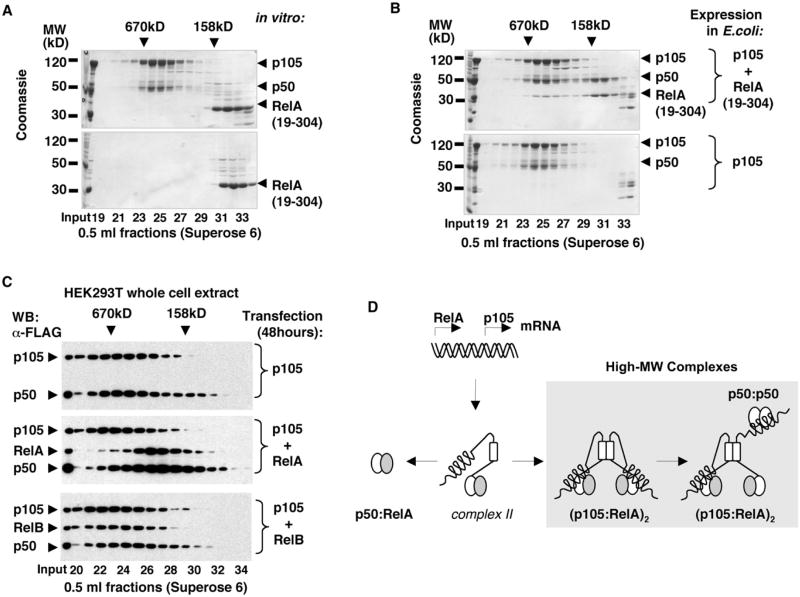
Figure 7. Biogenesis of p50:RelA Heterodimers and RelA Complexes with p105.
(A) Recombinant p105:p50 complexes were incubated with RelA RHD homodimers, RelA(19–304), and the resulting complexes were analyzed by gel filtration chromatography (GF) followed by SDS-PAGE and Coomassie staining (top) and compared to the similarly treated isolated RelA(19–304) (bottom).
(B) His-tagged p105 was coexpressed with untagged RelA(19–304) in E. coli, and the resulting complexes were isolated by Ni affinity chromatography followed by GF (top) and compared to the recombinant p105:p50 complexes (bottom).
(C) FLAG-tagged p105 was expressed in HEK293T cells alone or coexpressed with FLAG-tagged RelA or RelB. At 48 hr after transfection, the whole-cell extracts were analyzed by GF followed by western blotting (WB).
(D) A model for the biogenesis of p50:RelA heterodimers and the high-MW p105:p50:RelA complexes.