinodermes. In: Grasse P, editor. Traite de Zoologie. 1948. pp. 1–363. [Google Scholar]
- David B, Guille A, Féral J-P, Roux M. Echinoderms through time. A. A. Balkema; Rotterdam: 1994. [Google Scholar]
- Delavault R. Determination of sex. In: Boolootain R, editor. Physiology of Echinodermata. John Wiley & Sons Press; New York: 1966. pp. 615–638. [Google Scholar]
- Emson R, Wilkie I. Fission and autotomy in echinoderms. Oceanogr Mar Biol Annu Rev. 1980;18:155–250. [Google Scholar]
- Erwin DH, Laflamme M, Tweedt SM, Sperling EA, Pisani D, Peterson KJ. The Cambrian conundrum: early divergence and later ecological success in the early history of animals. Science. 2011;334:1091–1097. doi: 10.1126/science.1206375. [DOI] [PubMed] [Google Scholar]
- Evaes A, Palmer A. Widespread cloning in echinoderm larvae. Nature. 2003;425:146. doi: 10.1038/425146a. [DOI] [PubMed] [Google Scholar]
- Ewen-Campen B, Schwager EE, Extavour CG. The molecular machinery of germ line specification. Mol Reprod Dev. 2010;77:3–18. doi: 10.1002/mrd.21091. [DOI] [PubMed] [Google Scholar]
- Extavour CG, Akam M. Mechanisms of germ cell specification across the metazoans: epigenesis and preformation. Development. 2003;130:5869–5884. doi: 10.1242/dev.00804. [DOI] [PubMed] [Google Scholar]
- Foltz K, Adams N, Runft L. Echinoderm eggs and embryos: procurement and culture. In: Ettensohn C, Wessel G, Wray G, editors. Methods Cell Biology. Academic Press; 2004. pp. 39–73. [DOI] [PubMed] [Google Scholar]
- Fresques T, Zazueta-Novoa V, Reich A, Wessel G. Selective accumulation of germ-line associated gene products in early development of the sea star and distinct differences from germ-line development in the sea urchin. Dev Dynam. 2013 doi: 10.1002/dvdy.24038. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gemmill J. The development and certain points in the adult structure of the starfish Asterias rubens. Phil Trans Royal Soc. 1914:205B. [Google Scholar]
- Greenstein BJ. Is the fossil record of regular echinoids really so poor? A comparison of living and subfossil assemblages. Palaios. 1993;8:587–601. [Google Scholar]
- Hayashi K, de Sousa Lopes SM, Surani MA. Germ cell specification in mice. Science. 2007;316:394–396. doi: 10.1126/science.1137545. [DOI] [PubMed] [Google Scholar]
- Heath H. The early development of a starfish, Pateria (Asterina) miniata. Journal of morphology. 1917;29:461–470. [Google Scholar]
- Hernroth B, Farahani F, Brunborg G, Dupont S, Dejmek A, Sköld H. Possibility of mixed progenitor cells in sea star arm regeneration. J Exp Zool B Mol Dev Evol. 2010;314:457–468. doi: 10.1002/jez.b.21352. [DOI] [PubMed] [Google Scholar]
- Hines G, Watts S, Voogt P, Walker C. Androgen metabolism in somatic and germinal tissues of the sea star Asterias vulgaris Comp. Biochem Physiol. 1992;102B:521–526. doi: 10.1016/0305-0491(92)90043-q. [DOI] [PubMed] [Google Scholar]
- Hinman V, Nguyen A, Cameron R, Davidson E. Developmental gene regulatory network architecture across 500 million years of echinoderm evolution. PNAS USA. 2003;100:13356–13361. doi: 10.1073/pnas.2235868100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinman VF, Davidson EH. Expression of AmKrox, a starfish ortholog of a sea urchin transcription factor essential for endomesodermal specification. Gene Expr Patterns. 2003;3:423–426. doi: 10.1016/s1567-133x(03)00083-8. [DOI] [PubMed] [Google Scholar]
- Hyman H. The Invertebrates: Echinodermata. McGraw-Hill Education; New York: 1955. [Google Scholar]
- Inoue C, Kiyomoto M, Shirai H. Germ cell differentiation in starfish: The posterior enterocoel as the origin of germ cells in Asterina pectinifera. Development Growth & Differentiation. 1992;34:413–418. doi: 10.1111/j.1440-169X.1992.00413.x. [DOI] [PubMed] [Google Scholar]
- Inoue C, Shirai H. Origin of germ cells and early differentiation of gonads in the starfish, Asterina pectinifera. Development Growth and Differentiation. 1991;33:217–226. doi: 10.1111/j.1440-169X.1991.00217.x. [DOI] [PubMed] [Google Scholar]
- Jaeckle W. Multiple modes of asexual reproduction by tropical and subtropical sea star larvae: an unusual adaptation for gene dispersal and survival. Biological Bulletin. 1994;186:62–71. doi: 10.2307/1542036. [DOI] [PubMed] [Google Scholar]
- Jaffe L, Terasaki M. Quantitative microinjection of oocytes, eggs, and embryos. In: Ettensohn C, Wessel G, Wray G, editors. Methods in Cell Biology. Academic Press; 2004. pp. 219–242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juliano C, Wessel G. Versatile Germline Genes. Science. 2010;329:640–641. doi: 10.1126/science.1194037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Juliano CE, Wessel GM. An evolutionary transition of vasa regulation in echinoderms. Evol Dev. 2009;11:560–573. doi: 10.1111/j.1525-142X.2009.00362.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knott K, Balser E, Jaeckle W, Wray G. Identification of asteroid genera with species capable of larval cloning. Biological Bulletin. 2003;204:246–255. doi: 10.2307/1543596. [DOI] [PubMed] [Google Scholar]
- Kroh A. Climate changes in the early to middle miocene of the central paratethys and the origin of its echinoderm fauna. Palaeogeography, Paleoclimatology, Paleoecology. 2007;253:185–223. [Google Scholar]
- Kroh A, Smith AB. The phylogeny and classification of post-Palaeozoic echinoids. Journal of Systematic Palaeontology. 2010;8:147–212. [Google Scholar]
- Kuraishi R, Osanai K. Cell movements during gastrulation of starfish larvae. Biological Bulletin. 1992;183:258–268. doi: 10.2307/1542213. [DOI] [PubMed] [Google Scholar]
- Kuraishi R, Osanai K. Contribution of maternal factors and cellular interaction to determination of archenteron in the starfish embryo. Development. 1994;120:2619–2628. [Google Scholar]
- Lawrence J. Starfish: Biology and Ecology of the Asteroidea. Johns Hopkins University Press; 2013. [Google Scholar]
- Livi CB, Davidson EH. Expression and function of blimp1/krox, an alternatively transcribed regulatory gene of the sea urchin endomesoderm network. Developmental biology. 2006;293:513–525. doi: 10.1016/j.ydbio.2006.02.021. [DOI] [PubMed] [Google Scholar]
- Luo YJ, Su YH. Opposing Nodal and BMP Signals Regulate Left-Right Asymmetry in the Sea Urchin Larva. Plos Biol. 2012;10 doi: 10.1371/journal.pbio.1001402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEdward L. Evolution of pelagic direct development in the starfish Pteraster tesselatus (Asteroidea: Velatida) Biological Journal of the Linnean Society. 1995;54:299–327. [Google Scholar]
- Menge B, Sanford E. Ecological role of sea stars from population to meta-ecosystems. Johns Hopkins Publisher; 2013. [Google Scholar]
- Mercier A, Hamel J-F. Reproduction in Asteroidea. In: Lawrence J, editor. Starfish: Biology and Ecology of the Asteroidea. The Johns Hopkins Press; Baltimore MD USA: 2013. pp. 37–50. [Google Scholar]
- Morris V, Selvakumaraswamy P, Whan R, Byrne M. Development of the five primary podia from the coeloms of a sea star larva: homology with the echinoid echinoderms and other deuterostomes. Proc R Soc B. 2009;276:1277–1284. doi: 10.1098/rspb.2008.1659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman H. An experimental analysis of asymmetry in the starfish, Patiria miniata. Biological Bulletin. 1925;49:111–138. [Google Scholar]
- O’Loughlin P, Waters J. A molecular and morphological revision of genera of Asterinidae (Echinodermata: Asteroidea) Memoirs of Museum Victoria. 2004;61:1–40. [Google Scholar]
- Oulhen N, Onorato T, Ramos I, Wessel G. Dysferlin is essential for endocytosis in the sea star oocyte. Developmental biology. 2014 doi: 10.1016/j.ydbio.2013.12.018. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pehrson J, Cohen L. The fate of the small micromeres in sea urchin development. Developmental biology. 1986;113:522–526. doi: 10.1016/0012-1606(86)90188-0. [DOI] [PubMed] [Google Scholar]
- Porter KM. The poor fossil record of the regular echinoid. Paleobiology. 1977;3:168–174. [Google Scholar]
- Ransick A, Cameron RA, Davidson EH. Postembryonic segregation of the germ line in sea urchins in relation to indirect development. Proceedings of the National Academy of Sciences of the United States of America. 1996;93:6759–6763. doi: 10.1073/pnas.93.13.6759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seervai RN, Wessel GM. Lessons for inductive germline determination. Mol Reprod Dev. 2013;80:590–609. doi: 10.1002/mrd.22151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith AB, Zamora S, Alvero J. The oldest echinoderm faunas from Gondwana show that echinoderm body plan diversification was rapid. Nature Communications. 2013;4:1835. doi: 10.1038/ncomms2391. [DOI] [PubMed] [Google Scholar]
- Tanaka S, Dan K. Study of the lineage and cell cycle of small micromeres in embryos of the sea urchin, Hemicentrotus pulcherrimus. Dev Growth Differ. 1991;32:145–156. doi: 10.1111/j.1440-169X.1990.00145.x. [DOI] [PubMed] [Google Scholar]
- Vandenberg L, Levin M. A unified model for left-right asymmetry? Comparison and synthesis of molecular models of embryonic laterality. Developmental biology. 2013;379:1–15. doi: 10.1016/j.ydbio.2013.03.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickery M, McClintock J. Regeneration in metazoan larvae. Nature. 1998;394:140. [Google Scholar]
- Vickery M, McClintock J. Effects of food concentration and availability on the incidence of cloning in planktotrophic larvae of the sea star Pisaster ochraceus. Biological Bulletin. 2000;199:298–304. doi: 10.2307/1543186. [DOI] [PubMed] [Google Scholar]
- Vickery M, Vickery M, McClintock J, Amsler C. Utilization of a novel deuterostome model for the study of regeneration genetics: molecular cloning of genes that are differentially expressed during early stages of larval sea star regeneration. Gene. 2001;262:73–80. doi: 10.1016/s0378-1119(00)00554-0. [DOI] [PubMed] [Google Scholar]
- Voronina E, Lopez M, Juliano CE, Gustafson E, Song JL, Extavour C, George S, Oliveri P, McClay D, Wessel G. Vasa protein expression is restricted to the small micromeres of the sea urchin, but is inducible in other lineages early in development. Developmental biology. 2008;314:276–286. doi: 10.1016/j.ydbio.2007.11.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessel GM, Reich AM, Klatsky PC. Use of sea stars to study basic reproductive processes. Systems biology in reproductive medicine. 2010;56:236–245. doi: 10.3109/19396361003674879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yajima M, Wessel GM. Small micromeres contribute to the germline in the sea urchin. Development. 2011;138:237–243. doi: 10.1242/dev.054940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yajima M, Wessel GM. Autonomy in specification of primordial germ cells and their passive translocation in the sea urchin. Development. 2012;139:3786–3794. doi: 10.1242/dev.082230. [DOI] [PMC free article] [PubMed] [Google Scholar]