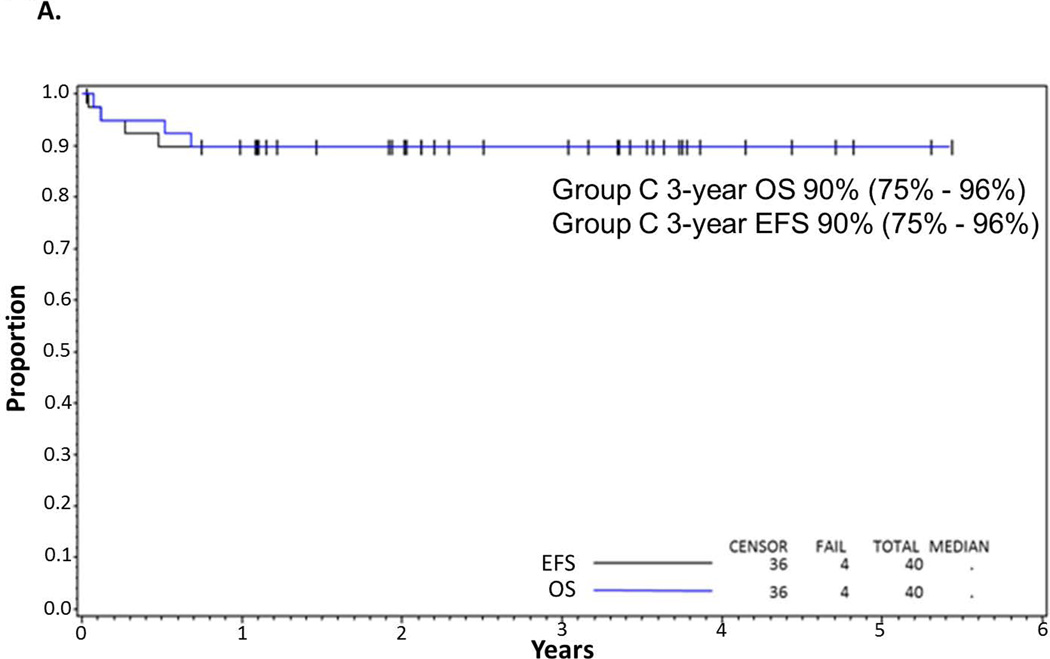
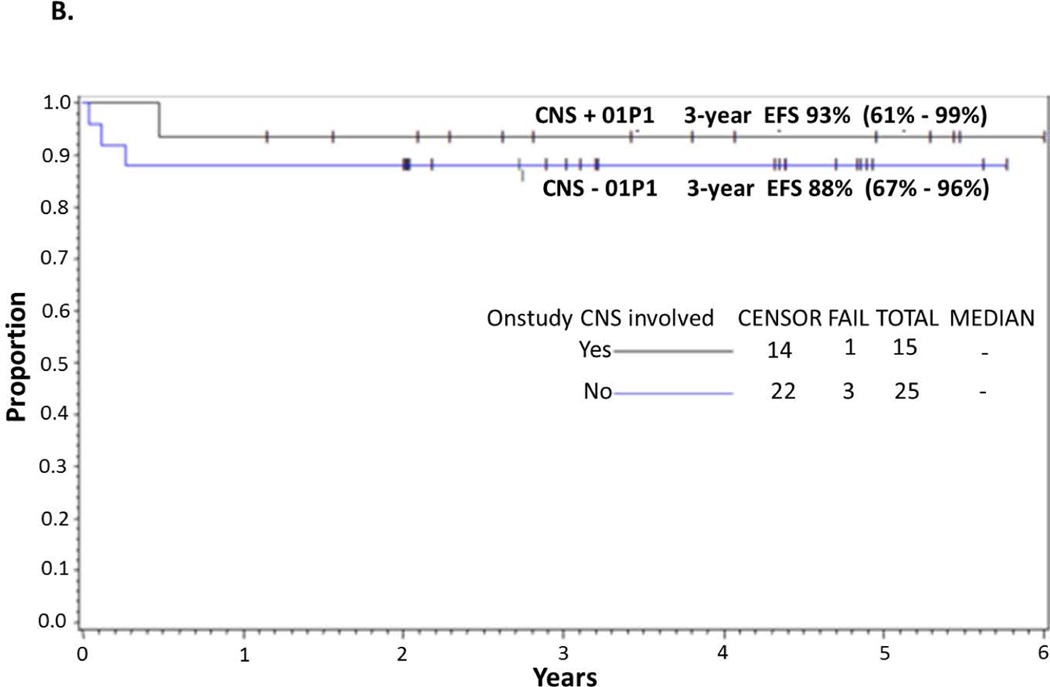
Figure 2.
(A) EFS and OS in all patients. Probability of EFS and OS in children and adolescents with BM and/or CNS disease with de novo mature B-cell non-Hodgkin lymphoma (B-NHL) treated with Rituximab and FAB Group C1 Chemotherapy Pilot on COG-ANHL01P1 as determined by Kaplan-Meier method. (B) EFS in CNS-positive and CNS-negative patients. Probability of EFS in children and adolescents with BM and/or CNS-positive and -negative disease with advanced de-novo mature B-NHL treated with Rituximab and FAB Group C1 chemotherapy stratified by CNS+ vs. CNS− on COG-ANHL01P1 as determined by Kaplan-Meier method.
EFS, event-free survival; OS, overall survival; BM, bone marrow; CNS, central nervous system; 01P1, ANHL01P1 protocol.