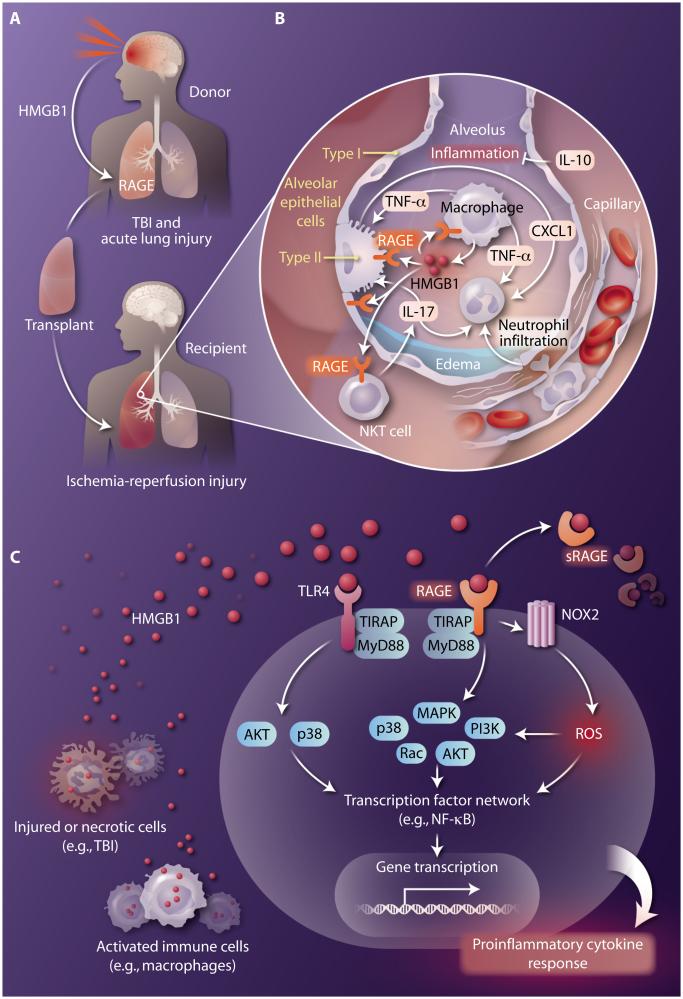
Fig. 1. HMGB1, TBI, and lung transplantation.
HMGB1 released in response to traumatic brain injury (TBI) or ischemia-reperfusion injury in the donor mediates lung dysfunction after transplantation. (A) TBI in a potential lung donor disrupts the blood-brain barrier, releasing HMGB1, which leads to acute lung injury and the binding of HMGB1 to RAGE. Transplantation of lungs from donors with TBI results in an increase in ischemia-reperfusion injury in the donor lungs in recipients. (B) Lung ischemia-reperfusion injury after transplant is mediated, in part, by the HMGB1/RAGE axis and can result in primary graft dysfunction involving edema formation and impaired gas exchange. HMGB1 (produced by alveolar macrophages) augments production of the proinflammatory cytokine IL-17 by NKT cells and CXCL1 production by type 2 alveolar epithelial cells, resulting in neutrophil infiltration and activation, which are key events in inflammation and tissue injury. Enhanced production of the anti-inflammatory cytokine IL-10 (from various sources) can deter innate immune cell activation and inflammation. (C) Common HMGB1-mediated signaling pathways in a target cell. HMGB1, released from necrotic cells or secreted by activated immune cells, signals by binding to RAGE and/or TLR4 (with associated regulators such as TIRAP and MyD88) to stimulate various signaling cascades that activate proinflammatory transcription factors such as NF-κB. The enzymatic cleavage of membrane-bound RAGE releases soluble RAGE (sRAGE), which also can bind to HMGB1 to serve as a decoy receptor. HMGB1-RAGE signaling can also induce production of reactive oxygen species (ROS) through activation of NADPH oxidase (NOX2), which also leads to activation of NF-κB. The end result is rapid transcription of genes encoding proinflammatory cytokines to elicit an inflammatory, injurious response.