Abstract
Tissue-specific gene expression is regulated by epigenetic modification involving trans-acting factors. Here, we identified that the human MAGEB16 gene and its mouse homolog, Mageb16, are only expressed in the testis. To investigate the mechanism governing their expression, the promoter methylation status of these genes was examined in different samples. Two CpG islands (CGIs) in the 5' upstream region of MAGEB16 were highly demethylated in human testes, whereas they were methylated in cells without MAGEB16 expression. Similarly, the CGI in Mageb16 was hypomethylated in mouse testes but hypermethylated in other tissues and cells without Mageb16 expression. Additionally, the expression of these genes could be activated by treatment with the demethylation agent 5'-aza-2'-deoxycytidine (5'-aza-CdR). Luciferase assays revealed that both gene promoter activities were inhibited by methylation of the CGI regions. Therefore, we propose that the testis-specific expression of MAGEB16 and Mageb16 is regulated by the methylation status of their promoter regions. [BMB Reports 2014; 47(2): 86-91]
Keywords: CpG island, DNA demethylation, MAGEB16, Mageb16, Testis-specific expression
INTRODUCTION
Mammalian spermatozoan development is a complex process and it has been hypothesized that an exceptionally large number of genes (approximately 4% of mammalian genes) are specifically expressed in the male germ line (1). The identification of these genes and their biological functions poses a considerablechallenge but may be useful in elucidating the molecular process of spermatogenesis.
The X and Y chromosomes are unique because men only have a single copy of each chromosome. The involvement of the Y chromosome in spermatogenesis is clear because the deletion of one or more azoospermia factor (AZF) regions leads to human spermatogenic failure. The X chromosome was also proposed to play an important role in spermatogenesis because multiple genes located on the X chromosome are predominantly expressed in mammalian testicular tissues (2). Up to now, the function of many genes on the X chromosome, especially the cancer-testis (CT) genes, has not been studied so far.
MAGE genes were initially identified because they encode tumor antigens that can be recognized by cytolytic T lymphocytes from cancer patients (3). Based on their expression patterns and functions, MAGE genes can be classified into type I or type II. Type I genes are composed of three subfamilies (MAGE-A, -B and -C) and are expressed in highly proliferating cells, such as tumors, placenta and germ line cells (4,5). And most type I MAGE genes are located on the X chromosome and encode tumor antigens; they are classified as members of the CT–X antigen gene family and play a key role in the proliferation and differentiation of germ and tumor cells (6-8).
Epigenetic modification often plays a role secondary to the presence or absence of trans-acting factors in the control of tissue-specific gene activation (9). Ectopic hypomethylation of genomic DNA has been associated with CT-X gene expression and the demethylation of critical CpG residues within their promoter regions (7). For example, the activation of MAGEA1 in melanoma is associated with hypomethylation of a restricted region overlapping the promoter, and its expression can be induced in certain cancer cell lines by treating non-expressing cells with 5'-aza-CdR (10). Recently, MAGEB2 was also found to be activated by promoter demethylation in head and neck squamous cell carcinoma (11).
In a search for testis-specific genes, we found that MAGEB16, one gene of the MAGE-B subfamily that is localized to Xp21.1 from the NCBI Unigene database (GenBank accession No.: NM_001099921.1), may be uniquely expressed in human testicular tissue similar to other MAGE-B subfamily genes. Hence, in this report, we further investigated the expression pattern of MAGEB16 and its homolog, Mageb16, in different tissues and cells from humans and mice, respectively, and discovered that their testis-specific expression was regulated by the methylation status of CpG sites in their promoter regions.
RESULTS
Expression profile of the human MAGEB16 and mouse Mageb16 genes
To investigate the expression profile of the MAGEB16 gene in human tissues and cells, we performed PCR amplification from 16 cDNA pools derived from different human tissues and cells, including heart, liver, brain, lung, kidney, spleen, pancreas, prostate, ovary, small intestine, colon, peripheral leukocytes, placenta, thymus, skeletal muscle and testis. The results showed that MAGEB16 mRNA was only expressed in the testis (Fig. 1A and C). In parallel, the expression of the Mageb16 gene was examined in 12 different mouse tissues and cells, including heart, liver, spleen, brain, lung, kidney, pancreas, ovary, small intestine, thymus, peripheral leukocytes and testis. The results indicated that Mageb16 mRNA was abundantly expressed in mouse testis (Fig. 1B and D). Additionally, the amount of Mageb16 mRNA in the mouse testis was approximately three-fold more than that of MAGEB16 in human testis (Fig. 1B).
Fig. 1. The expression profile of the MAGEB16 and Mageb16 genes in human and mouse tissues and cells. (A) RT-PCR analysis of MAGEB16 mRNA from 16 human tissue and cell samples. (B) RT-PCR analysis of Mageb16 mRNA from 12 mouse tissue and cell samples. (C) QRT-PCR analysis of MAGEB16 mRNA from 16 human tissue and cell samples. The amount of Mageb16 mRNA in mouse testis was used as a control and was set to 1 arbitrary unit. (D) QRT-PCR analysis of Mageb16 mRNA from 12 mouse tissue and cell samples. (E) QRT-PCR analysis of Mageb16 mRNA from embryos and testis from different developmental stages. The column labeled “4 m” indicates mRNA extracted from the testis of 4-month-old mice.
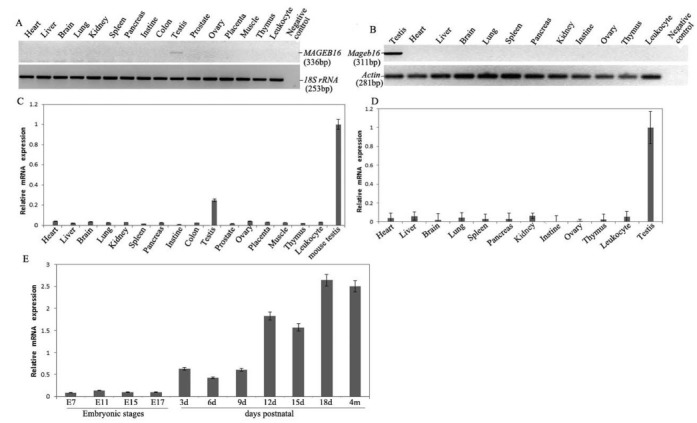
We also investigated the expression pattern of the Mageb16 gene at four mouse embryonic stages and in testis tissues from different postnatal developmental time points. Low expression of Mageb16 was observed in the embryonic stages. The mRNA level increased with development and reached its highest level at day 18 postnatal, and the expression stayed approximately at this level in the testes of adult mice (Fig. 1E).
Methylation of CGIs in the promoter regions of the MAGEB16 and Mageb16 genes was correlated with gene expression in different tissues and cells
To understand the mechanism governing the specificity of MAGEB16 and Mageb16 gene expression, we investigated the methylation status of their CpG sites. First, the CGIs in the 5' upstream regions were analyzed with the following parameters, using the online tool MethPrimer (http://www.urogene.org/cgibin/methprimer/methprimer.cgi): length >100 bp, C+ Gs/total bases >50%, and CpG observed/CpG expected >0.6. Two typical CGIs containing 12 and 17 CpG sites were found in the upstream region of the human MAGEB16 gene at positions -520 to -420 bp and -173 to +39 bp relative to the putative transcription initiation site (TIS). One CGI situated in the mouse Mageb16 gene included 16 CpG sites from -63bp to +101 bp relative to the TIS (Fig. 2A and B).
Fig. 2. The methylation status of CGIs in the 5' upstream regions of MAGEB-16 and Mageb16. (A) Two CGIs located in the 5' upstream region of the MAGEB16 gene and their methylation status in genomic DNA from different human tissues and cells. (B) One CGI located in the 5' upstream region of the Mageb16 gene and its methylation status in genomic DNA from different mouse tissues and cells. TIS: transcription initiation site.
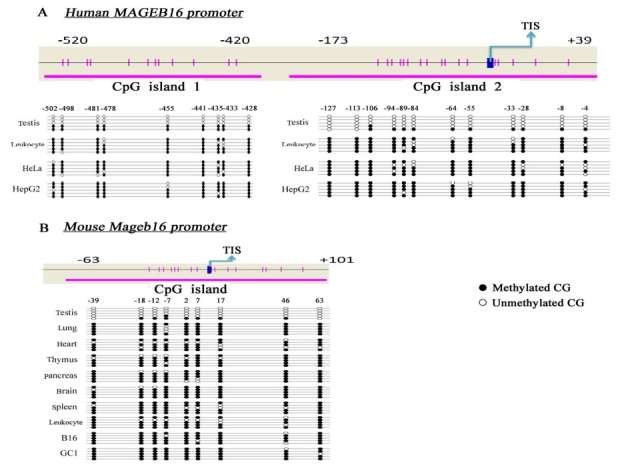
Then, bisulfite genomic sequencing was performed to examine the methylation status of the selected CGIs in genomic DNA extracted from different human and mouse tissues and cells. It was found that the CpG sites in the two CGIs of the MAGEB16 gene were highly unmethylated in human testis tissue, in contrast to the strong methylation of these CpG sites in peripheral leukocytes and two tumor cell lines, HeLa and HepG2, in which the MAGEB16 gene is unexpressed (Fig. 2A). Similarly, the bisulfite genomic sequencing results showed that CpG sites in the CGI of Mageb16 were predominantly unmethylated in mouse testis. In contrast, these CpG sites were highly methylated in mouse lung, heart, thymus, pancreas, brain and spleen tissues, peripheral leukocytes and B16 and GC-1 cells, all of which lack Mageb16 expression (Fig. 2B).
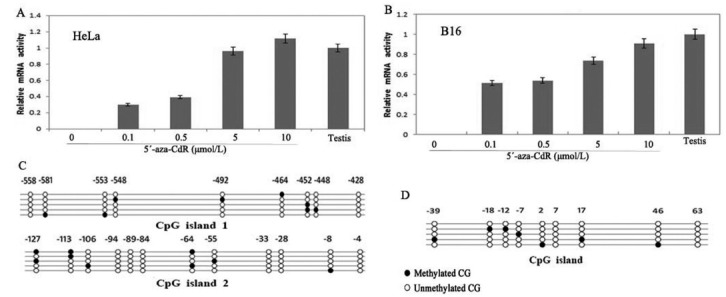
5'-aza-CdR is a commonly used DNA methyltransferase inhibitor and is able to demethylate DNA genome-wide in almost all cell culture systems (12). To further confirm that hypermethylation is responsible for the silencing of MAGEB16 and Mageb16 expression, HeLa, HepG2, B16 and GC-1 cells were treated with 5'-aza-CdR. The expression of MAGEB16 and Mageb16 was restored in HeLa and B16 cells (Fig. 3A and B). The up-regulation occurred in a dose-dependent manner, and the maximal induction of MAGEB16 and Mageb16 mRNA was observed at 10 μmol/L 5'-aza-CdR (Fig. 3A and B). Then, bisulfite sequencing was performed to analyze the methylation status in cells treated with 5'-aza-CdR, and the results showed that most of CpG sites in both the MAGEB16 and Mageb16 genes were demethylated in the treated cells (Fig. 3C and D). The similar results were obtained from HepG2 and GC-1 cell lines (Data not shown).
Fig. 3. MAGEB16 and Mageb16 mRNA expression in HeLa and B16 cells after treatment with 5'-aza-CdR. (A) HeLa cells were treated with DMSO (0) or 5'-aza-CdR at the designated concentrations (0.1, 0.5, 1, 5 or 10 μmol/L) and were evaluated for MAGEB16 mRNA expression. (B) B16 cells were treated with DMSO (0) or 5'-aza-CdR at the designated concentrations (0.1, 0.5, 1, 5 or 10 μmol/L) and were evaluated for Mageb16 mRNA expression. The levels of mRNA were determined by QRT-PCR on isolated RNA. The testis values were used as 1 arbitrary unit=100%. (C) The methylation status of the MAGEB16 CGIs in HeLa cells treated by 10 μmol/L 5'-aza-CdR. (D) The methylation status of the Mageb16 CGI in B16 cells treated by 10 μmol/L 5'-aza-CdR. Bisulfite genomic sequencing displayed extensive demethylation in treated cells.
The methylation status of CGIs in the 5' upstream regions of MAGEB16 and Mageb16 mediates the expression of a luciferase reporter gene
To examine the effect of the methylation status of CGIs in the 5' upstream regions of MAGEB16 and Mageb16 in the regulation of gene expression in vitro, we cloned several 5' upstream sequences of MAGEB16 and Mageb16 and sub-cloned them into the pGL3-Basic luciferase reporter vector (Fig. 4A and C). The MAGEB16 and Mageb16 constructs were transiently transfected into human HEK293T and mouse GC-1 cells, respectively. The results showed that the largest MAGEB16 construct, (-1102/+50), displayed the highest activity. The transcriptional activity of construct (-272/+50) was significantly decreased by approximately 3-fold compared with that of the largest construct, but the smallest construct, (-171/+50), displayed higher transcriptional activity (Fig. 4A). These results suggest that the basal promoter of the MAGEB16 gene is located in the region from -171 to +50 nt. It is worth noting that CGI-2 of MAGEB16 is also located in the putative basal promoter region (Fig. 4A).
Fig. 4. In vitro analysis of the methylation status of the CGIs in the 5' upstream regions of MAGEB16 and Mageb16 in regulating the expression of a luciferase reporter gene. (A) Functional deletion constructs of the MAGEB16 gene promoter region and the relative luciferase activity of each deletion construct following transient transfection into HEK293 cells. (B) The luciferase activity of each methylated deletion construct of the MAGEB16 gene. (C) Functional deletion constructs of the Mageb16 gene promoter region and the relative luciferase activity of each deletion construct following transient transfection into GC-1 cells. (D) The luciferase activity of each methylated deletion construct of the Mageb16 gene.
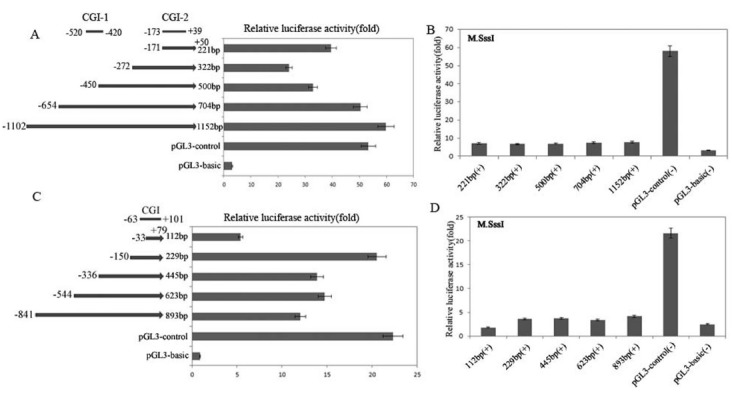
As shown in Fig. 4C, in GC-1 cells, the Mageb16 construct (-150/+79) displayed the highest activity, approximately 2-fold over that of the largest construct, (-814/+79). This result indicates that the core promoter of the Mageb16 gene is present in the -150 to +79 nt region. Additionally, the -63 to +101 bp CGI region largely fell within the promoter region of the Mageb16 gene.
Further, we performed in vitro methylation analysis to confirm whether the promoter activities of the MAGEB16 and Mageb16 genes were inhibited by CGI methylation. All the constructs were treated with the methyltransferase SssI, which resulted in the methylation of the CpG sites in the constructs. Then, the methylated MAGEB16 and Mageb16 constructs were transiently transfected into HEK293T and GC-1 cells, respectively, and the luciferase activity was measured. As shown in Fig. 4B and D, the luciferase activity of the treated constructs was clearly repressed. This further confirmed the hypothesis that the promoter activity of the MAGEB16 and Mageb16 genes is mediated by the methylation status of the CGI region.
DISCUSSION
The MAGEB16 gene is located on Xp21.1, and here, we have shown that it is restrictively expressed in human testes, which indicates that MAGEB16 may be a CT-X gene, although it was not detected in HeLa and HepG2 tumor cells. To confirm that the MAGEB16 gene encodes a CT antigen, future investigations should be undertaken to screen its expression and to test the sero-reactivity of MAGEB16 antibodies in cancer patients, in a manner similar to previous reports (13,14). The mouse Mageb16 gene was also uniquely expressed in the testis, and the mRNA level in the testis increased through development and reached the highest level of expression at day 18 postnatal. These results indicate that Mageb16 may play a role in mouse spermatogenesis. In addition, a recent study described that the mouse Mageb16 gene was expressed at a high level in undifferentiated embryonic stem cells (ESCs), but the expression was very low in somatic cells derived from ESCs (15), which indicates that the Mageb16 gene may also be involved in the proliferation and differentiation of ESCs. Further investigations into these potential biological functions of the Mageb16 gene could be undertaken using ESCs as an in vitro model.
Genome–wide DNA demethylation is often observed in male germ cells and is known to be an important mechanism involved in mammalian germ-cell development (16,17). In this study, we found that the status of DNA methylation correlated with the specific expression of MAGEB16 and Mageb16 genes. Growing evidence suggests that many transcription factors have methylation sensitivity, including SP1 and CREB (18,19). There are also several potential binding sites for transcription factors, such as GATA-1, CREB, SOX5 and SOX9, in both the CGI-2 region of MAGEB16 and CGI of Mageb16, identified by searching the TRANSFACⓇ Public 6.0 database (http://www.gene-regulation.com, data not shown). Hence, the hypermethylation of these CGI sequences may interfere with the binding of these proteins with these regions and lead to the silencing of MAGEB16 and Mageb16 gene expression. However, to confirm this hypothesis, more evidence from chromatin immunoprecipitation assays using antibodies to the above trans-acting factors is necessary.
In summary, we identified that the MAGEB16 and Mageb16 genes are specifically expressed in human and mouse testis, respectively, and that the methylation status of the CGIs in their 5'upstream regions is the primary mechanism for the activation of MAGEB16 and Mageb16 gene expression.
MATERIALS AND METHODS
Human samples
Normal human testicular tissues were obtained from two body donors, who died in accidents, from West China Hospital, Sichuan University. Informed consent for the samples was obtained from the relatives of the body donors. The present study was authorized by the Ethical Committee of West China Hospital, Sichuan University, China.
Animals
The C57BL/6Jmice used in the study were obtained from the Laboratory Animal Center of the State Key Laboratory of Biotherapy, Sichuan University. The animals were treated in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals (USA).
Reverse transcription (RT)-PCR and Quantitative (Q) RT-PCR
Normal human tissue cDNA panels (multiple tissue cDNA panels I and II) were purchased from Clontech (Mountain View, USA). Total RNA was isolated from human cells and mouse tissues, cells, embryos, and testes at different stages of development. RT was performed using 1 μg RNA, oligo (dT), and RevertAidTM Reverse Transcriptase (Fermentas,USA). The primers used to amplify MAGEB16 and Mageb16 genes were listed in the Supplementary Table 1. The QRT-PCR analysis was performed with cDNA using the SYBRⓇ Premix Ex TaqTM II kit (Takara, China) plus 10 mM of the corresponding set of sense and antisense primers. Reactions were run on a Bio-Rad MyiQ Cycler using the following parameters: 95℃ for 1min; 95℃ for 10s and 60℃ for 30 s for 40 cycles. Three separate assays were performed in duplicate on each tissue. The CT values from each run were averaged per tissue. The CT method was then applied for data analysis, in which the value of MAGEB16 and Mageb16mRNA expressed in each tissue and cell was normalized with that of 18S rRNA and Actin, respectively. All QRT-PCR data are expressed as the mean ± standard deviation (S.D.), using the value of mouse testis as 1 arbitrary unit=100%.
Cell culture, 5'-aza-CdR treatment and transfection
The cell lines used in this study were grown in Dulbecco’s modified Eagle’s medium (DMEM), supplied with 10% fetal bovine serum. All cells were maintained at 37℃ in an incubator containing 5% CO2.
As previously described (20), cells were seeded at low density in six-well plates and treated with 5'-aza-CdR (Sigma, USA) at different concentrations (0, 0.1, 0.5, 5, or 10 μmol/L). After 72 h, the cells were directly lysed in Trizol (Invitrogen, USA), and total RNA was extracted and analyzed, as described above.
The cells were transfected with various plasmids using Lipofectamine 2000 (Invitrogen).
Genomic DNA isolation and bisulfite sequencing
Genomic DNA from human and mouse tissues and cells was isolated using a Bioteke whole genomic DNA isolation kit, according to the manufacturer’s instruction (Bioteke, China). Bisulfite treatment was carried out using a DNA Methylation-Gold kit (Zymo research, USA).
The primers used to amplify the 5' upstream CGI regions of MAGEB16 and Mageb16 were listed in the Supplementary Table 1. The PCR conditions were 10 minutes at 95℃; 40 cycles of 30 seconds at 95℃, 30 seconds at 55℃ for the human MAGEB16 gene and 60℃ for mouse Mageb16 gene, and 30 seconds at 72℃; with a final 10-minute extension at 72℃. PCR products were separated on a 1.5% agarose gel, excised, and gel- purified. TA cloning was used to insert the PCR products into the pMDⓇ20-T vector (TaKaRa). The insert was sequenced with the T7/M13 universal primers. More than five subclones were picked and sequenced for each DNA sample.
MAGEB16 and Mageb16 gene promoter plasmid construction
Ten pairs of primers (Supplementary Table 1) corresponding to the 5' upstream regions of the human MAGEB16 and mouse Mageb16 genes were designed and used to amplify the putative promoter regions from genomic DNA. All the amplified fragments were cut by the appropriate enzymes and inserted into the pGL3-Basic vector (Promega, USA). All the constructs were sequenced.
In vitro methylation and luciferase assay
The promoter constructs were incubated overnight with three units of the CpG methyltransferase (M.SssI) (New England Biolabs, UK) in the presence of 1 mmol/L S-adenosylmethionine, following the manufacturer’s recommendation. Efficient methylation of the plasmid DNA was confirmed by its resistance to digestion with the methylation-sensitive restriction enzyme HpaII (New England Biolabs). After DNA isolation, 2 μg of the methylated MAGEB16 and Mageb16 gene reporter constructs were transiently co-transfected with pRL-CMV vector (Promega) into HEK293T and B16 cells, respectively, and the luciferase activities were measured by the dual-luciferase reporter assay system (Promega). The luciferase activity was normalized using Renilla luciferase activity.
Acknowledgments
This work was funded by a grant from the National Basic Program of China (Program 973) from the Ministry of Science and Technology of China (2012CB947600) and the Fundamental Research Funds for the Central Universities (2010SCU11025).
References
- 1.Schultz N., Hamra F. K., Garbers D. L. A multitude of genes expressed solely in meiotic or postmeiotic spermatogenic cells offers a myriad of contraceptive targets. Proc. Natl. Acad. Sci. U.S.A. (2003);100:12201–12206. doi: 10.1073/pnas.1635054100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Koslowski M., Sahin U., Huber C., Türeci Ö. The human X chromosome is enriched for germline genes expressed in premeiotic germ cells of both sexes. Hum. Mol. Genet. (2006);15:2392–2399. doi: 10.1093/hmg/ddl163. [DOI] [PubMed] [Google Scholar]
- 3.van der Bruggen P., Traversari C., Chomez P., Lurquin C., De Plaen E., Van den Eynde B., Knuth A., Boon T. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science. (1991);254:1643–1647. doi: 10.1126/science.1840703. [DOI] [PubMed] [Google Scholar]
- 4.Osterlund C., Tohonen V., Forslund K. O., Nordqvist K. Mage-b4, a novel melanoma antigen (MAGE) gene specifically expressed during germ cell differentiation. Cancer Res. (2000);60:1054–1061. [PubMed] [Google Scholar]
- 5.Chomez P., De Backer O., Bertrand M., De Plaen E., Boon T., Lucas S. An overview of the MAGE gene family with the identification of all human members of the family. Cancer Res. (2001);61:5544–5551. [PubMed] [Google Scholar]
- 6.Zhao Q., Caballero O. L., Simpson A. J., Strausberg R. L. Differential evolution of MAGE genes based on expression pattern and selection pressure. PloS One. (2012);7:e48240. doi: 10.1371/journal.pone.0048240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gure A. O., Chua R., Williamson B., Gonen M., Ferrera C. A., Gnjatic S., Ritter G., Simpson A. J., Chen Y. T., Old L. J., Altorki N. K. Cancer-testis genes are coordinately expressed and are markers of poor outcome in non-small cell lung cancer. Clin. Cancer Res. (2005);11:8055–8062. doi: 10.1158/1078-0432.CCR-05-1203. [DOI] [PubMed] [Google Scholar]
- 8.Kim J., Reber H. A., Hines O. J., Kazanjian K. K., Tran A., Ye X., Amersi F. F., Martinez S. R., Dry S. M., Bilchik A. J., Hoon D. S. The clinical significance of MAGEA3 expression in pancreatic cancer. Int. J. Cancer. (2006);118:2269–2275. doi: 10.1002/ijc.21656. [DOI] [PubMed] [Google Scholar]
- 9.De Smet C., Lurquin C., Lethe B., Martelange V., Boon T. DNA methylation is the primary silencing mechanism for a set of germ line- and tumor-specific genes with a CpG-rich promoter. Mol. Cell. Biol. (1999);19:7327–7335. doi: 10.1128/mcb.19.11.7327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Smet C., Loriot A., Boon T. Promoter-dependent mechanism leading to selective hypomethylation within the 5' region of gene MAGE-A1 in tumor cells. Mol. Cell. Biol. (2004);24:4781–4790. doi: 10.1128/MCB.24.11.4781-4790.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pattani K. M., Soudry E., Glazer C. A., Ochs M. F., Wang H., Schussel J., Sun W., Hennessey P., Mydlarz W., Loyo M., Demokan S., Smith I. M., Califano J. A. MAGEB2 is activated by promoter demethylation in head and neck squamous cell carcinoma. PloS One. (2012);7:e45534. doi: 10.1371/journal.pone.0045534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhang J., Yu J., Gu J., Gao B. M., Zhao Y. J., Wang P., Zhang H. Y., De Zhu J. A novel protein-DNA interaction involved with the CpG dinucleotide at -30 upstream is linked to the DNA methylation mediated transcription silencing of the MAGE-A1 gene. Cell Res. (2004);14:283–294. doi: 10.1038/sj.cr.7290229. [DOI] [PubMed] [Google Scholar]
- 13.Bai G., Liu Y., Zhang H., Su D., Tao D., Yang Y., Ma Y., Zhang S. Promoter demethylation mediates the expression of ZNF645, a novel cancer/testis gene. BMB Rep. (2010);6:400–406. doi: 10.5483/BMBRep.2010.43.6.400. [DOI] [PubMed] [Google Scholar]
- 14.Dong X., Yang X., Wang Y., Chen W. Zinc-finger protein ZNF165 is a novel cancer-testis antigen capable of eliciting antibody response in hepatocellular carcinoma patients. Br. J. Cancer. (2004);91:1566–1570. doi: 10.1038/sj.bjc.6602138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gaspar J. A., Doss M. X., Winkler J., Wagh V., Hescheler J., Kolde R., Vilo J., Schulz H., Sachinidis A. Gene expression signatures defining fundamental biological processes in pluripotent, early, and late differentiated embryonic stem cells. Stem Cells Dev. (2012);21:2471–2484. doi: 10.1089/scd.2011.0637. [DOI] [PubMed] [Google Scholar]
- 16.Sasaki H., Matsui Y. Epigenetic events in mammalian germ-cell development: reprogramming and beyond. Nat. Rev. Genet. (2008);9:129–140. doi: 10.1038/nrg2295. [DOI] [PubMed] [Google Scholar]
- 17.del Mazo J., Prantera G., Torres M., Ferraro M. DNA methylation changes during mouse spermatogenesis. Chromosome Res. (1994);2:147–152. doi: 10.1007/BF01553493. [DOI] [PubMed] [Google Scholar]
- 18.Zhu W. G., Srinivasan K., Dai Z., Duan W., Druhan L. J., Ding H., Yee L., Villalona-Calero M. A., Plass C., Otterson G. A. Methylation of adjacent CpG sites affects Sp1/Sp3 binding and activity in the p21(Cip1) promoter. Mol. Cell. Biol. (2003);23:4056–4065. doi: 10.1128/MCB.23.12.4056-4065.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Iguchi-Ariga S., Schaffner W. CpG methylation of the cAMP-responsive enhancer/promoter sequence TGACGTCA abolishes specific factor binding as well as transcriptional activation. Genes Dev. (1989);3:612–619. doi: 10.1101/gad.3.5.612. [DOI] [PubMed] [Google Scholar]
- 20.Maraskovsky E., Jager E., Seliger B., Maio M. 5'-aza-2'-deoxycytidine induced expression of functional cancer testis antigens in human renal cell carcinoma: immunotherapeutic implications. Clin. Cancer. Res. (2002);8:2690–2695. [PubMed] [Google Scholar]


