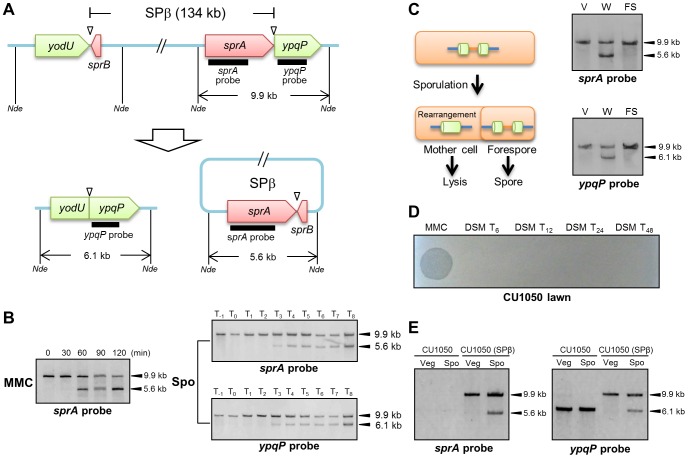
Figure 1. DNA rearrangement at the spsM locus.
(A) Diagram showing SPβ excision in Bacillus subtilis 168. The thick lines indicate the location of the digoxigenin (DIG)-labeled probes used for Southern blotting. Nde indicates NdeI sites. Triangles point to the attachment sites for SPβ. (B) SPβ excision upon mitomycin C (MMC) treatment and during sporulation. Left panel shows induction of SPβ excision by MMC treatment. B. subtilis 168 cells were grown in LB medium. Vegetative cells in the early log phase (OD600 = 0.25) were treated with 0.5 µg/ml MMC. Time 0 indicates the time point immediately after MMC addition. Right panels show SPβ excision (top panel) and spsM reconstitution (bottom panel) during sporulation. B. subtilis 168 cells were grown in DSM, and samples were taken at the indicated times (in h) after the onset of sporulation (T0). The DNA samples were digested with NdeI and subjected to Southern blotting. (C) Mother cell-specific SPβ excision. Chromosomal DNA from the vegetative cells (V) at T−1, whole sporangia (W) at T8, and the forespores (FS) at T8 were isolated, digested with NdeI, and subjected to Southern blotting. (D) Lytic activity of SPβ phages. SPβ phage lysate, which was prepared by treating the B. subtilis 168 vegetative cells with MMC, was spotted on the plate (MMC). The DSM culture of B. subtilis 168 at T6, T12, T24, and T48 was centrifuged and the supernatant was filtrated with 0.44 µm Millex filter (Millipore). The filtrate was spotted on the lawn of a SPβ sensitive strain CU1050 (DSM T24 and DSM T48). (E) Horizontal transfer of spsM rearrangement system. A new SPβ-lysogen, CU1050 (SPβ) was obtained by infecting CU1050 cells with the SPβ phage lysate. The CU1050 and CU1050 (SPβ) cells were induced to sporulate on DSM-agar plates at 37°C for 3 (Vegetative cells, Veg) and 12 hours (Sporulating cells, Spo). Chromosomal DNA of the CU1050 and CU1050 (SPβ) cells was subjected to Southern blotting.