Abstract
We showed that tumor cells with wild-type p53 and high levels of Bcl-xL are cisplatin resistant but are induced to undergo apoptosis by (−)-gossypol, making this a promising agent for overcoming cisplatin resistance. However, some cells in a population with this phenotype are not killed and continue to survive. Conversely, tumor cells with low Bcl-xL expression and either wild type or mutant p53 are relatively cisplatin sensitive and do not exhibit such high levels of apoptosis. However, these do undergo progressive loss of viability after (−)-gossypol that may not be tumor specific. We sought to elucidate the basis for these observations using cDNA microarray analysis of (−)-gossypol treated cisplatin sensitive and resistant cells. Genes in the reactive oxygen species (ROS) pathway were highly up-regulated in response to (−)-gossypol. The up-regulation was of much greater magnitude in cisplatin sensitive than resistant cells. Staining with an oxidation reporter dye confirmed differential induction of ROS in tumor cells with low Bcl-xL. As (−)-gossypol is known to undergo oxidative metabolism in vivo, ROS generation may be responsible for both off-target cytotoxicity and inactivation of the drug. In agreement with this hypothesis, oxidation of (−)-gossypol by pre-treatment with hydrogen peroxide eliminated its activity. Combined treatment with the antioxidant N-acetyl-cysteine (NAC) to block ROS increased (−)-gossypol-induced cytotoxicity to tumor but not normal cells. Furthermore, NAC increased the induction of apoptosis as measured by the sub G1 population, in both cisplatin sensitive and resistant cells. We postulate that concurrent treatment with antioxidant to block ROS prevents oxidative inactivation of (−)-gossypol and limits off-target toxicity allowing more potent (−)-gossypol-induced anti tumor activity.
Keywords: Cisplatin resistance, (−)-gossypol, Bcl-xL, reactive oxygen species, head and neck squamous cell carcinoma, apoptosis
Introduction
Evasion or blockade of apoptosis plays a key role in tumor progression and poor response to chemotherapy and radiation. The Bcl-2-family proteins function as regulators of the intrinsic mitochondrial-mediated apoptosis pathway 1-3. Bcl-2, Bcl-xL and Mcl-1 are homologous Bcl-2 family members that act to suppress the intrinsic apoptotic pathway by binding and blocking the action of pro-apoptotic Bcl-2 family members (Bax, Bak, Bad, etc.), and prevent the release of apoptotic factors from the mitochondria 1, 4, 5. Over-expression of Bcl-xL has been shown to inhibit chemotherapy- and radiation-induced apoptosis in both solid and hematologic tumors 6, 7. In previous studies of laryngeal tumors, 15% of tumors were found to over-express Bcl-2, but over-expression did not correlate with resistance to chemotherapy. However, 74% of tumors were found to over-express Bcl-xL. Low Bcl-xL expression was associated with response to therapy and larynx preservation 6. These results suggest a need for therapeutic agents that target Bcl-xL, as over-expression of Bcl-xL may be a key mechanism in chemoresistance and HNSCC tumor cell survival after therapy both in vitro and in vivo.
Gossypol is a natural compound isolated from cottonseeds, and has been used as an anti-tumor agent for several decades. The racemic form of gossypol, (±)-gossypol, was well tolerated in clinical trials 8-11. However, the mechanism of action of gossypol was not well understood until sophisticated protein binding experiments demonstrated that (−)-gossypol binds to the BH3 pocket of Bcl-2, Mcl-1 and Bcl-xL and can liberate pro-apoptotic BH3 peptides such as Bax and Bak 12. Thus, interest in gossypol as a novel therapeutic to target anti-apoptotic mechanisms in cancer has been rekindled. Our group has shown that (−)-gossypol has the capacity to induce apoptosis in vitro in HNSCC cell lines that express high levels of Bcl-xL and contain wild-type tumor-suppressor p53 13, 14. We also demonstrated that (−)-gossypol can inhibit tumor growth in an orthotopic mouse model of aggressive HNSCC 15. With (−)-gossypol showing promising results in pre-clinical xenograft mouse models of HNSCC, as well as in recent Phase I and II clinical trials in several cancer types, reducing the non-specific toxicities and improving the anti-tumor activity of (−)-gossypol will be important for developing optimized clinical treatment and combination strategies.
We previously observed that at therapeutic concentrations of (−)-gossypol, induction of apoptosis was pronounced in cisplatin-resistant (wild-type p53 and high Bcl-xL) HNSCC cell lines, but not in cisplatin-sensitive lines (mutant p53 and low Bcl-xL) 13, 16. However, cisplatin-sensitive cell lines had similar overall reductions in cell viability and survival following (−)-gossypol treatment. The discrepancy between levels of apoptosis and overall viability indicated that (−)-gossypol induces other non-apoptotic cytotoxic effects that may potentially be less selective for tumor cells. In the present study, we performed cDNA microarray analysis of HNSCC cell lines following (−)-gossypol treatment to identify patterns of induction of gene expression that would provide clues to alternative, non-apoptotic mechanisms of cell death induced by (−)-gossypol. ROS responsive genes were found to be among the most significantly up-regulated genes in response to (−)-gossypol treatment. Therefore, we examined ROS generation in vitro in response to (−)-gossypol treatment in HNSCC cell lines and investigated antioxidant treatment as a mechanism to block ROS generation and non-specific cytotoxicity in HNSCC cell lines.
Materials and Methods
Reagents
(−)-Gossypol was isolated and purified from racemic gossypol as previously described 14, and dissolved in DMSO at a stock concentration of 30 mM. NAC (Sigma, St. Louis, MO) was dissolved in media (below) immediately prior to use at a stock concentration of 1 M. 5-(and-6)-chloromethyl-2',7'-dichlorodihydrofluorescein diacetate, acetyl ester (CM-H2DCFDA) was from Molecular Probes (Eugene, OR), and dissolved in DMSO at 500 mM stock concentration. Bovine liver catalase (Calbiochem, San Diego, CA) was reconstituted in water immediately prior to use at ~16,000 units/μL.
Cell Culture
Human HNSCC cell lines were established at the University of Michigan 17, 18. Informed consent was obtained from all patients for the use and development of these cell lines. UM-SCC-5, with mutant p53 and low Bcl-xL expression, is from a primary tumor of the larynx and UM-SCC-74B, with wild-type p53 and low Bcl-xL expression, is from an intraoral recurrence of a primary tumor of the base of tongue. Pt-R (platinum resistant) cells (formerly UM-SCC-5PT) were selected for cisplatin resistance as previously described 13, 19. UM-SCC-74B was stably transfected to over-express Bcl-xL and the resulting subline was designated UM-SCC-74BxL. Cell lines were grown in DMEM containing 2 mM L-glutamine, 1% nonessential amino acids, 1% penicillin-streptomycin (Invitrogen, Carlsbad, CA), and 10% fetal bovine serum in a humidified atmosphere of 7% CO2 at 37°C. Pt-R cells were cultured in the continuous presence of 0.9 μM cisplatin to maintain the resistant phenotype.
The immortalized human oral keratinocyte cell line HOK16B 20 was grown in EpiLife media (Cascade Biologics, Portland, OR) supplemented with provided calcium (60 μM), human keratinocyte growth supplement (Cascade Biologics), and 1% penicillin-streptomycin. Media was supplemented with 1% bovine serum albumin for all experiments using these cells.
cDNA Microarray Analysis
UM-SCC-5 and Pt-R cells were plated at 2×106 cells/75 cm2, and allowed to grow overnight prior to treatment with 10 μM (−)-gossypol (~IC50). Treatment began concurrently, and cells from individual flasks were harvested for mRNA after 0, 4, 8, 12, and 24 hrs. Untreated control cells were grown and harvested simultaneously with each treatment timepoint to control for gene expression changes with growth time. mRNA was isolated using the RNeasy Mini kit according to the manufacturer’s protocol (Qiagen, Valencia, CA). In order to survey gene expression changes on a genome wide scale, we compared changes in gene expression between treated and untreated cells using a microarray containing 27,323 cDNA clones. Linear amplification of total RNA and subsequent fluorescent labeling of corresponding cDNA was carried out using the MessageAmp T7 linear amplification kit (Ambion) and cDNA labeling protocols developed at the AECOM Microarray Facility (http://microarray1k.aecom.yu.edu) 21. The ratio of the fluorescence intensities of the two dyes therefore represented a measure of differential gene expression between the two samples of interest. We considered a gene to be differentially expressed if we observed at least a 2-fold induction (or repression) of a given gene between treated and untreated cells at a given timepoint.
RT-PCR
To validate results obtained by microarray analysis, mRNA was harvested from UM-SCC-5 and Pt-R cells treated as described above. Reverse transcriptase PCR (RT-PCR) was performed according to the manufacturer’s protocol using random hexamers (Reverse Transcriptase System, Promega, Madison, WI). PCR primers were designed to span exon junctions to eliminate amplification of any contaminating genomic DNA and to generate amplicons of 300-500 base pairs. Specific primer sequences for ATF3, BNIP3, BTG1, DDIT4, GADD45A, NOXA, and VEGF are available upon request. GAPDH was used as a loading control. PCR products were run in 1% agarose gels with ethidium bromide in tris-borate-EDTA buffer, and band intensity was analyzed using ImageJ densitometry software (NIH).
Detection of Intracellular ROS
Cells were stained with CM-H2DCFDA to detect intracellular generation of ROS. CM-H2DCFDA is a cell permeable fluorescein derivative that is non-fluorescent until oxidized in the cytoplasm of live cells. After entering live cells, the diacetate groups are cleaved by intracellular esterases. Oxidation of the reduced dyes can then occur in the presence of ROS, causing the dyes to fluoresce green. CM-H2DCFDA is retained in cells because of the thiol-reactive chloromethyl group, which reacts with intracellular glutathione and other thiols (Invitrogen).
For ROS detection by fluorescence microscopy, cells were plated in 8-well chamber slides at 35,000 cells per well. After 2-3 days of growth (60-70% confluency), cells were treated with 10 μM (−)-gossypol. Following 0-24 hrs of treatment, cells were washed with Hank’s buffered salt solution (HBSS), and stained with 2.5 μM CM-H2DCFDA and 1:750 dilution of MitoTracker (from 1mM stock in DMSO, Molecular Probes) in HBSS to detect intracellular ROS and mitochondrial morphology and localization, respectively. Cells were incubated with staining solution for 15’ at 37°C, then washed and stored in HBSS.
For flow cytometry analysis, cells were plated 500,000 per flask, and treated for 0-24 hours with 10 μM (−)-gossypol. Cells were stained with 2.5 μM CM-H2DCFDA and propidium iodide (PI, Sigma, 50 μg/mL) with RNase type I-A (Roche, 100 μg/mL) in HBSS and incubated with staining solution for 15’ at 37°C. Cells were harvested and washed twice with HBSS. CMH2DCFDA fluorescence was detected by flow cytometry using FITC (488 nm) channel. PI was detected at 620 nm; cells were gated for viability by PI exclusion.
Cell Viability Assay
Cell lines were plated in 24-well plates at a density of 40,000 cells per well. After 24 hours, cells were treated and assessed for viability every 24 hours by comparison to untreated cells grown during the same time period. Cells (including media containing non-attached cells) were harvested, washed and stained with trypan blue (0.4%, Invitrogen), and viable cells (trypan blue negative) were counted using a hemacytometer following each time point. Viability was plotted as a percentage of total cells ± standard deviation. Total cell number was obtained using a cell counter (Beckman-Coulter, Fullerton, CA).
Peroxide Inactivation of (−)-Gossypol
Culture media (described above) with or without 10 μM (−)-gossypol was incubated with hydrogen peroxide (0.00003-0.03%) for 60 minutes in a humidified atmosphere of 7% CO2 at 37°C. A fresh aliquot of peroxide was used for each experiment. To inactivate peroxide ~30,000 units of bovine catalase was added to the media and allowed to incubate at room temperature for 15 minutes. Media containing the oxidized (−)-gossypol and/or catalase inactivated peroxide was then added to cells, which were assessed for viability after a 48 hour incubation as described above.
Sub-G1 Analysis
Cells were plated at 1×106 cells per flask and allowed to grow for 24 hrs prior to treatment. Cells were then treated for 48 hrs with 10 μM (−)-gossypol and/or 10 mM NAC. Following treatment, cells were trypsinized and washed with PBS, then fixed in 70% ethanol at 4°C overnight. Cellular DNA was stained with PI (50 μg/mL) and RNase type I-A (100 μg/mL) in PBS at 4°C overnight. Total DNA content and percentage of cells with sub-G1 DNA content was analyzed by flow cytometry.
Statistical Methods
All p-values reported are determined by Student's t-tests of representative experiments repeated in triplicate.
Results
(−)-Gossypol Toxicity in HNSCC Cell Lines
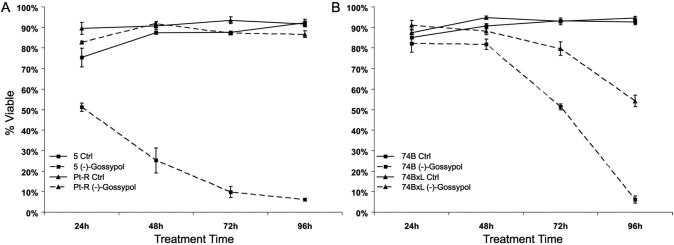
We previously reported that (−)-gossypol is a potent inducer of apoptosis in cisplatin resistant HNSCC cell lines. Apoptosis induction was more robust in the platinum resistant cell line Pt-R than the cisplatin sensitive cell line UM-SCC-5 13. In those experiments, treatment of UM-SCC-5 and Pt-R with 10 μM (−)-gossypol for 48 hours resulted in ~75% TUNEL-positive cells in Pt-R, but only ~25% in UM-SCC-5. To determine the fate of the cells that do not immediately undergo apoptosis, we examined viability at intervals after (−)-gossypol treatment. In Pt-R, a subpopulation of cells that does not undergo apoptosis shows no additional loss of viability over 96 hours, and continues to survive indefinitely. In contrast, UM-SCC-5 cells exhibit progressive loss of viability such that only 6% are viable by 96 hours (Fig. 1A).
Figure 1.
Percentage of viable cells by trypan blue exclusion assay in response to 10 μM (−)-gossypol for 24-96 hours. A. UM-SCC-5 and Pt-R, untreated versus treated. B. UM-SCC-74B and -74BxL, untreated versus treated.
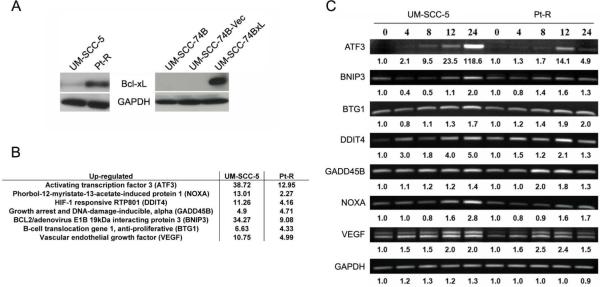
Since UM-SCC-5 and Pt-R differ in Bcl-xL expression (4.4-fold greater in Pt-R (Figure 2A)), we performed the same experiments in UM-SCC-74B, which expresses low levels of BclxL, and in UM-SCC-74BxL, which was stably transfected to over-express Bcl-xL (Figure 2A). Like UM-SCC-5 and Pt-R, treatment with 10 μM (−)-gossypol for 48 hours induces apoptosis in ~70% of UM-SCC-74BxL cells, but in only ~26% of UM-SCC-74B cells. Subsequent decreases in cell viability in UM-SCC-74B and -74BxL are like those seen in UM-SCC-5 and Pt-R (Fig. 1B), so that by 96 hours of treatment, only ~6% of UM-SCC-74B cells were viable, while ~54% of UM-SCC-74BxL cells remained viable.
Figure 2.
A. Bcl-xL and GAPDH expression in cell lines discussed. UM-SCC-74B-Vec represents a vector-only control. B. Fold increase in mRNA levels following 10 μM (−)-gossypol treatment for 24 hours for selected ROS family genes found by cDNA microarray analysis in UM-SCC-5 and Pt-R. Fold increase is shown versus paired untreated control (described in materials and methods). C. RT-PCR/gel electrophoresis validation of increases in gene expression following (−)-gossypol treatment seen in microarray analysis. Values underneath represent densitometry values of fold increase versus the respective cell line untreated control.
Changes in Gene Expression in Response to (−)-Gossypol
To examine changes in gene expression that might elucidate the mechanisms of cell death induced by (−)-gossypol treatment in HNSCC cells, microarray analysis was performed. A total of 5231 and 2045 changes in gene expression across the treatment time-course (0-24 hrs) were identified in UM-SCC-5 and Pt-R, respectively, following 10 μM (−)-gossypol treatment. Of this large set, we noted that ROS/hypoxia response genes were the only family of genes strongly up-regulated in both UM-SCC-5 and Pt-R (Figure 2B). Expression of these ROS responsive genes increased at each time point (4, 8, 12, 24 hrs), and to a greater extent in UM-SCC-5 than in Pt-R. Furthermore, this family of genes exhibited the highest levels of up-regulation of any gene family in the array. For example, at 24 hours ATF3 was up-regulated ~38-fold and 13-fold and BNIP3 was up-regulated ~34-fold and 11-fold in UM-SCC-5 and Pt-R, respectively. The microarray results were validated using RT-PCR with primers for ATF3, BNIP3, BTG1, DDIT4, GADD45B, NOXA, and VEGF. GAPDH was used as a loading/PCR control. Densitometry analysis confirmed general trends in gene expression changes following (−)-gossypol treatment (Fig. 2C).
Induction of ROS by (−)-Gossypol
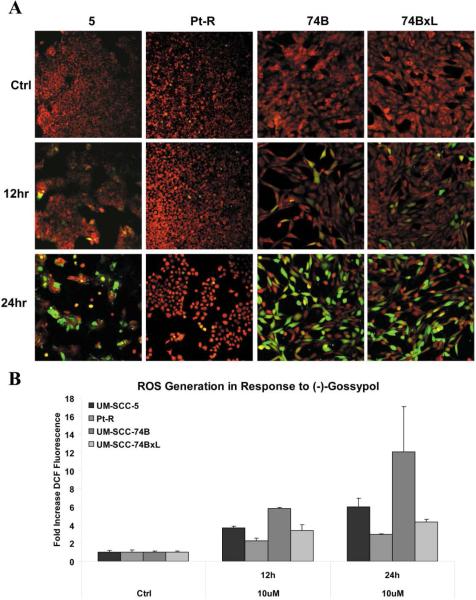
The intracellular generation of ROS by (−)-gossypol in HNSCC was examined over the same time course as the gene expression studies, using CM-H2DCFDA (fluoresces green in the presence of ROS) and MitoTracker (fluoresces red in healthy mitochondria). Following (−)-gossypol exposure, induction of ROS was observed by both fluorescence microscopy and flow cytometry. As shown in Figure 3A, increases in levels of intracellular ROS (indicated by increased green fluorescence) are apparent at 12 and 24 hrs after treatment with 10 μM (−)-gossypol. Increased green fluorescence corresponded with a loss in MitoTracker intensity (decreased red fluorescence), suggesting mitochondrial dysfunction, a known mechanism of (−)-gossypol. Flow cytometry confirmed increasing levels of ROS over time (0, 12, 24 hours) (Fig. 3B) and allowed quantification of the change in ROS activity. In the low Bcl-xL cell lines, UM-SCC-5 and -74B, the same treatment induced approximately 6.0-fold and 14.2-fold increases in ROS, respectively, relative to controls. In contrast, in the high Bcl-xL cell lines, Pt-R and UM-SCC-74BxL, 10 μM (−)-gossypol induced lower levels of ROS which were approximately 3.0-fold and 4.3-fold increases compared to vehicle control, respectively, by 24 hours.
Figure 3.
A. Representative images from confocal microscopy of indicated cell lines following treatment with 10 μM (−)-gossypol for 0, 12, or 24 hrs. MitoTracker (red) indicates functioning mitochondria, and increased fluorescence of CM-H2DCFDA (green) denotes generation of intracellular ROS. B. Quantification of increased intracellular ROS generation demonstrated in (A) by flow cytometry, as fold increase in DCF fluorescence versus untreated control. All increases in fluorescence versus control were statistically significant (p < .05). Increases in fluorescence between each high Bcl-xL and low Bcl-xL cell line were also statistically significant. For UM-SCC-5 vs. Pt-R, p = .0026 and p = .0045, at 12 and 24 hrs, respectively. For UM-SCC-74B vs. -74BxL, p < .0001 and p = .032 at 12 and 24 hrs, respectively.
Antioxidant Increases (−)-Gossypol-Induced Cytotoxicty
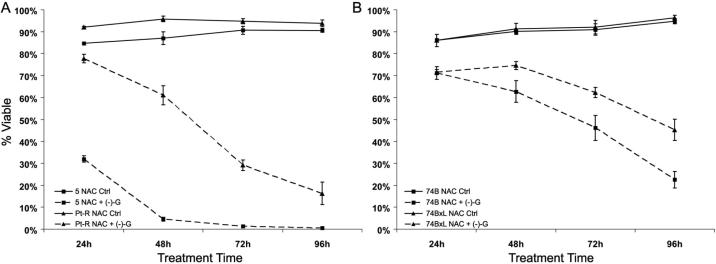
Blockade of ROS induced by (−)-gossypol through the use of antioxidants (e.g. vitamin C, tiron, trolox, N-acetyl-L-cysteine (NAC)) may prevent (−)-gossypol-induced oxidative stress in HNSCC. NAC is non-toxic in vitro and in vivo at high concentrations 22-24 and has been shown to block ROS generation and its associated cytotoxicity. To examine whether treatment with NAC will block (−)-gossypol-induced ROS generation and cell death, cells were treated concurrently with 10 μM (−)-gossypol and 10 mM NAC, and analyzed for viability by trypan blue exclusion as described below (Fig. 4).
Figure 4.
Percentage of viable cells by trypan blue exclusion assay in response to 10mM NAC with or without 10 μM (−)-gossypol for 24-96 hours. A. UM-SCC-5 and Pt-R, NAC versus combination. B. UM-SCC-74B and -74BxL, NAC versus combination.
Treatment with NAC alone did not adversely affect cell viability (Fig. 4) in any of the cell lines. However, concurrent treatment with NAC enhanced (−)-gossypol-induced cytotoxicity. In UM-SCC-5, treatment with NAC increased (−)-gossypol-induced cytotoxicity from 94% (Fig. 1A) to 99% (Fig. 4A). Even more dramatic decreases in viability (versus (−)-gossypol alone) were seen in Pt-R in which ~90% of surviving cells are viable at 96 hours after treatment with (−)-gossypol alone (Fig. 1A). In contrast, combining NAC and (−)-gossypol reduced the surviving population to ~16% (Fig. 4A). Similar effects were seen in UM-SCC-74B and -74BxL with combination treatment, though the increased toxicity was less dramatic than that in UM-SCC-5/Pt-R (Fig. 1B versus 4B). Increases in cytotoxicity with combination antioxidant/(−)-gossypol treatment were also observed in all four cells lines using other antioxidants (10 mM tiron or 2 mM trolox, data not shown).
Oxidative Inactivation of (−)-Gossypol
The increased induction of (−)-gossypol cytotoxicity by antioxidants suggested that ROS was inhibiting the (−)-gossypol activity. The lack of toxicity caused by high concentrations of antioxidants alone, and the similar effects of three unrelated antioxidants in combination with (−)-gossypol, suggest that the increased (−)-gossypol activity is a result of ROS blockade. We postulated that (−)-gossypol-induced ROS might lead to oxidation and inactivation of (−)-gossypol. If so, then antioxidant treatment can prevent oxidation of (−)-gossypol, and allow it to remain active.
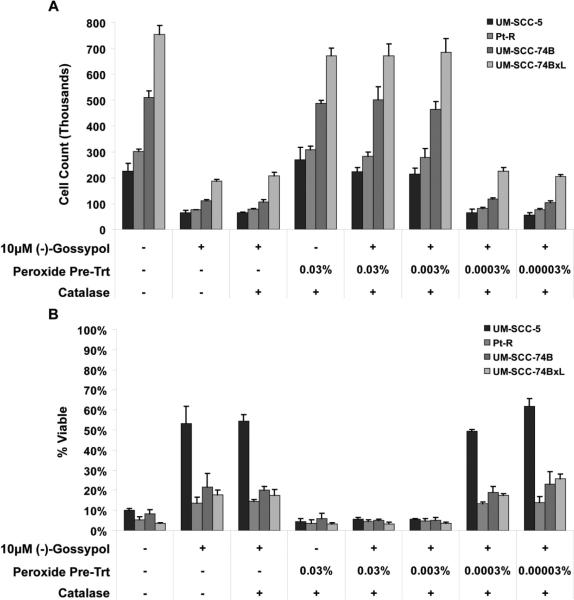
To test the hypothesis that gossypol is inactivated by oxidation, (−)-gossypol was pre-treated with hydrogen peroxide to simulate oxidation by mitochondrial ROS and ROS generated by (−)-gossypol. Pre-incubation with hydrogen peroxide effectively neutralized the cytotoxicity of (−)-gossypol in a dose dependent manner (Fig. 5). Peroxide concentrations of 0.03% and 0.003% (approximately 10 mM and 1 mM, respectively) prevented (−)-gossypol-induced cytotoxicity. Total cell counts and trypan blue positive cell counts (Fig. 5A and 5B, respectively) were almost identical to the untreated controls (columns 5 and 6 versus column 1). However, much lower peroxide concentrations of 0.0003% and 0.00003% (approximately 100 μM and 10 μM, respectively) had no inhibitory effect on (−)-gossypol cytotoxicity; cell counts and trypan blue positive cell counts were no different than (−)-gossypol treated samples (columns 7 and 8 versus 2). Similar results were obtained for all cell lines tested (UM-SCC-5, Pt-R, UMSCC-74B, -74BxL). Catalase alone (used to inactivate residual peroxide) had no effect on (−)-gossypol toxicity (column 3)., Catalase prevented the peroxide from having any adverse effect on the cells (column 4), and alone had no effect on cell counts or viability (data not shown).
Figure 5.
Cell viability following 48 hrs treatment with peroxide-inactivated (−)-gossypol. A. Whole cell counts following treatment. B. Percentage of viable (trypan-blue negative) cells following treatment. In both parts (A) and (B), lanes 1, 4, 5, and 6 represent the effect of absent or inactivated (−)-gossypol, while lanes 2, 3, 7, and 8 represent the effect of active (−)-gossypol.
Increased Apoptosis with (−)-Gossypol/Antioxidant Combination
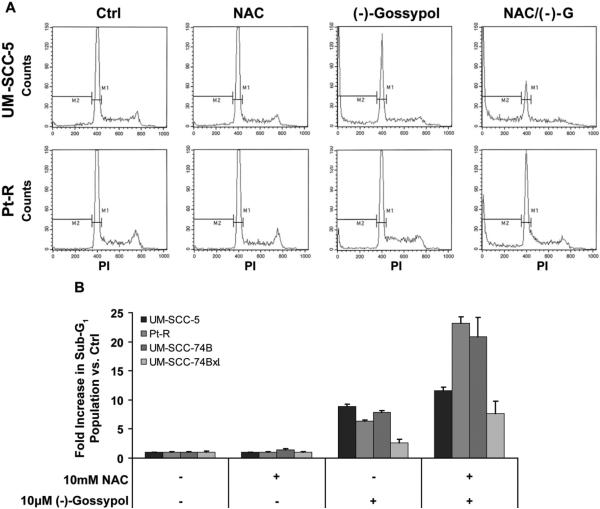
Blocking the oxidative metabolism of (−)-gossypol with antioxidant may lead to increased (−)-gossypol-induced apoptosis since without oxidative inactivation, more (−)-gossypol is available to bind Bcl-2/Bcl-xL. To test this hypothesis, cells with sub-G1 DNA content were used as a marker of apoptosis. The (−)-gossypol and antioxidant combination significantly increased the proportion of cells with sub-G1 DNA content versus (−)-gossypol treatment alone in all cell lines tested (Fig. 6). With (−)-gossypol alone, sub-G1 populations were 8.8-, 6.4-, 7.9-, and 2.6-fold greater than untreated controls in UM-SCC-5, Pt-R, UM-SCC-74B, and -74BxL, respectively. With the combination of (−)-gossypol and antioxidant, sub-G1 populations increased over control by 11.6-, 23.2-, 20.8-, and 7.7-fold in UM-SCC-5, Pt-R, UM-SCC-74B, and -74BxL, respectively. Antioxidant treatment alone caused no significant increase in sub-G1 population in any of the cell lines.
Figure 6.
Sub-G1 populations as a marker of apoptosis following NAC (10 mM) and (−)-gossypol (10 μM), alone and in combination. A. Flow cytometry histograms representative of experiments performed in triplicate for (B). Sub-G1 population PI staining cut-offs are shown by the M2 region. B. Increases in sub-G1 (apoptotic) populations following treatment shown as fold increase versus untreated control. NAC treatment was identical to untreated sub-G1 population, while (−)-gossypol and (−)-gossypol/NAC treatments each caused significant increases in sub-G1 population versus control (p < .05). For all four cell lines, increases seen in sub-G1 population with (−)-gossypol/NAC versus (−)-gossypol alone were statistically significant (p < .05).
Normal Cells are Resistant to (−)-Gossypol/Antioxidant Combination
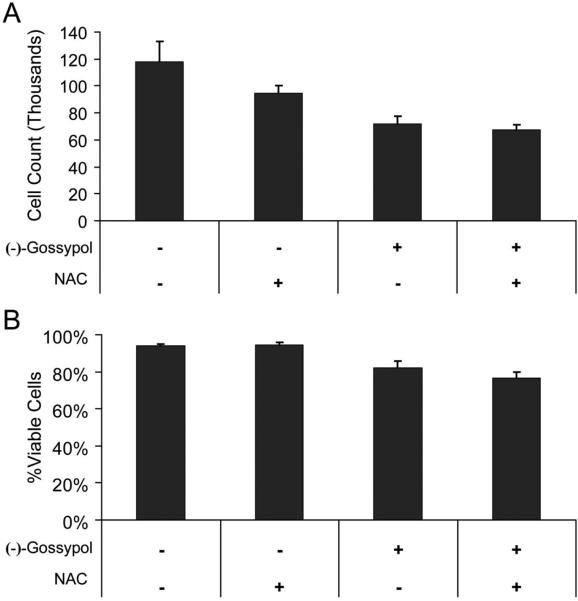
To test whether the synergistic toxicity caused by the combination of (−)-gossypol and antioxidant would specifically target HNSCC and not surrounding normal cells, immortalized oral keratinocytes (HOK16B) were treated as described below and assessed for total cell count and viability (Fig. 7). The combination of (−)-gossypol and antioxidant did not exhibit the synergistic induction of cytotoxicity that was seen in the HNSCC cell lines. Although there was some reduction in cell proliferation in the treated HOK16B cells, following 48 hours of treatment, differences in total cell count and viability by trypan blue exclusion (Figure 7A and 7B, respectively) seen between cells treated with (−)-gossypol alone and cells treated with (−)-gossypol and antioxidant was not significant. Antioxidant alone caused no significant change in cell count or viability versus untreated controls.
Figure 7.
Cell viability following 48 hrs treatment of NAC (10 mM) and (−)-gossypol (10 μM), alone and in combination, in immortalized human keratinocyte cell line HOK16B. A. Whole cell counts following treatment. B. Percentage of viable (trypan-blue negative) cells following treatment. Changes in viability measurement between (−)-gossypol alone and (−)-gossypol/NAC was not significant in (A) or (B) (p > .05).
Discussion
Chemotherapy combined with radiation provides an alternative to the potential morbidity and decreased quality of life associated with radical surgery required to treat advanced cancers of the head and neck 25. However, tumor resistance to chemotherapy and recurrence limit the usefulness of this approach. Our group has identified the anti-apoptotic protein Bcl-xL and the tumor-suppressor protein p53 as key components in chemotherapy resistance in HNSCC 6, 7, 16. The negative enantiomer of the natural compound gossypol, (−)-gossypol, has shown promise as a novel small-molecule therapeutic that targets Bcl-xL and Bcl-2 proteins 13-15. Although (−)-gossypol has been shown to selectively induce apoptosis in tumor cells in several in vitro and in vivo systems, the exact mechanisms of (−)-gossypol-induced cell death are not completely understood. Broad effects of (−)-gossypol other than binding to Bcl-2 proteins have been reported (summarized in 12), but the role of these alternative effects in tumor cytotoxicity is unknown. Understanding the mechanisms of (−)-gossypol-induced cell death in tumor cells will play an important role in the development of clinical treatment strategies.
(−)-Gossypol induces HNSCC cell lines with high levels of Bcl-xL and wild-type p53 (the cisplatin resistant phenotype) to undergo extensive apoptosis 13, 14, 16, but despite similar reductions in overall cell viability, the apoptotic response is less robust in lines with low levels of Bcl-xL or mutant p53 (the cisplatin sensitive phenotype). The observations that cisplatin sensitive cell lines undergo cell death in response to (−)-gossypol in the absence of extensive apoptosis suggests that (−)-gossypol also induces alternative mechanisms of cell death. Since (−)-gossypol has high affinity for Bcl-xL, and since it activates the intrinsic apoptotic pathway, the lack of ‘target’ in low Bcl-xL expressing cell lines or lack of functional p53 may allow (−)-gossypol to exert cytotoxic effects via alternative mechanisms 26-31.
The increase of ROS responsive genes in HNSCC cells following (−)-gossypol treatment represents an alternative mechanism of (−)-gossypol cytotoxicity. Changes in gene expression found by microarray analysis suggest that the production of free radicals by gossypol may lead to death by oxidative stress or ROS signaling. Kovacic and colleagues also suggested that the pro-oxidant properties of racemic (±)-gossypol play a role in its effects on mitochondrial respiration, anti-spermatogenic activity, and tumor cytotoxicity 32-35. Early experiments by de Peyster and colleagues showed that gossypol promotes oxygen radical generation in rat liver microsomes and human sperm, consistent with ROS production as an underlying basis for its other biological activities 33.
The primary oxidation product of gossypol, gossypolone, has been implicated in the formation of a redox system leading to additional free radical generation 32, 34, 35. In a study examining the route of oxidative degradation of gossypol, Haas and Shirley demonstrated that formation of gossypolone is the primary oxidative metabolite of gossypol 36. We have observed that (−)-gossypol accumulates at the mitochondria, which is consistent with ROS generation during electron transfer and oxidative phosphorylation. Supporting this view, studies in liver microsomes have implicated cytochrome P450 enzymes in the oxidation of gossypol to gossypolone 35. Conversion of gossypol to gossypolone may result in a stable radical system that generates additional ROS. ROS can induce cell death by oxidative stress or via downstream ROS/hypoxia signaling through induction of genes such as NOXA 37-39 and BNIP3 40-42, which we observed to be up-regulated in the microarray analysis. Thus, generation of ROS is likely to play a key role in non-apoptotic death induced by (−)-gossypol in HNSCC cells. Interestingly, we saw little or no change in glutathione-S-transferase gene expression in response to (−)-gossypol (data not shown) suggesting that the increased ROS is not secondary to reduced levels of endogenous antioxidants.
Pre-treatment of (−)-gossypol with hydrogen peroxide, a potent oxidant, resulted in loss of its biological activity. The lack of (−)-gossypol inactivation with peroxide concentrations below 1mM suggests that (−)-gossypol reacts in a stoichiometric ratio with oxygen radicals. This model of (−)-gossypol oxidation fits with in vivo models of gossypol oxidative metabolism presented by Abou-Donia and colleagues, in which each step was predicted to require 2-4 oxidations per molecule 32. These oxidized metabolites are unstable, water soluble and readily excreted in the urine 32, 34. Thus, blocking ROS with antioxidant treatment appears to protect (−)-gossypol from oxidation to inactive forms.
Greater increases in ROS responsive genes were observed in UM-SCC-5 versus Pt-R cells, consistent with the hypothesis that ROS plays a greater role in cells unable to undergo apoptosis in response to (−)-gossypol, i.e. cisplatin sensitive cells with low Bcl-xL expression. In fact, cells with high levels of (−)-gossypol-induced ROS (e.g., UM-SCC-5 and -74B) are those with low levels of (−)-gossypol-induced apoptosis. Conversely, cells with relatively low levels of (−)-gossypol-induced ROS generation (Pt-R and UM-SCC-74BxL) are those with higher levels of (−)-gossypol-induced apoptosis. Thus, we postulate that Bcl-xL could influence the levels of ROS generated by treatment with (−)-gossypol. High levels of Bcl-xL in a cell may provide an excess of ‘target’ for (−)-gossypol to bind, thereby reducing its alternative effects. Cells with high Bcl-xL may be more dependent on Bcl-xL expression for survival, such that targeting BclxL rapidly induces apoptosis by activating the intrinsic apoptotic pathway. In contrast, without excess Bcl-xL, free (−)-gossypol would be more readily oxidized to form a redox system generating further ROS as described above. Further, Bcl-xL has been shown to directly prevent generation of ROS in several systems 43, 44. Thus, over-expression of Bcl-xL may have a direct protective role against the ROS-induced mechanism of cell death, providing an explanation for why cells with high Bcl-xL favor (−)-gossypol-induced apoptosis, whereas those with low Bcl-xL expression exhibit ROS as the primary mechanism of cell death. It is therefore reasonable to postulate that antioxidant treatment plays a protective role against ROS parallel to that of Bcl-xL, causing cells to favor (−)-gossypol-induced apoptosis over non-apoptotic cell death. Interestingly, Figure 6B shows that the ability of NAC to specifically increase (−)-gossypol-induced apoptosis (versus (−)-gossypol alone) is markedly more significant in the low Bcl-xL cell lines UM-SCC-5 and -74B, suggesting that these cells have reduced antioxidant capacity without Bcl-xL. However, wild-type p53 may be required for efficient induction of apoptosis by (−)-gossypol, as the wild-type p53 cell line UM-SCC-74B shows much greater increase in apoptosis with the addition of antioxidant versus UM-SCC-5. We observed that while NAC was able to block (−)-gossypol-induced increases in ROS at early time points (12 hours) as determined by DCF fluorescence, ROS at 24 hours in cells treated with the combination was higher than cells treated with (−)-gossypol alone (data not shown). We believe this is due to the increased induction of apoptosis by active (−)-gossypol in cells treated with antioxidant and (−)-gossypol, which will cause mitochondrial dysfunction and increased ROS.
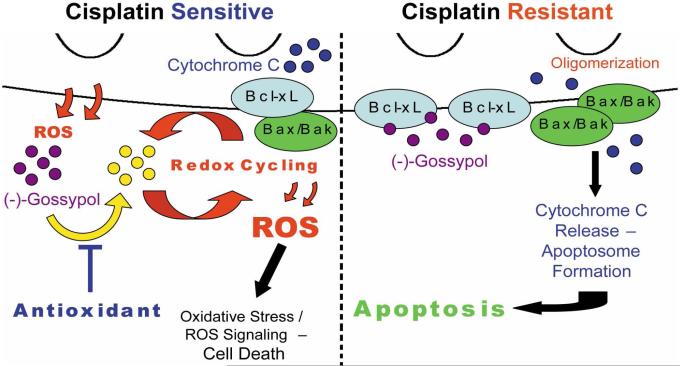
Our hypothesis that oxidation inactivates (−)-gossypol, and our demonstration that increased apoptosis occurs when antioxidants were added leads us to propose a new model of (−)-gossypol cytotoxicity in HNSCC (Fig. 8). In cisplatin sensitive HNSCC cells, (−)-gossypol cannot efficiently induce apoptosis due to lack of functional p53, or may be rapidly oxidized in the absence of high levels of Bcl-xL. ROS generated by (−)-gossypol oxidizes (−)-gossypol, leading to increased redox cycling and additional ROS generation that inactivates more (−)-gossypol. High levels of ROS generated in this manner can also lead to oxidative stress-induced cell death, which may play a role in undesired (−)-gossypol toxicity. Conversely, in cisplatin resistant cells, functional p53 allows for efficient induction of apoptosis, and high levels of Bcl-xL prevent the oxidation of (−)-gossypol. (−)-Gossypol remains active, and available to bind more Bcl-2/Bcl-xL and further increase the induction of apoptosis. The absence of significantly increased cell death in the human oral keratinocyte cell line HOK16B following treatment with (−)-gossypol and antioxidant suggests that this combination does not adversely affect normal cells, which are not dependent on anti-apoptotic proteins for survival. In contrast, tumor cells that are reliant on anti-apoptotic proteins to balance accumulated pro-death signals readily undergo apoptosis when Bcl-xL is blocked. Similarly, addition of antioxidant more efficiently kills the residual surviving cells from the tumors that express the highest levels of Bcl-xL. Thus, we expect the combination of anti-oxidant and (−)-gossypol to be more effective as an anti-cancer agent, particularly in cisplatin resistant tumor cells.
Figure 8.
Proposed molecular model of (−)-gossypol toxicity. Details are discussed in the text. Activation of Bak and Bax may instead be caused by activated BH3-only proteins sequestered by Bcl-xL and released by (−)-gossypol, but is not shown in this model.
Assessment of combined antioxidant and (−)-gossypol treatment in HNSCC in vivo models will be an important area of further investigation. NAC is well tolerated by humans 23, 24, 45, 46 and mice, and has been shown to block ROS generation in mouse tumors 22, 23, 47. Dietary supplements with high levels of NAC during (−)-gossypol treatment may inhibit ROS generation in tumor cells, prevent oxidation of (−)-gossypol, and increase its efficacy and specificity against HNSCC cells. Targeting Bcl-xL/Bcl-2 while minimizing alternative effects of (−)-gossypol could increase specificity for tumors versus normal tissue, thereby increasing the therapeutic index. Additionally, preserving (−)-gossypol in an active form with antioxidants may allow (−)-gossypol treatment doses to be lowered, further decreasing undesired toxicity, in vivo.
Acknowledgments
NIH NIDCR (R01 DE13346) & NIDCD (P30 DC 05188), NIH NCI (R01 CA83087), University of Michigan's Head and Neck Cancer SPORE grant (P50 CA97248) & Cancer Center Support Grant (P30 CA46592). We thank the Albert Einstein College of Medicine Microarray Facility and the University of Michigan Flow Cytometry Core for technical assistance.
Literature Cited
- 1.Chao DT, Korsmeyer SJ. BCL-2 family: regulators of cell death. Annual review of immunology. 1998;16:395–419. doi: 10.1146/annurev.immunol.16.1.395. [DOI] [PubMed] [Google Scholar]
- 2.Chao DT, Linette GP, Boise LH, White LS, Thompson CB, Korsmeyer SJ. Bcl-XL and Bcl-2 repress a common pathway of cell death. The Journal of experimental medicine. 1995;182:821–8. doi: 10.1084/jem.182.3.821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Korsmeyer SJ. Programmed cell death: Bcl-2. Important advances in oncology. 1993:19–28. [PubMed] [Google Scholar]
- 4.Korsmeyer SJ. BCL-2 gene family and the regulation of programmed cell death. Cancer research. 1999;59:1693s–700s. [PubMed] [Google Scholar]
- 5.Johnstone RW, Ruefli AA, Lowe SW. Apoptosis: a link between cancer genetics and chemotherapy. Cell. 2002;108:153–64. doi: 10.1016/s0092-8674(02)00625-6. [DOI] [PubMed] [Google Scholar]
- 6.Trask DK, Wolf GT, Bradford CR, Fisher SG, Devaney K, Johnson M, Singleton T, Wicha M. Expression of Bcl-2 family proteins in advanced laryngeal squamous cell carcinoma: correlation with response to chemotherapy and organ preservation. The Laryngoscope. 2002;112:638–44. doi: 10.1097/00005537-200204000-00009. [DOI] [PubMed] [Google Scholar]
- 7.Bradford CR. Predictive factors in head and neck cancer. Hematology/oncology clinics of North America. 1999;13:777–85. doi: 10.1016/s0889-8588(05)70092-7. [DOI] [PubMed] [Google Scholar]
- 8.Bushunow P, Reidenberg MM, Wasenko J, Winfield J, Lorenzo B, Lemke S, Himpler B, Corona R, Coyle T. Gossypol treatment of recurrent adult malignant gliomas. Journal of neuro oncology. 1999;43:79–86. doi: 10.1023/a:1006267902186. [DOI] [PubMed] [Google Scholar]
- 9.Flack MR, Pyle RG, Mullen NM, Lorenzo B, Wu YW, Knazek RA, Nisula BC, Reidenberg MM. Oral gossypol in the treatment of metastatic adrenal cancer. The Journal of clinical endocrinology and metabolism. 1993;76:1019–24. doi: 10.1210/jcem.76.4.8473376. [DOI] [PubMed] [Google Scholar]
- 10.Stein RC, Joseph AE, Matlin SA, Cunningham DC, Ford HT, Coombes RC. A preliminary clinical study of gossypol in advanced human cancer. Cancer chemotherapy and pharmacology. 1992;30:480–2. doi: 10.1007/BF00685601. [DOI] [PubMed] [Google Scholar]
- 11.Van Poznak C, Seidman AD, Reidenberg MM, Moasser MM, Sklarin N, Van Zee K, Borgen P, Gollub M, Bacotti D, Yao TJ, Bloch R, Ligueros M, Sonenberg M, Norton L, Hudis C. Oral gossypol in the treatment of patients with refractory metastatic breast cancer: a phase I/II clinical trial. Breast cancer research and treatment. 2001;66:239–48. doi: 10.1023/a:1010686204736. [DOI] [PubMed] [Google Scholar]
- 12.Zhang M, Liu H, Guo R, Ling Y, Wu X, Li B, Roller PP, Wang S, Yang D. Molecular mechanism of gossypol-induced cell growth inhibition and cell death of HT-29 human colon carcinoma cells. Biochemical pharmacology. 2003;66:93–103. doi: 10.1016/s0006-2952(03)00248-x. [DOI] [PubMed] [Google Scholar]
- 13.Bauer JA, Trask DK, Kumar B, Los G, Castro J, Lee JS, Chen J, Wang S, Bradford CR, Carey TE. Reversal of cisplatin resistance with a BH3 mimetic, (-)-gossypol, in head and neck cancer cells: role of wild-type p53 and Bcl-xL. Molecular cancer therapeutics. 2005;4:1096–104. doi: 10.1158/1535-7163.MCT-05-0081. [DOI] [PubMed] [Google Scholar]
- 14.Oliver CL, Bauer JA, Wolter KG, Ubell ML, Narayan A, O'Connell KM, Fisher SG, Wang S, Wu X, Ji M, Carey TE, Bradford CR. In vitro effects of the BH3 mimetic, (-)-gossypol, on head and neck squamous cell carcinoma cells. Clin Cancer Res. 2004;10:7757–63. doi: 10.1158/1078-0432.CCR-04-0551. [DOI] [PubMed] [Google Scholar]
- 15.Wolter KG, Wang SJ, Henson BS, Wang S, Griffith KA, Kumar B, Chen J, Carey TE, Bradford CR, D'Silva NJ. (-)-gossypol inhibits growth and promotes apoptosis of human head and neck squamous cell carcinoma in vivo. Neoplasia (New York, NY. 2006;8:163–72. doi: 10.1593/neo.05691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bauer JA, Kumar B, Cordell KG, Prince ME, Tran HH, Wolf GT, Chepeha DB, Teknos TN, Wang S, Eisbruch A, Tsien CI, Urba SG, Worden FP, Lee J, Griffith KA, Taylor JM, D'Silva N, Wang SJ, Wolter KG, Henson B, Fisher SG, Carey TE, Bradford CR. Targeting apoptosis to overcome cisplatin resistance: a translational study in head and neck cancer. Int J Radiat Oncol Biol Phys. 2007;69:S106–8. doi: 10.1016/j.ijrobp.2007.05.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carey TE, Kimmel KA, Schwartz DR, Richter DE, Baker SR, Krause CJ. Antibodies to human squamous cell carcinoma. Otolaryngol Head Neck Surg. 1983;91:482–91. doi: 10.1177/019459988309100503. [DOI] [PubMed] [Google Scholar]
- 18.Krause CJ, Carey TE, Ott RW, Hurbis C, McClatchey KD, Regezi JA. Human squamous cell carcinoma. Establishment and characterization of new permanent cell lines. Arch Otolaryngol. 1981;107:703–10. doi: 10.1001/archotol.1981.00790470051012. [DOI] [PubMed] [Google Scholar]
- 19.Nakata B, Albright KD, Barton RM, Howell SB, Los G. Synergistic interaction between cisplatin and tamoxifen delays the emergence of cisplatin resistance in head and neck cancer cell lines. Cancer chemotherapy and pharmacology. 1995;35:511–8. doi: 10.1007/BF00686837. [DOI] [PubMed] [Google Scholar]
- 20.Park NH, Min BM, Li SL, Huang MZ, Cherick HM, Doniger J. Immortalization of normal human oral keratinocytes with type 16 human papillomavirus. Carcinogenesis. 1991;12:1627–31. doi: 10.1093/carcin/12.9.1627. [DOI] [PubMed] [Google Scholar]
- 21.Belbin TJ, Singh B, Smith RV, Socci ND, Wreesmann VB, Sanchez-Carbayo M, Masterson J, Patel S, Cordon-Cardo C, Prystowsky MB, Childs G. Molecular profiling of tumor progression in head and neck cancer. Archives of otolaryngology--head & neck surgery. 2005;131:10–8. doi: 10.1001/archotol.131.1.10. [DOI] [PubMed] [Google Scholar]
- 22.Sablina AA, Budanov AV, Ilyinskaya GV, Agapova LS, Kravchenko JE, Chumakov PM. The antioxidant function of the p53 tumor suppressor. Nature medicine. 2005;11:1306–13. doi: 10.1038/nm1320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sener G, Sehirli AO, Ayanoglu-Dulger G. Protective effects of melatonin, vitamin E and N-acetylcysteine against acetaminophen toxicity in mice: a comparative study. Journal of pineal research. 2003;35:61–8. doi: 10.1034/j.1600-079x.2003.00050.x. [DOI] [PubMed] [Google Scholar]
- 24.Tsai CL, Chang WT, Weng TI, Fang CC, Walson PD. A patient-tailored N-acetylcysteine protocol for acute acetaminophen intoxication. Clinical therapeutics. 2005;27:336–41. doi: 10.1016/j.clinthera.2005.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Urba S, Wolf G, Eisbruch A, Worden F, Lee J, Bradford C, Teknos T, Chepeha D, Prince M, Hogikyan N, Taylor J. Single-cycle induction chemotherapy selects patients with advanced laryngeal cancer for combined chemoradiation: a new treatment paradigm. J Clin Oncol. 2006;24:593–8. doi: 10.1200/JCO.2005.01.2047. [DOI] [PubMed] [Google Scholar]
- 26.Hou DX, Uto T, Tong X, Takeshita T, Tanigawa S, Imamura I, Ose T, Fujii M. Involvement of reactive oxygen species-independent mitochondrial pathway in gossypol-induced apoptosis. Archives of biochemistry and biophysics. 2004;428:179–87. doi: 10.1016/j.abb.2004.06.007. [DOI] [PubMed] [Google Scholar]
- 27.Jan CR, Lin MC, Chou KJ, Huang JK. Novel effects of gossypol, a chemical contraceptive in man: mobilization of internal Ca(2+) and activation of external Ca(2+) entry in intact cells. Biochimica et biophysica acta. 2000;1496:270–6. doi: 10.1016/s0167-4889(00)00033-1. [DOI] [PubMed] [Google Scholar]
- 28.Jarvis WD, Turner AJ, Povirk LF, Traylor RS, Grant S. Induction of apoptotic DNA fragmentation and cell death in HL-60 human promyelocytic leukemia cells by pharmacological inhibitors of protein kinase C. Cancer research. 1994;54:1707–14. [PubMed] [Google Scholar]
- 29.Ligueros M, Jeoung D, Tang B, Hochhauser D, Reidenberg MM, Sonenberg M. Gossypol inhibition of mitosis, cyclin D1 and Rb protein in human mammary cancer cells and cyclin-D1 transfected human fibrosarcoma cells. British journal of cancer. 1997;76:21–8. doi: 10.1038/bjc.1997.330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shidaifat F, Canatan H, Kulp SK, Sugimoto Y, Chang WY, Zhang Y, Brueggemeier RW, Somers WJ, Lin YC. Inhibition of human prostate cancer cells growth by gossypol is associated with stimulation of transforming growth factor-beta. Cancer letters. 1996;107:37–44. doi: 10.1016/0304-3835(96)04340-6. [DOI] [PubMed] [Google Scholar]
- 31.Shidaifat F, Canatan H, Kulp SK, Sugimoto Y, Zhang Y, Brueggemeier RW, Somers WJ, Chang WY, Wang HC, Lin YC. Gossypol arrests human benign prostatic hyperplastic cell growth at G0/G1 phase of the cell cycle. Anticancer research. 1997;17:1003–9. [PubMed] [Google Scholar]
- 32.Abou-Donia MB. Physiological effects and metabolism of gossypol. Residue reviews. 1976;61:125–60. doi: 10.1007/978-1-4613-9401-3_5. [DOI] [PubMed] [Google Scholar]
- 33.de Peyster A, Quintanilha A, Packer L, Smith MT. Oxygen radical formation induced by gossypol in rat liver microsomes and human sperm. Biochemical and biophysical research communications. 1984;118:573–9. doi: 10.1016/0006-291x(84)91341-x. [DOI] [PubMed] [Google Scholar]
- 34.Kovacic P. Mechanism of drug and toxic actions of gossypol: focus on reactive oxygen species and electron transfer. Current medicinal chemistry. 2003;10:2711–8. doi: 10.2174/0929867033456369. [DOI] [PubMed] [Google Scholar]
- 35.Yu YW. Probing into the mechanism of action, metabolism and toxicity of gossypol by studying its (+)- and (-)-stereoisomers. Journal of ethnopharmacology. 1987;20:65–78. doi: 10.1016/0378-8741(87)90120-6. [DOI] [PubMed] [Google Scholar]
- 36.Haas RH, Shirley DA. The Oxidation of Gossypol. II. Formation of Gossypolone with Ferric Chloride. Journal of organic chemistry. 1965;30:4111–3. [Google Scholar]
- 37.Armstrong JL, Veal GJ, Redfern CP, Lovat PE. Role of Noxa in p53-independent fenretinide-induced apoptosis of neuroectodermal tumours. Apoptosis. 2007 doi: 10.1007/s10495-006-0020-1. [DOI] [PubMed] [Google Scholar]
- 38.Punj V, Chakrabarty AM. Redox proteins in mammalian cell death: an evolutionarily conserved function in mitochondria and prokaryotes. Cellular microbiology. 2003;5:225–31. doi: 10.1046/j.1462-5822.2003.00269.x. [DOI] [PubMed] [Google Scholar]
- 39.Webster KA, Graham RM, Thompson JW, Spiga MG, Frazier DP, Wilson A, Bishopric NH. Redox stress and the contributions of BH3-only proteins to infarction. Antioxidants & redox signaling. 2006;8:1667–76. doi: 10.1089/ars.2006.8.1667. [DOI] [PubMed] [Google Scholar]
- 40.Fei P, Wang W, Kim SH, Wang S, Burns TF, Sax JK, Buzzai M, Dicker DT, McKenna WG, Bernhard EJ, El-Deiry WS. Bnip3L is induced by p53 under hypoxia, and its knockdown promotes tumor growth. Cancer cell. 2004;6:597–609. doi: 10.1016/j.ccr.2004.10.012. [DOI] [PubMed] [Google Scholar]
- 41.Guo K, Searfoss G, Krolikowski D, Pagnoni M, Franks C, Clark K, Yu KT, Jaye M, Ivashchenko Y. Hypoxia induces the expression of the pro-apoptotic gene BNIP3. Cell death and differentiation. 2001;8:367–76. doi: 10.1038/sj.cdd.4400810. [DOI] [PubMed] [Google Scholar]
- 42.Manka D, Spicer Z, Millhorn DE. Bcl-2/adenovirus E1B 19 kDa interacting protein-3 knockdown enables growth of breast cancer metastases in the lung, liver, and bone. Cancer research. 2005;65:11689–93. doi: 10.1158/0008-5472.CAN-05-3091. [DOI] [PubMed] [Google Scholar]
- 43.Shimizu S, Eguchi Y, Kosaka H, Kamiike W, Matsuda H, Tsujimoto Y. Prevention of hypoxia-induced cell death by Bcl-2 and Bcl-xL. Nature. 1995;374:811–3. doi: 10.1038/374811a0. [DOI] [PubMed] [Google Scholar]
- 44.Trancikova A, Weisova P, Kissova I, Zeman I, Kolarov J. Production of reactive oxygen species and loss of viability in yeast mitochondrial mutants: protective effect of Bcl-xL. FEMS yeast research. 2004;5:149–56. doi: 10.1016/j.femsyr.2004.06.014. [DOI] [PubMed] [Google Scholar]
- 45.Perry HE, Shannon MW. Efficacy of oral versus intravenous N-acetylcysteine in acetaminophen overdose: results of an open-label, clinical trial. The Journal of pediatrics. 1998;132:149–52. doi: 10.1016/s0022-3476(98)70501-3. [DOI] [PubMed] [Google Scholar]
- 46.Tucker JR. Late-presenting acute acetaminophen toxicity and the role of N-acetylcysteine. Pediatric emergency care. 1998;14:424–6. doi: 10.1097/00006565-199812000-00013. [DOI] [PubMed] [Google Scholar]
- 47.Tomko RJ, Jr., Bansal P, Lazo JS. Airing out an antioxidant role for the tumor suppressor p53. Molecular interventions. 2006;6:23–5. 2. doi: 10.1124/mi.6.1.5. [DOI] [PubMed] [Google Scholar]