Abstract
Oral busulfan is the historical backbone of the busulfan+cyclophosphamide regimen for autologous stem cell transplantation. However intravenous busulfan has more predictable pharmacokinetics and less toxicity than oral busulfan; we, therefore, retrospectively analyzed data from 952 patients with acute myeloid leukemia who received intravenous busulfan for autologous stem cell transplantation. Most patients were male (n=531, 56%), and the median age at transplantation was 50.5 years. Two-year overall survival, leukemia-free survival, and relapse incidence were 67±2%, 53±2%, and 40±2%, respectively. The non-relapse mortality rate at 2 years was 7±1%. Five patients died from veno-occlusive disease. Overall leukemia-free survival and relapse incidence at 2 years did not differ significantly between the 815 patients transplanted in first complete remission (52±2% and 40±2%, respectively) and the 137 patients transplanted in second complete remission (58±5% and 35±5%, respectively). Cytogenetic risk classification and age were significant prognostic factors: the 2-year leukemia-free survival was 63±4% in patients with good risk cytogenetics, 52±3% in those with intermediate risk cytogenetics, and 37 ± 10% in those with poor risk cytogenetics (P=0.01); patients ≤50 years old had better overall survival (77±2% versus 56±3%; P<0.001), leukemia-free survival (61±3% versus 45±3%; P<0.001), relapse incidence (35±2% versus 45±3%; P<0.005), and non-relapse mortality (4±1% versus 10±2%; P<0.001) than older patients. The combination of intravenous busulfan and high-dose melphalan was associated with the best overall survival (75±4%). Our results suggest that the use of intravenous busulfan simplifies the autograft procedure and confirm the usefulness of autologous stem cell transplantation in acute myeloid leukemia. As in allogeneic transplantation, veno-occlusive disease is an uncommon complication after an autograft using intravenous busulfan.
Introduction
Despite recent improvements in the prognosis and treatment of acute myeloid leukemia (AML), disease relapse continues to affect most patients who are not allografted.1 Autologous hematopoietic stem cell transplantation (ASCT) offers an alternative possibility of delivering high-dose myeloablative treatment in AML. Several historical randomized trials have reported significantly lower relapse incidences after ASCT than after conventional chemotherapy. Unfortunately ASCT is associated with a non-negligible non-relapse mortality linked to the use of total body irradiation or the combination of oral busulfan with cyclophosphamide, as well as slow kinetics of engraftment with bone marrow grafts. The use of peripheral blood stem cells since 1994 has resulted in more rapid engraftment kinetics and lower rates of non-relapse mortality.2
Busulfan is an alkylating agent that has been used as a chemotherapeutic agent since 1950. High-dose busulfan combined with cyclophosphamide is frequently used for chemotherapy dose intensification in patients undergoing ASCT for AML3,4 and other malignant and non-malignant diseases. Until recently busulfan was available only in an oral form and given four times daily. Although busulfan was shown to be effective when used in this regimen, the therapeutic potential of the oral drug is compromised by unpredictable exposure.5,6 A high area under the curve of busulfan plasma concentration versus time is associated with a high-risk of regimen-related toxicity and, in particular, sinusoidal obstructive syndrome/hepatic veno-occlusive disease.6–9 Conversely, low busulfan concentrations are associated with a higher risk of graft rejection and leukemia relapse.6,10–13
To reduce both intra- and inter-individual variability of busulfan pharmacokinetics, an intravenous formulation of busulfan has recently been developed. In adults, intravenous busulfan has shown predictable and consistent pharmacokinetic profiles with acceptable toxicity.14–16 In addition, it is easier to administer and is associated with lower rates of sinusoidal obstructive syndrome, partially due to the elimination of first-pass metabolism, in children and in allogeneic stem cell transplantation.
In adults with AML, historical series of ASCT from various institutions,17 retrospective surveys from the European Group for Blood and Marrow Transplantation (EBMT)18–22 and the Center for International Blood and Marrow Transplant Research,23 and randomized trials24,25 have revealed long-term leukemia-free survival rates of 45–55% in patients transplanted in first remission and 25–35% for those transplanted in second remission.26 However, in contrast to the considerable experience using oral busulfan in high-dose regimens, no studies have reported outcomes using intravenous busulfan-based conditioning regimens for ASCT. For that reason the EBMT Acute Leukemia Working Party evaluated outcomes of adult patients who underwent ASCT following a high-dose regimen containing intravenous busulfan.
Methods
The study design was approved by the Acute Leukemia Working Party, in accordance with the EBMT guidelines for retrospective studies.
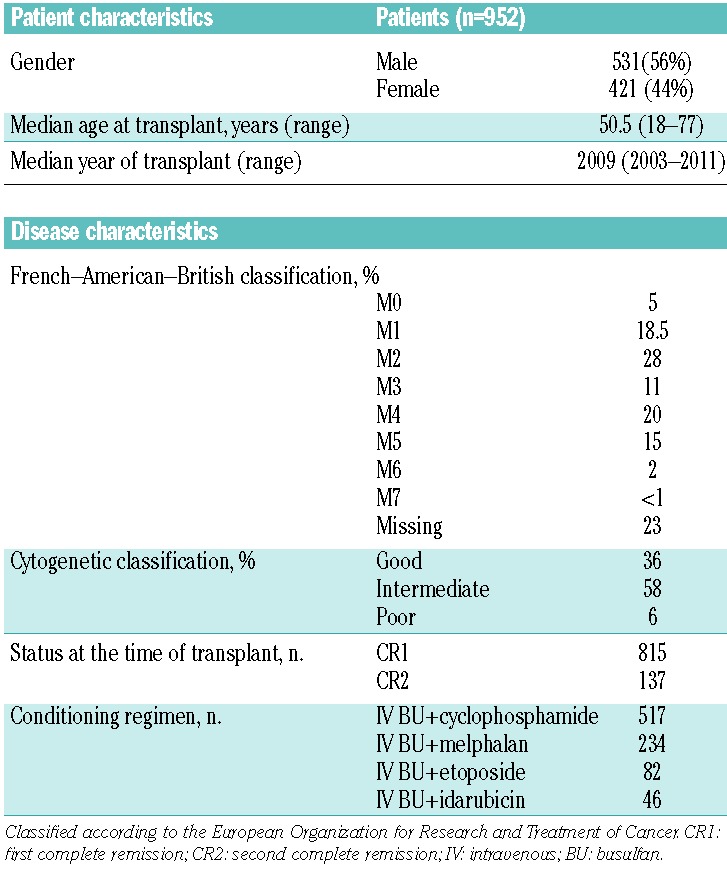
Data from 952 adult AML patients undergoing intravenous busulfan-based conditioning prior to ASCT from January 2003 to December 2011 were reported to the Acute Leukemia Working Party of the EBMT. The diagnosis of AML was based on morphological criteria according to the French–American–British classification. The median age of the patients was 50.5 years (range, 18–77), 56% of the patients were male, and the median transplant year was 2009 (2003–2011) (Table 1). Internal tandem duplication of the FLT3 gene was positive in 32 (22%) of the 142 patients assessed. The median follow-up was 16 months (range, 1–110 months). The conditioning regimen consisted of intravenous busulfan median total dose 12.8 mg/kg; (range, 6.4–16.3) combined with cyclophosphamide (total dose 120 mg/kg, n=517), melphalan (140 mg/m2, n=234), VP-16 (n=82), idarubicin (n=46), or other agents (n=73).
Table 1.
Patient and disease characteristics.
End-point definitions and statistical analysis
Four outcomes were evaluated in this series: (i) non-relapse mortality, defined as death without previous relapse; (ii) relapse incidence, defined on the basis of morphological evidence of leukemia in bone marrow or extramedullary organs; (iii) leukemia-free survival, defined as the time interval from the transplant to first event (either relapse or death in complete remission); and (iv) overall survival. Cumulative incidence curves were used for relapse incidence and non-relapse mortality in a competing risks setting,27 since death and relapse are competing events. The Gray test was used for univariate comparisons.28 Probabilities of overall survival and leukemia-free survival were calculated using the Kaplan–Meier estimate,29 and the log-rank test was used for univariate comparisons. Relationships between outcomes and patient, disease, and graft characteristics were evaluated in multivariate analyses using the Cox proportional hazards model.30 All tests were two-sided. The type I error rate was fixed at 0.05 for the determination of factors associated with time-to-event outcomes. Statistical analyses were performed with SPSS 19 (SPSS Inc., Chicago, IL, USA) and R 2.13.2 (R Development Core Team, Vienna, Austria) software packages.
Results
Nine hundred and twenty-three patients engrafted with a median time to neutrophil recovery (>500/mm3) of 12 days (range, 2–110); 12 patients did not engraft, and three additional patients had a late graft failure. A total of 256 patients died: 161 from relapse and disease progression, 45 from infection, and 5 from hepatic veno-occlusive disease. At 2 years, the overall survival rate was 67±2%, the leukemia-free survival rate was 53±2%, the relapse incidence was 40± 2%, and the non-relapse mortality rate was 7±1%. Of the five patients who died from sinusoidal obstructive syndrome, four were transplanted in first complete remission and one in second complete remission; the doses of intravenous busulfan used in these patients were 16 mg/kg (n=1), 12.8 mg/kg (n=3), and 12 mg/kg (n=1).
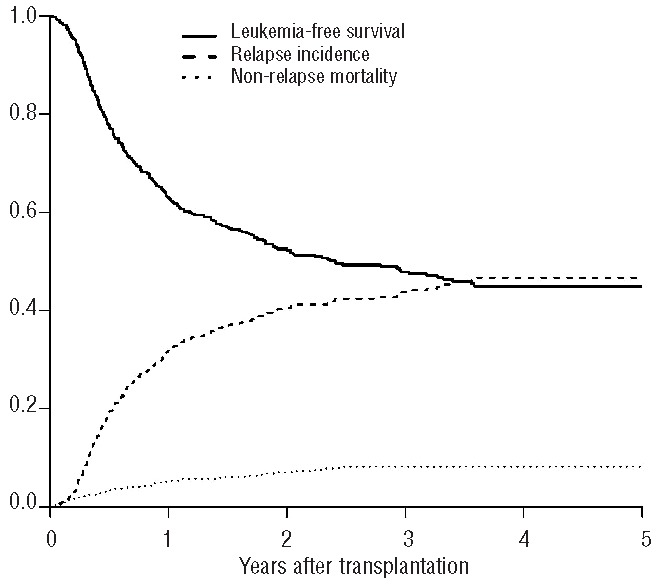
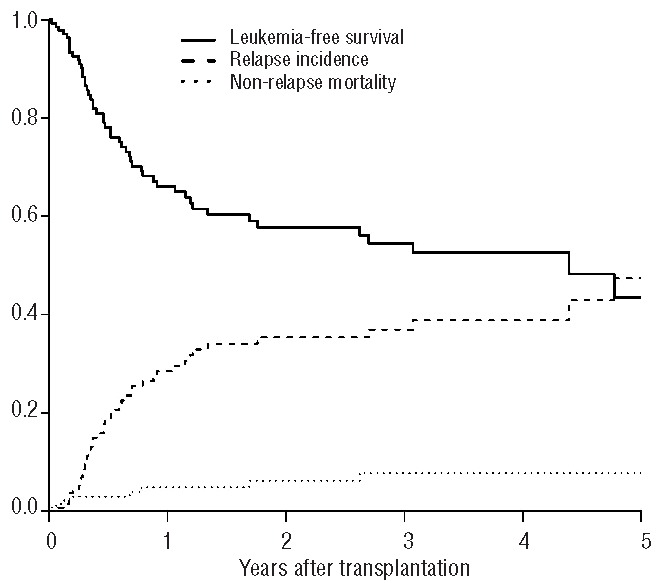
The univariate analyses showed that there were no differences in outcome for patients transplanted in first complete remission or second complete remission (Table 2). For patients transplanted in first complete remission, the overall survival rate was 67±2%, the leukemia-free survival rate was 52±2%, and the relapse incidence was 40±2% (Figure 1). For patients transplanted in second complete remission, the overall survival rate was 69±4%, the leukemia-free survival rate was 58±5%, and the relapse incidence was 35±5% (Figure 2). The non-relapse mortality rate did not differ between the two groups of patients (first complete remission 7±1% versus second complete remission 6±2%). As expected, cytogenetic risk classification was predictive of outcome, with significantly better results for the good risk group compared with intermediate risk and poor risk groups (overall survival: 75±2% versus 69±3% versus 46±10%, P=0.003; leukemia-free survival: 63±4% versus 52±3% versus 37±10%, P=0.01; relapse incidence 31±4% versus 43±3% versus 55±11%, P=0.02). Younger patients (≤50 years old) had better outcomes than older patients (overall survival: 77±2% versus 56±3%, P<0.001; leukemia-free survival: 61±3% versus 45±3%, P<0.001; relapse incidence: 35±2% versus 45±3%, P=0.005) (Figure 3). The non-relapse mortality was 4±1% in patients ≤50 years, and 10±2% in patients >50 years (P=0.0002).
Table 2.
Univariate analysis of prognostic factors.
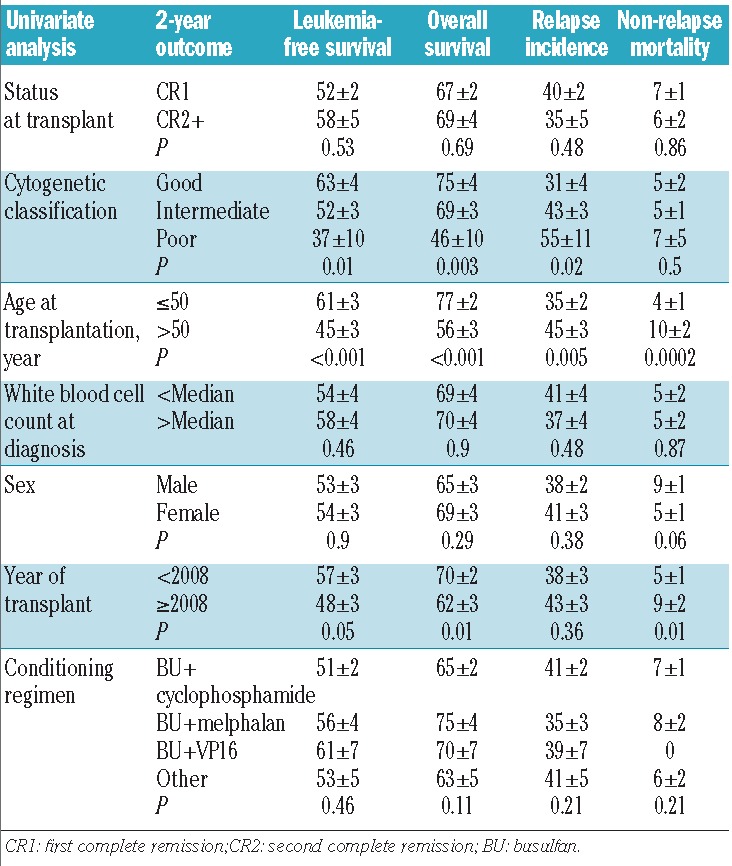
Figure 1.
Outcomes of patients with AML autografted in first complete remission using a pretransplant conditioning regimen containing intravenous busulfan.
Figure 2.
Outcomes of patients with AML autografted in second complete remission using a pretransplant conditioning regimen containing intravenous busulfan.
Figure 3.
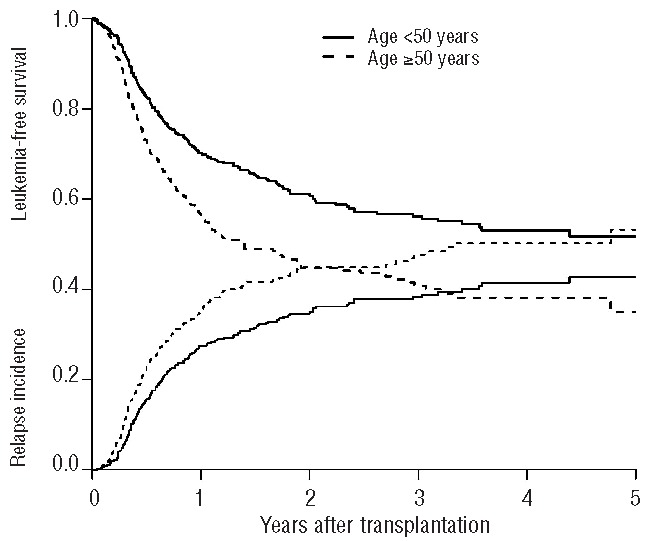
Leukemia-free survival and relapse incidence according to the patients’ age (<50 years, ≥50 years).
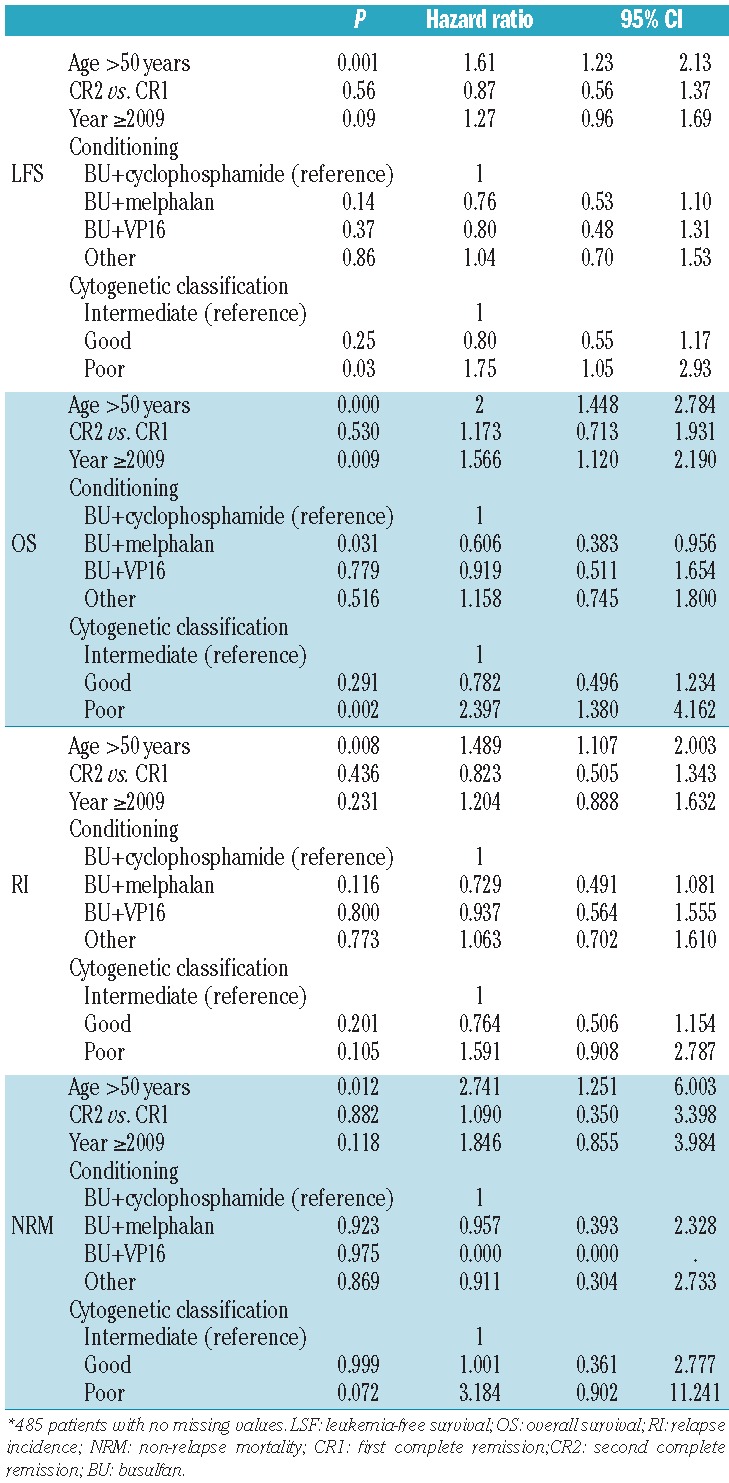
Results of multivariate analysis (Table 3) showed that age was a significant prognostic factor for overall survival, leukemia-free survival, relapse incidence, and non-relapse mortality, and cytogenetic classification was a significant prognostic factor for overall survival and leukemia-free survival. The combination of intravenous busulfan and high-dose melphalan was associated with a better overall survival (P=0.03; hazard ratio: 0.6; 95% confidence interval: 0.38–0.95).
Table 3.
Multivariate analyses*.
Discussion
ASCT has been widely used for consolidation chemotherapy in patients with AML in first or second complete remission in the past decades. Indeed, the EBMT registry presently contains information on more than 17,000 autografts for AML. Although ASCT for AML remains a therapeutic option, it has become less popular for two major reasons. The first reason is the high relapse incidence post-ASCT and the risk of late relapse (11% at 5 years, 16% at 10 years) in patients still in complete remission 2 years post-ASCT, suggesting a possible role for some maintenance therapy.31 A second reason is the recent development of allogeneic stem cell transplantation with reduced-intensity conditioning, which has made transplantation feasible in older patients (≤70 years of age) and enabled the use of alternative donors e.g., HLA-matched unrelated donors, cord blood, and even haplo-mismatched family donors, allowing the possibility of an allograft for almost all patients. Allogeneic stem cell transplantation is associated with a lower relapse incidence from the graft-versus-leukemia effect but unfortunately is also associated with higher rates of non-relapse mortality, graft-versus-host disease, and infections; surviving recipients of allogeneic grafts do, therefore, tend to have a poorer quality of life compared with patients who undergo ASCT.32
Several randomized studies24,25,33–36 comparing allogeneic stem cell transplantation using HLA-identical siblings after a myeloablative conditioning regimen with ASCT and chemotherapy have reported better outcomes after allogeneic transplantation, but they have also consistently shown a decreased relapse incidence after ASCT compared with conventional chemotherapy. However, to date no randomized study has demonstrated the superiority of allogeneic transplantation using an alternative donor (unrelated or cord blood) over ASCT. A retrospective EBMT study of elderly patients with de novo AML found that outcomes after allogeneic peripheral blood stem cell transplantation from HLA-identical sibling donors with reduced-intensity conditioning were similar to outcomes after autologous peripheral blood stem cell transplantation.37
A recent retrospective study from the Center for International Blood and Marrow Transplant Research concluded that in the absence of a matched sibling donor, ASCT may provide an acceptable alternative post-remission therapy for patients with AML in first complete remission.23 Furthermore results of ASCT in AML may be improved. Several recent studies, including two retrospective surveys from the EBMT, reported similar outcomes after ASCT and allogeneic stem cell transplantation in patients with AML in the good risk classification who carried an inversion on chromosome 16, the t(8 ;21) translocation, or the NPM1 mutation.20,21 More recently Schlenk et al.38 evaluated patients included in prospective randomized trials with prognostically favorable AML and double CEBPA mutations. They found that allogeneic or autologous hematopoietic stem cell transplantation produced identical outcomes, with leukemia-free survival and overall survival rates >70% at 4 years, which were significantly better than outcomes after conventional chemotherapy.
In addition, the quality of the autograft is of considerable importance. Clinical observations from patients with acute promyelocytic leukemia (AML3) in second complete remission39 or acute lymphocytic leukemia expressing the BCR-ABL transcript and receiving tyrosine kinase inhibitors40 have shown that patients with no detectable minimal residual disease may benefit from dose intensification and autografting. Regarding the introduction of maintenance therapy, trials in progress41 are looking for a possible benefit from the use of hypomethylating agents in allogeneic transplantation. This approach may also be useful in ASCT to reduce the incidence of late relapse. Overall, these considerations may support the view that patients receiving only chemotherapy nowadays in fact should receive ASCT whenever possible, and ASCT would be a more appropriate control than chemotherapy alone for allogeneic transplantation with any comparison taking into account the quality of life.
The present study is the first retrospective study to include a large series of patients with AML receiving an intravenous busulfan-based conditioning regimen prior to ASCT. The absorption and bioavailability of oral busulfan are erratic and unpredictable; therefore, monitoring busulfan levels and making dose adjustments cannot be easily achieved with the oral formulation.8,42–44 In contrast, the initial experience with intravenous busulfan showed that pharmacokinetic values are more predictable, with 85% of patients achieving and maintaining the targeted therapeutic window (area under the curve: 900–1500 mM/min). This allows a tight control of plasma levels and less need for plasma level testing and dose adjustment.14 Lee et al.45 evaluated 253 patients with malignant disorders (49% with breast cancer) who underwent ASCT after receiving a conditioning regimen of 12 mg/kg oral busulfan, 100 mg/m2 melphalan, and 500 mg/m2 thiotepa and found that 70 (28%) experienced veno-occlusive disease, which was moderate in 31 (12%) patients and severe in 11 (4%). Our observation of fatal sinusoidal obstructive syndrome/veno-occlusive disease in only 5% of patients receiving intravenous busulfan is in sharp contrast with these historical data. Similar low incidences of sinusoidal obstructive syndrome/veno-occlusive disease have been claimed when using pharmacokinetics and dose adjustment for oral busulfan. However, with this approach, there is still considerable intrapatient variability with a reported coefficient of variation of 36%;46 furthermore, this technology is not available everywhere, so that the benefit in terms of cost/effectiveness concerns only selected centers. The observation that patients autografted in second complete remission had outcomes similar to those autografted in first complete remission, which has never been reported in numerous series and trials, further attests to the potential benefits of intravenous busulfan. Recent studies evaluated the toxicity and outcomes of intravenous busulfan-based conditioning in patients with multiple myeloma,47 non-Hodgkin lymphoma, neuroblastoma, and Ewing sarcoma.48 Taken together, the results demonstrated that intravenous busulfan has high efficacy with very low toxicity; the rate of mild-to-moderate sinusoidal obstructive syndrome was <5% and no deaths without prior relapse (non-relapse mortality) were observed.
We recently reported for the Acute Leukemia Working Party of the EBMT on the comparison of intravenous busulfan plus cyclophosphamide versus total body irradiation plus cyclophosphamide in allotransplanted patients.49 Patients who received intravenous busulfan plus cyclophosphamide had lower rates of acute and chronic graft-versus-host disease and a trend toward lower non-relapse mortality. Leukemia-free survival did not differ significantly between the two groups of patients. In the present retrospective study, patients received intravenous busulfan at the recommended dosage of 0.8 mg/kg four times a day for 4 days before ASCT. New modalities of administered intravenous busulfan are being tested with only one perfusion per day.50 Likewise, high-dose melphalan is now given intravenously over 90 to 120 minutes. In the present study patients who received intravenous busulfan plus melphalan had a better overall survival rate than other patients but did not differ in terms of relapse incidence or leukemia-free survival rate. The combination of intravenous busulfan and high-dose melphalan may be one of the simplest conditioning regimens before autografting.
Along the lines described above, there are presently three ongoing phase 2 studies testing the role of ASCT, two in good and intermediate risk AML within the Spanish CETLAM group with a specific interest in evaluation of minimal residual disease and in vivo purging by gemtuzumab ozogamicin and one in good risk patients only, using intravenous busulfan four times a day and etoposide as a pretransplant regimen in South Korea. In addition, a randomized phase 3 study comparing ASCT with intravenous busulfan included in the pretransplant regimen and haplo-mismatched transplants in AML is currently underway in China, where these two therapeutic strategies are both used, since most families have only one child.
Based on our results we suggest that intravenous busulfan may be an important step forward to improve results of ASCT in patients with AML. Prospective studies comparing intravenous busulfan-based conditioning for ASCT with allogeneic stem cell transplantation using matched unrelated or matched-related donors should be performed.
Acknowledgments
Cardarelli Hospital, Napoli, Italy; Hospital Reina Sofia, Córdoba, Spain; Hopital Saint Antoine-Hotel Dieu, Paris, France; Azienda Ospedaliera S. Giovanni, Torino, Italy; CHRU Limoges, France; Hospital dos Capuchos, Lisboa, Portugal; Ospedali Riuniti di Bergamo, Italy; CHU Nantes, France; University Hospital, Basel, Switzerland; Ospedale Vito Fazzi, Lecce, Italy; Ospedale A. Businco, Cagliari, Italy; Azienda Ospedali Riuniti di Ancona, Ancona-Torrete, Italy; King Faisal Specialist Hospital & Research Centre, Riyadh, Saudi Arabia; University Medical Center St. Radboud, Nijmegen, The Netherlands, The; Gazi Universitesi Tip Fakültesi, Ankara, Turkey; Hospital Gregorio Marañón, Madrid, Spain; Rome Transplant Network, Italy; Hospital Clínico, Salamanca, Spain; Hospital Universitario Son Espases, Palma De Mallorca, Spain; CHRU d’Angers, France; Hospital Covadonga, Oviedo, Spain; Hopital Bretonneau, Tours, France; H SS. Antonio e Biagio, Alessandria, Italy; CHU de Dijon - Hopital Le Bocage, Dijon, France; University Medical Center, Ljubljana, Slovenia; Ospedale di Careggi, Firenze, Italy; Mazzoni Hospital, Ascoli Piceno, Italy; Hospital son Llatzer, Palma De Mallorca, Spain; University of Freiburg, Germany; Constantiaberg Medi-Clinic, Cape Town, South Africa; Tel-Aviv University, Tel-Hashomer, Israel; Service d’Onco Hematologie, Strasbourg, France; Centre For Clinical Haematology, Birmingham, United Kingdom; Spedali Civili - Brescia, Italy; Hadassah University Hospital, Jerusalem, Israel; Medizinische Klinik und Poliklinik, Ulm, Germany; Centre Henri Becquerel, Rouen, France; Clinica Puerta de Hierro, Madrid, Spain; Hospital Universitario Puerta del Mar, Cádiz, Spain; Klinikum Bremen-Mitte, Bremen, Germany; Gaziantep University Medical School, Gaziantep, Turkey; GATA BMT Center, Ankara, Turkey; Kaplan Hospital, Rehovot, Israel; Cliniques Universitaires St. Luc, Brussels, Belgium; Robert-Bosch-Krankenhaus, Stuttgart, Germany.
Footnotes
Authorship and Disclosures
Information on authorship, contributions, and financial & other disclosures was provided by the authors and is available with the online version of this article at www.haematologica.org.
References
- 1.Buchner T, Schlenk RF, Schaich M, Dohner K, Krahl R, Krauter J, et al. Acute myeloid leukemia (AML): different treatment strategies versus a common standard arm–combined prospective analysis by the German AML Intergroup. J Clin Oncol. 2012;30(29):3604–10 [DOI] [PubMed] [Google Scholar]
- 2.Gorin NC. Autologous stem cell transplantation in acute myelocytic leukemia. Blood. 1998;92(4):1073–90 [PubMed] [Google Scholar]
- 3.Santos GW. Busulfan (Bu) and cyclophosphamide (Cy) for marrow transplantation. Bone Marrow Transplant. 1989;4(Suppl 1):236–9 [PubMed] [Google Scholar]
- 4.Santos GW. Allogeneic and autologous bone marrow transplantation in AML. Leukemia. 1992;6(Suppl 2):102–3 [PubMed] [Google Scholar]
- 5.Bleyzac N, Souillet G, Magron P, Janoly A, Martin P, Bertrand Y, et al. Improved clinical outcome of paediatric bone marrow recipients using a test dose and Bayesian pharmacokinetic individualization of busulfan dosage regimens. Bone Marrow Transplant. 2001;28(8):743–51 [DOI] [PubMed] [Google Scholar]
- 6.Slattery JT, Sanders JE, Buckner CD, Schaffer RL, Lambert KW, Langer FP, et al. Graft-rejection and toxicity following bone marrow transplantation in relation to busulfan pharmacokinetics. Bone Marrow Transplant. 1995;16(1):31–42 [PubMed] [Google Scholar]
- 7.Dix SP, Wingard JR, Mullins RE, Jerkunica I, Davidson TG, Gilmore CE, et al. Association of busulfan area under the curve with veno-occlusive disease following BMT. Bone Marrow Transplant. 1996;17(2):225–30 [PubMed] [Google Scholar]
- 8.Grochow LB, Jones RJ, Brundrett RB, Braine HG, Chen TL, Saral R, et al. Pharmacokinetics of busulfan: correlation with veno-occlusive disease in patients undergoing bone marrow transplantation. Cancer Chemother Pharmacol. 1989;25(1):55–61 [DOI] [PubMed] [Google Scholar]
- 9.Ljungman P, Hassan M, Bekassy AN, Ringden O, Oberg G. High busulfan concentrations are associated with increased transplant-related mortality in allogeneic bone marrow transplant patients. Bone Marrow Transplant. 1997;20(11):909–13 [DOI] [PubMed] [Google Scholar]
- 10.Bolinger AM, Zangwill AB, Slattery JT, Glidden D, DeSantes K, Heyn L, et al. An evaluation of engraftment, toxicity and busulfan concentration in children receiving bone marrow transplantation for leukemia or genetic disease. Bone Marrow Transplant. 2000;25(9):925–30 [DOI] [PubMed] [Google Scholar]
- 11.McCune JS, Gooley T, Gibbs JP, Sanders JE, Petersdorf EW, Appelbaum FR, et al. Busulfan concentration and graft rejection in pediatric patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant. 2002;30(3):167–73 [DOI] [PubMed] [Google Scholar]
- 12.Slattery JT, Clift RA, Buckner CD, Radich J, Storer B, Bensinger WI, et al. Marrow transplantation for chronic myeloid leukemia: the influence of plasma busulfan levels on the outcome of transplantation. Blood. 1997; 89(8):3055–60 [PubMed] [Google Scholar]
- 13.Gibbs JP, Murray G, Risler L, Chien JY, Dev R, Slattery JT. Age-dependent tetrahydrothiophenium ion formation in young children and adults receiving high-dose busulfan. Cancer Res. 1997;57(24):5509–16 [PubMed] [Google Scholar]
- 14.Andersson BS, Gajewski J, Donato M, Giralt S, Gian V, Wingard J, et al. Allogeneic stem cell transplantation (BMT) for AML and MDS following i.v. busulfan and cyclophosphamide (i.v. BuCy). Bone Marrow Transplant. 2000;(Suppl 2):S35–8 [DOI] [PubMed] [Google Scholar]
- 15.Vassal G, Michel G, Esperou H, Gentet JC, Valteau-Couanet D, Doz F, et al. Prospective validation of a novel IV busulfan fixed dosing for paediatric patients to improve therapeutic AUC targeting without drug monitoring. Cancer Chemother Pharmacol. 2008;61(1):113–23 [DOI] [PubMed] [Google Scholar]
- 16.Andersson BS, Kashyap A, Gian V, Wingard JR, Fernandez H, Cagnoni PJ, et al. Conditioning therapy with intravenous busulfan and cyclophosphamide (IV BuCy2) for hematologic malignancies prior to allogeneic stem cell transplantation: a phase II study. Biol Blood Marrow Transplant. 2002;8(3):145–54 [DOI] [PubMed] [Google Scholar]
- 17.Gorin NC, Labopin M, Laporte JP, Douay L, Lopez M, Lesage S, et al. Importance of marrow dose on posttransplant outcome in acute leukemia: models derived from patients autografted with mafosfamide-purged marrow at a single institution. Exp Hematol. 1999;27(12):1822–30 [DOI] [PubMed] [Google Scholar]
- 18.Gorin NC, Aegerter P, Auvert B, Meloni G, Goldstone AH, Burnett A, et al. Autologous bone marrow transplantation for acute myelocytic leukemia in first remission: a European survey of the role of marrow purging. Blood. 1990;75(8):1606–14 [PubMed] [Google Scholar]
- 19.Gorin NC, Labopin M, Blaise D, Reiffers J, Meloni G, Michallet M, et al. Higher incidence of relapse with peripheral blood rather than marrow as a source of stem cells in adults with acute myelocytic leukemia autografted during the first remission. J Clin Oncol. 2009;27(24):3987–93 [DOI] [PubMed] [Google Scholar]
- 20.Gorin NC, Labopin M, Frassoni F, Milpied N, Attal M, Blaise D, et al. Identical outcome after autologous or allogeneic genoidentical hematopoietic stem-cell transplantation in first remission of acute myelocytic leukemia carrying inversion 16 or t(8;21): a retrospective study from the European Cooperative Group for Blood and Marrow Transplantation. J Clin Oncol. 2008;26(19):3183–8 [DOI] [PubMed] [Google Scholar]
- 21.Gorin NC, Labopin M, Meloni G, Pigneux A, Esteve J, Mohamad M. Impact of FLT3 ITD/NPM1 mutation status in adult patients with acute myelocytic leukemia autografted in first remission. Haematologica. 2013;(2): e12–4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gorin NC, Labopin M, Reiffers J, Milpied N, Blaise D, Witz F, et al. Higher incidence of relapse in patients with acute myelocytic leukemia infused with higher doses of CD34+ cells from leukapheresis products autografted during the first remission. Blood. 2010;116(17):3157–62 [DOI] [PubMed] [Google Scholar]
- 23.Keating A, DaSilva G, Perez WS, Gupta V, Cutler CS, Ballen KK, et al. Autologous blood cell transplantation versus HLA-identical sibling transplantation for acute myeloid leukemia in first complete remission: a registry study from the Center for International Blood and Marrow Transplantation Research. Haematologica. 2013;98(2):185–92 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang J, Ouyang J, Zhou R, Chen B, Yang Y. Autologous hematopoietic stem cell transplantation for acute myeloid leukemia in first complete remission: a meta-analysis of randomized trials. Acta Haematol. 2010;124(2):61–71 [DOI] [PubMed] [Google Scholar]
- 25.Levi I, Grotto I, Yerushalmi R, Ben-Bassat I, Shpilberg O. Meta-analysis of autologous bone marrow transplantation versus chemotherapy in adult patients with acute myeloid leukemia in first remission. Leuk Res. 2004;28(6):605–12 [DOI] [PubMed] [Google Scholar]
- 26.Meloni G, Vignetti M, Avvisati G, Capria S, Micozzi A, Giona F, et al. BAVC regimen and autograft for acute myelogenous leukemia in second complete remission. Bone Marrow Transplant. 1996;18(4):693–8 [PubMed] [Google Scholar]
- 27.Fine JP, Gray RJ. A proportional hazards model for subdistribution of a competing risk. J Am Stat Assoc. 1999;94(446):496–509 [Google Scholar]
- 28.Gooley TA, Leisenring W, Crowley J, Storer BE. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med. 1999;18(6):695–706 [DOI] [PubMed] [Google Scholar]
- 29.Kaplan EL, Meier P. Non parametric estimation from incomplete observations. J Am Stat Assoc. 1958;53:457–81 [Google Scholar]
- 30.Cox DR. Regression models and life tables. J R Stat Soc. 1972;34:187–202 [Google Scholar]
- 31.Czerw T, Labopin M, Gorin NC, Giebel S, Blaise D, Meloni G, et al. Long-term follow-up of autologous hematopoietic stem cell transplantation (AHSCT) for acute myeloid leukemia (AML): a survey of 3567 patients in CR at two years post transplantation, from the Acute Leukemia Working Party of the EBMT. Blood. 2012;120(21):abstr 3112 [Google Scholar]
- 32.Sun CL, Francisco L, Kawashima T, Leisenring W, Robison LL, Baker KS, et al. Prevalence and predictors of chronic health conditions after hematopoietic cell transplantation: a report from the Bone Marrow Transplant Survivor Study. Blood. 2010;116(17):3129–39; quiz 3377 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Burnett AK, Goldstone AH, Stevens RM, Hann IM, Rees JK, Gray RG, et al. Randomised comparison of autologous bone marrow transplantation to intensive chemotherapy for acute myeloid leukaemia in first remission: results of MRC-AML 10 trial. Lancet. 1998; 351(9104):700–8 [DOI] [PubMed] [Google Scholar]
- 34.Cassileth PA, Harrington DP, Appelbaum FR, Lazarus HM, Rowe JM, Paietta E, et al. Chemotherapy compared with autologous or allogeneic bone marrow transplantation in the management of acute myeloid leukemia in first remission. N Engl J Med. 1998;339(23):1649–56 [DOI] [PubMed] [Google Scholar]
- 35.Harousseau JL, Cahn JY, Pignon B, Witz F, Milpied N, Delain M, et al. Comparison of autologous bone marrow transplantation and intensive chemotherapy as postremission therapy in adult acute myeloid leukemia. The Groupe Ouest Est Leucemies Aigues Myeloblastiques (GOELAM). Blood. 1997;90(8):2978–86 [PubMed] [Google Scholar]
- 36.Zittoun RA, Mandelli F, Willemze R, de Witte T, Labar B, Resegotti L, et al. Autologous or allogeneic bone marrow transplantation compared with intensive chemotherapy in acute myelogenous leukemia. European Organization for Research and Treatment of Cancer (EORTC) and the Gruppo Italiano Malattie Ematologiche Maligne dell’Adulto (GIMEMA) Leukemia Cooperative Groups. N Engl J Med. 1995;332(4):217–23 [DOI] [PubMed] [Google Scholar]
- 37.Herr AL, Labopin M, Blaise D, Milpied N, Potter M, Michallet M, et al. HLA-identical sibling allogeneic peripheral blood stem cell transplantation with reduced intensity conditioning compared to autologous peripheral blood stem cell transplantation for elderly patients with de novo acute myeloid leukemia. Leukemia. 2007;21(1):129–35 [DOI] [PubMed] [Google Scholar]
- 38.Schlenk RF, Taskesen E, van Norden Y, Krauter J, Ganser A, Bullinger L, et al. The value of allogeneic and autologous hematopoietic stem cell transplantation in prognostically favorable acute myeloid leukemia with double mutant CEBPA. Blood. 2013;122(9):1576–82 [DOI] [PubMed] [Google Scholar]
- 39.Holter-Chakrabarty JL, Rubinger M, Grigg A, Szer J, Selby G, Rowe JM, et al. Autologous hematopoietic cell transplantation is superior to allogeneic transplantation for acute promyelocytic leukemia in second complete remission. Blood. 2013;122(21):418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Giebel S, Labopin M, Gorin NC, Caillot D, Leguay T, Schaap N, et al. Improving results of autologous stem cell transplantation for Philadelphia-positive acute lymphoblastic leukaemia in the era of tyrosine kinase inhibitors: a report from the Acute Leukaemia Working Party of the European Group for Blood and Marrow Transplantation. Eur J Cancer. 2014;50(2):411–7 [DOI] [PubMed] [Google Scholar]
- 41.De Lima M, Giralt S, Thall PF, de Padua Silva L, Jones RB, Komanduri K, et al. Maintenance therapy with low-dose azacitidine after allogeneic hematopoietic stem cell transplantation for recurrent acute myelogenous leukemia or myelodysplastic syndrome: a dose and schedule finding study. Cancer. 2010;116(23):5420–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bolinger AM, Zangwill AB, Slattery JT, Risler LJ, Sultan DH, Glidden DV, et al. Target dose adjustment of busulfan in pediatric patients undergoing bone marrow transplantation. Bone Marrow Transplant. 2001;28(11):1013–8 [DOI] [PubMed] [Google Scholar]
- 43.Deeg HJ, Storer B, Slattery JT, Anasetti C, Doney KC, Hansen JA, et al. Conditioning with targeted busulfan and cyclophosphamide for hemopoietic stem cell transplantation from related and unrelated donors in patients with myelodysplastic syndrome. Blood. 2002;100(4):1201–7 [DOI] [PubMed] [Google Scholar]
- 44.Hassan M. The role of busulfan in bone marrow transplantation. Med Oncol. 1999;16(3):166–76 [DOI] [PubMed] [Google Scholar]
- 45.Lee JL, Gooley T, Bensinger W, Schiffman K, McDonald GB. Veno-occlusive disease of the liver after busulfan,melphalan and thiotepa conditioning therapy: incidence, risk factors and outcome. Biol Blood Marrow Transplant. 1999;5(5):306–15 [DOI] [PubMed] [Google Scholar]
- 46.Lindley C, Shea T, McCune J, Shord S, Decker J, Harvey D, et al. Intraindividual variability in busulfan pharmacokinetics in patients undergoing a bone marrow transplant: assessment of a test dose and first dose strategy. Anticancer Drugs. 2004;15(5):453–9 [DOI] [PubMed] [Google Scholar]
- 47.Blanes M, de la Rubia J, Lahuerta JJ, Gonzalez JD, Ribas P, Solano C, et al. Single daily dose of intravenous busulfan and melphalan as a conditioning regimen for patients with multiple myeloma undergoing autologous stem cell transplantation: a phase II trial. Leuk Lymphoma. 2009;50(2):216–22 [DOI] [PubMed] [Google Scholar]
- 48.Hovi L, Saxen H, Saarinen-Pihkala UM, Vettenranta K, Meri T, Richardson M. Prevention and monitoring of invasive fungal infections in pediatric patients with cancer and hematologic disorders. Pediatr Blood Cancer. 2007;48(1):28–34 [DOI] [PubMed] [Google Scholar]
- 49.Nagler A, Rocha V, Labopin M, Unal A, Ben Othman T, Campos A, et al. Allogeneic hematopoietic stem-cell transplantation for acute myeloid leukemia in remission: comparison of intravenous busulfan plus cyclophosphamide (Cy) versus total-body irradiation plus Cy as conditioning regimen–a report from the acute leukemia working party of the European group for blood and marrow transplantation. J Clin Oncol. 2013;31(28):3549–56 [DOI] [PubMed] [Google Scholar]
- 50.Almog S, Kurnik D, Shimoni A, Loebstein R, Hassoun E, Gopher A, et al. Linearity and stability of intravenous busulfan pharmacokinetics and the role of glutathione in busulfan elimination. Biol Blood Marrow Transplant. 2011;17(1):117–23 [DOI] [PubMed] [Google Scholar]


