TABLE VI.
Randomized trials of patients with multiple myeloma: adverse events
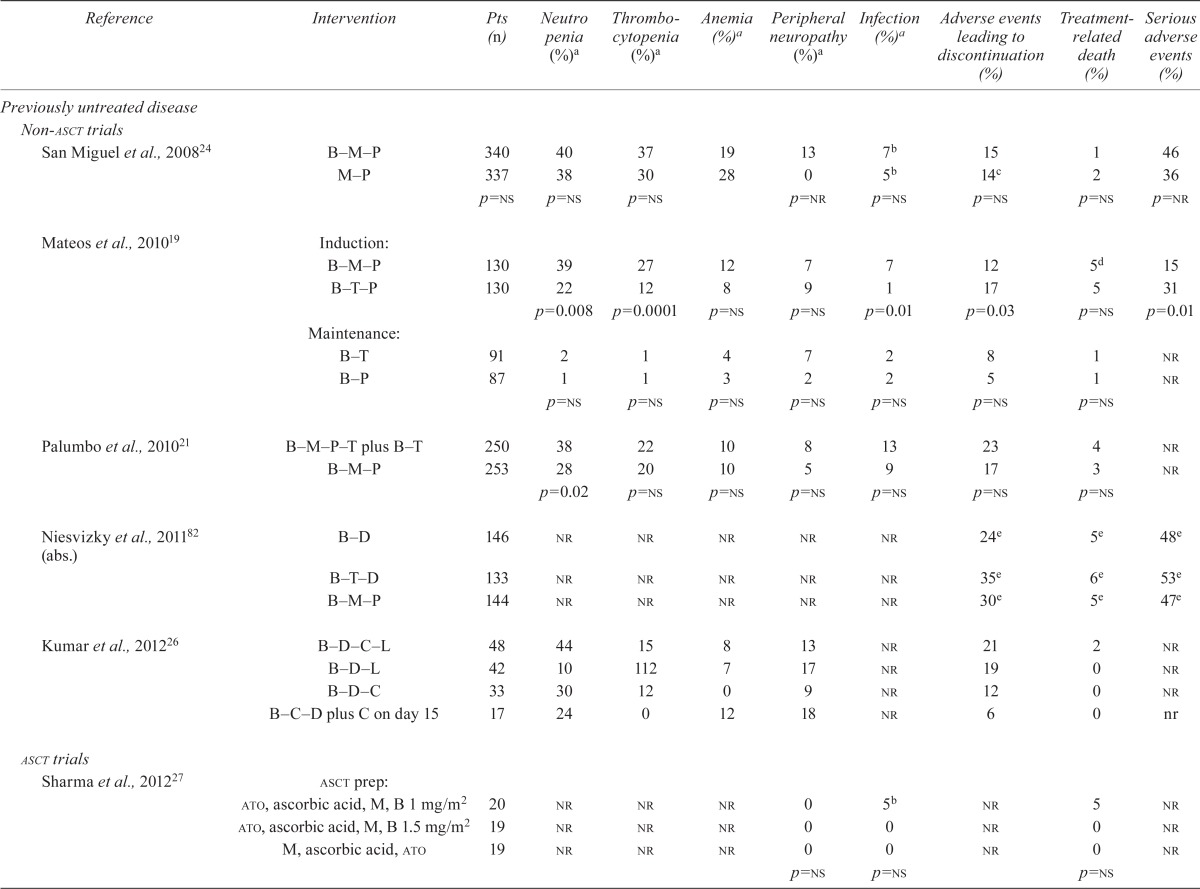
| Reference | Intervention | Pts (n) | Neutro penia (%)a | Thrombocytopenia (%)a | Anemia (%)a | Peripheral neuropathy (%)a | Infection (%)a | Adverse events leading to discontinuation (%) | Treatment-related death (%) | Serious adverse events (%) |
|---|---|---|---|---|---|---|---|---|---|---|
| Previously untreated disease | ||||||||||
| Non-asct trials | ||||||||||
| San Miguel et al., 200824 | B–M–P | 340 | 40 | 37 | 19 | 13 | 7b | 15 | 1 | 46 |
| M–P | 337 | 38 | 30 | 28 | 0 | 5b | 14c | 2 | 36 | |
| p=ns | p=ns | p=ns | p=nr | p=ns | p=ns | p=ns | p=nr | |||
| Mateos et al., 201019 | Induction: B–M–P | 130 | 39 | 27 | 12 | 7 | 7 | 12 | 5d | 15 |
| B–T–P | 130 | 22 | 12 | 8 | 9 | 1 | 17 | 5 | 31 | |
| p=0.008 | p=0.0001 | p=ns | p=ns | p=0.01 | p=0.03 | p=ns | p=0.01 | |||
| Maintenance: B–T | 91 | 2 | 1 | 4 | 7 | 2 | 8 | 1 | nr | |
| B–P | 87 | 1 | 1 | 3 | 2 | 2 | 5 | 1 | nr | |
| p=ns | p=ns | p=ns | p=ns | p=ns | p=ns | p=ns | ||||
| Palumbo et al., 201021 | B–M–P–T plus B–T | 250 | 38 | 22 | 10 | 8 | 13 | 23 | 4 | nr |
| B–M–P | 253 | 28 | 20 | 10 | 5 | 9 | 17 | 3 | nr | |
| p=0.02 | p=ns | p=ns | p=ns | p=ns | p=ns | p=ns | ||||
| Niesvizky et al., 201182 (abs.) | B–D | 146 | nr | nr | nr | nr | nr | 24e | 5e | 48e |
| B–T–D | 133 | nr | nr | nr | nr | nr | 35e | 6e | 53e | |
| B–M–P | 144 | nr | nr | nr | nr | nr | 30e | 5e | 47e | |
| Kumar et al., 201226 | B–D–C–L | 48 | 44 | 15 | 8 | 13 | nr | 21 | 2 | nr |
| B–D–L | 42 | 10 | 112 | 7 | 17 | nr | 19 | 0 | nr | |
| B–D–C | 33 | 30 | 12 | 0 | 9 | nr | 12 | 0 | nr | |
| B–C–D plus C on day 15 | 17 | 24 | 0 | 12 | 18 | nr | 6 | 0 | nr | |
| asct trials | ||||||||||
| Sharma et al., 201227 | asct prep:ato, ascorbic acid, M, B 1 mg/m2 | 20 | nr | nr | nr | 0 | 5b | nr | 5 | nr |
| ato, ascorbic acid, M, B 1.5 mg/m2 | 19 | nr | nr | nr | 0 | 0 | nr | 0 | nr | |
| M, ascorbic acid, ato | 19 | nr | nr | nr | 0 | 0 | nr | 0 | nr | |
| p=ns | p=ns | p=ns | ||||||||
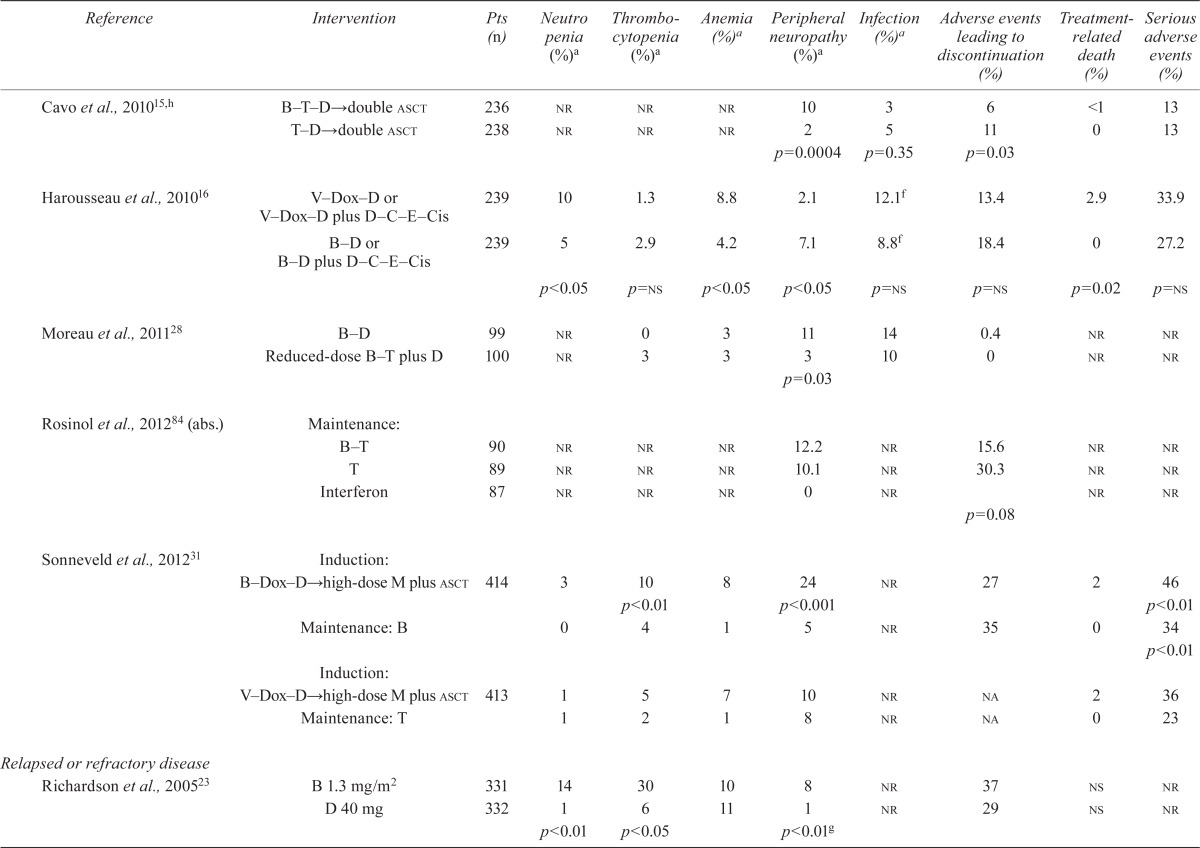
| Cavo et al., 201015,h | B T D→double asct | 236 | nr | nr | nr | 10 | 3 | 6 | <1 | 13 |
| T D→double asct | 238 | nr | nr | nr | 2 | 5 | 11 | 0 | 13 | |
| p=0.0004 | p=0.35 | p=0.03 | ||||||||
| Harousseau et al., 201016 | V–Dox–D or V–Dox–D plus D–C–E–Cis | 239 | 10 | 1.3 | 8.8 | 2.1 | 12.1f | 13.4 | 2.9 | 33.9 |
| B–D or B–D plus D–C–E–Cis | 239 | 5 | 2.9 | 4.2 | 7.1 | 8.8f | 18.4 | 0 | 27.2 | |
| p<0.05 | p=ns | p<0.05 | p<0.05 | p=ns | p=ns | p=0.02 | p=ns | |||
| Moreau et al., 201128 | B–D | 99 | nr | 0 | 3 | 11 | 14 | 0.4 | nr | nr |
| Reduced-dose B–T plus D | 100 | nr | 3 | 3 | 3 | 10 | 0 | nr | nr | |
| p=0.03 | ||||||||||
| Rosinol et al., 201284 (abs.) | Maintenance: B–T | 90 | nr | nr | nr | 12.2 | nr | 15.6 | nr | nr |
| T | 89 | nr | nr | nr | 10.1 | nr | 30.3 | nr | nr | |
| Interferon | 87 | nr | nr | nr | 0 | nr | nr | nr | ||
| p=0.08 | ||||||||||
| Sonneveld et al., 201231 | Induction: B Dox D→high-dose M plus asct | 414 | 3 | 10 | 8 | 24 | nr | 27 | 2 | 46 |
| p<0.01 | p<0.001 | p<0.01 | ||||||||
| Maintenance: B | 0 | 4 | 1 | 5 | nr | 35 | 0 | 34 | ||
| p<0.01 | ||||||||||
| Induction:V Dox D→high-dose M plus asct | 413 | 1 | 5 | 7 | 10 | nr | na | 2 | 36 | |
| Maintenance: T | 1 | 2 | 1 | 8 | nr | na | 0 | 23 | ||
| Relapsed or refractory disease | ||||||||||
| Richardson et al., 200523 | B 1.3 mg/m2 | 331 | 14 | 30 | 10 | 8 | nr | 37 | ns | nr |
| D 40 mg | 332 | 1 | 6 | 11 | 1 | nr | 29 | ns | nr | |
| p<0.01 | p<0.05 | p<0.01g | ||||||||
| Orlowski et al., 200720 | B 1.3 mg/m2 | 318 | 15 | 16 | 9 | 9 | nr | nr | 4d | nr |
| B 1.3 mg/m2 plus peg-Dox 30 mg/m2 | 318 | 29 | 23 | 9 | 4 | nr | 5 | 3d | nr | |
| p<0.001 | p=ns | p=ns | p=ns | |||||||
| Lonial et al., 201017 | Escalating dose (B 1.0, 1.3, or 1.6 mg/m2): | |||||||||
| 24 Hours before high-dose M | 19 | 45 | nr | nr | nr | 16 | nr | nr | nr | |
| 24 Hours after high-dose M | 20 | 65 | nr | nr | nr | 10 | nr | nr | nr | |
| p=ns | ||||||||||
| Reece et al., 201122 | B 1.3 mg/m2 | 21 | nr | 33 | 10 | 24 | nr | 33 | nr | nr |
| B 1.0 mg/m2 | 21 | nr | 10 | 10 | 24 | nr | 24 | nr | nr | |
| Garderet et al., 201230 | B–T–D | 135 | 11 | 17 | 8 | 16 | 14 | nr | nr | nr |
| T–D | 134 | 16 | 7 | 5 | 12 | 7 | nr | nr | nr | |
| p=0.02 | ||||||||||
| Hjorth et al., 201225 | B–D | 64 | 17 | 34 | nr | 19 | 33 | nr | nr | nr |
| T–D | 67 | 13 | 6 | nr | 7 | 24 | nr | nr | nr | |
| p=nr | p=nr | p=nr | ||||||||
| Orlowski et al., 201283 (abs.) | Siltuximab plus B | 142 | 49 | 48 | nr | nr | nr | nr | 8 | 29 |
| B | 144 | nr | nr | nr | nr | nr | nr | 5 | 24 | |
Grade 3 or 4 adverse events.
Patients with pneumonia.
In addition, bortezomib alone was discontinued in another 19% of patients.
Deaths within 30 days after the last study medication.
Adverse events reported during induction.
A significant difference was reported for between-groups grades 1–4 herpes zoster (p < 0.05).
Grade 3 adverse event.
Adverse events reported during induction.
Pts = patients; asct= autologous stem-cell transplantation; B = bortezomib; M = melphalan; P = prednisone; ns = statistically nonsignificant; nr = not reported; abs.= abstract; T = thalidomide; D = dexamethasone; C = cyclophosphamide; L = lenalidomide; ato = arsenic trioxide; V = vincristine; Dox = doxorubicin; E = etoposide; Cis = cisplatinum; peg-Dox = pegylated liposomal doxorubicin.
