Table 2.
Scope studies for hydroxamic acid synthesis.
| entrya | product | yield (%)b | |
|---|---|---|---|
| 1 |
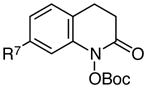
|
R7 = H | 83 |
| 2 | R7 = OMe | 77 | |
| 3 | R7 = Me | 85 | |
| 4 | R7 = CF3 | 81 | |
| 5 | R7 = CN | 72 | |
| 6 | R7 = Br | 76 | |
| 7 |
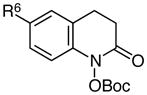
|
R6 = F | 79 |
| 8 | R6 = OMe | 0c | |
| 9 |
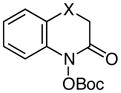
|
X = O | 78 |
| 10d | X = NTs | 53 | |
| 11d |
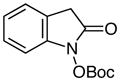
|
64 | |
| 12d |
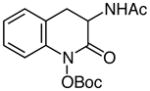
|
58 |
Reactions conducted using 2.5 mol% Ru(bpy)3Cl2, 2.1 equiv 5, and 0.1 equiv CSA unless otherwise noted.
Values represent the averaged isolated yields from two reproducible experiments.
Quinolinone 10 was isolated in 54% yield (Scheme 2).
Reaction conducted using 3 equiv of 5 and 1 equiv of CSA.
