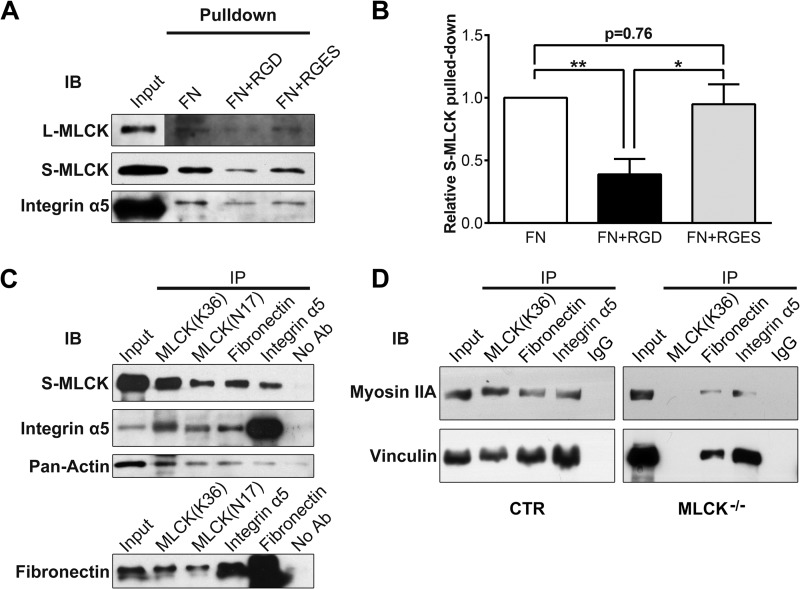
FIGURE 7.
MLCK participates in the formation of fibronectin-integrin-cytoskeleton transmembrane protein complexes. A, with a fibronectin pull-down assay, MLCK isoforms and integrin α5 were detected in the protein complexes binding FN. In the “input” signal panel, total protein of SMC lysate used in the pull-down assay was loaded. In other signal panels, fibronectin pulled-down proteins were probed. RGD peptide was used to block the FN-integrin interaction, and RGES peptide was used as a negative control. B, quantitation of FN pulled-down S-MLCK (n = 5). C and D, co-immunoprecipitation was performed, and the immunoprecipitates were then subjected to immunoblotting (IB) using antibodies against MLCK, integrin α5, FN, pan-actin, myosin IIA, and vinculin. Whole-SMC lysate (Input) was used as a positive control. Protein G-Sepharose without antibody (No Ab) or with rabbit IgG (IgG) was used as a negative control.