Abstract
AIM: To systematically review the surgical outcomes of totally laparoscopic gastrectomy (TLG) vs open gastrectomy (OG) for gastric cancer.
METHODS: A systematic search of PubMed, Embase, Cochrane Library, and Web of Science was conducted. All original studies comparing TLG with OG were included for critical appraisal. Data synthesis and statistical analysis were carried out using RevMan 5.1 software.
RESULTS: One RCT and 13 observational studies involving 1532 patients were included (721 TLG and 811 OG). TLG was associated with longer operation time [weighted mean difference (WMD) = 58.04 min, 95%CI: 37.77-78.32, P < 0.001], less blood loss [WMD = -167.57 min, 95%CI: -208.79-(-126.34), P < 0.001], shorter hospital stay [WMD = -3.75 d, 95%CI: -4.88-(-2.63), P < 0.001] and fewer postoperative complications (RR = 0.71, 95%CI: 0.58-0.86, P < 0.001). The number of harvested lymph nodes, surgical margin, mortality and cancer recurrence rate were similar between the two groups.
CONCLUSION: TLG may be a technically safe, feasible and favorable approach in terms of better cosmesis, less blood loss and faster recovery compared with OG.
Keywords: Gastric cancer, Gastrectomy, Intracorporeal anastomosis, Laparoscopy, Meta-analysis, Survival
Core tip: Laparoscopy-assisted gastrectomy for gastric cancer has rapidly become popular in the past decades due to its minimally invasive advantages over open gastrectomy (OG). However, totally laparoscopic gastrectomy (TLG) remains controversial in terms of safety and technical issues. This study evaluated the safety and efficacy of TLG compared with OG for gastric cancer by performing a systematic review and meta-analysis of the literature. The existing research shows that TLG is safe and feasible, and can achieve similar lymph node dissection effects to those of OG, and is characterized by advantages such as less pain, fewer postoperative complications, and rapid recovery, and is expected to achieve the same effect in oncological treatment as OG.
INTRODUCTION
Laparoscopy-assisted gastrectomy (LAG) for gastric cancer was first reported in 1994[1], and has undergone rapid development and gained popularity in the past few decades. Laparoscopic surgery has multiple benefits compared with open gastrectomy (OG) such as minimal invasiveness, the possibility of practical maneuvers based on anatomic understanding through a good visual field and magnification, earlier patient recovery after surgery and better postoperative quality of life[2-5]. During LAG, lymph node dissection is performed laparoscopically. However, a mini-laparotomy is performed in the epigastrium, through which the anastomosis is performed under direct vision. Totally laparoscopic gastrectomy (TLG) preserves the integrity of the abdominal wall, which is considered to be incisionless, except for the trocar wounds[6], and is a laparoscopic approach for intracorporeal anastomosis without auxiliary incision and contact with the tumor. TLG represents the evolution of LAG. However, there are some technical difficulties when performing intracorporeal anastomosis, thus LAG is still a common approach in laparoscopic surgery[7]. The safety and efficacy of LAG has been demonstrated in large retrospective studies and randomized controlled trials (RCTs)[2,3,8-11]. In addition, several meta-analyses and systematic reviews have been published on LAG[12-15]. Similar studies have not been conducted to assess the potential benefits and disadvantages of TLG. The aim of this study was to compare TLG with OG with respect to surgical outcomes, morbidity, mortality and functional recovery. Long-term outcomes after TLG and OG in patients with any stage of gastric cancer were also evaluated in a systematic review of the literature and meta-analyses were performed.
MATERIALS AND METHODS
Search strategy
Systematic searches of PubMed, Embase, Cochrane Library, and Web of Science were performed to identify articles published up to February 2014 that compared TLG and OG. The search terms “gastric adenocarcinoma”, “gastric cancer”, “laparoscopic”, “laparoscopy”, “gastrectomy”, “completely”, “totally”, “intracorporeal” and “endocorporeal” were utilized. The links of each search result and all references in the original articles identified were reviewed to identify additional literature that was not indexed. Only studies written in English were considered for inclusion.
Eligibility criteria
Studies meeting the following criteria were included: comparative, peer-reviewed studies of TLG vs OG in patients with gastric cancer for which the full text of the article was available. If two studies from the same group were identified, the most recent study or that including more subjects was selected unless the reports were from different time periods. The papers containing any of the following were excluded: (1) laparoscopic-assisted, hand-assisted, or robot-assisted gastrectomy; (2) non-gastric carcinoma cases; (3) tumors not in the stomach such as the esophagus; and (4) studies in which < 2 of the indices under study were reported, or it was difficult to calculate these from the results.
Data extraction and quality assessment
Two authors independently extracted the data using a unified datasheet, and controversial issues were decided by discussion. The extracted data included: author, study period, geographical region, number of patients, operation time, blood loss, number of retrieved lymph nodes, proximal and distal margin distance, time to flatus, time to oral intake, length of hospital stay, morbidity, mortality, and long-term outcomes. Postoperative complications were classified as medical (cardiovascular, respiratory, or metabolic events; nonsurgical infections; deep vein thrombosis; and pulmonary embolism) or surgical (any anastomotic leakage or fistula, any complication that required reoperation, intra-abdominal collections, wound complications, bleeding events, pancreatitis, ileus, delayed gastric emptying, and anastomotic stricture). This classification system was based on the Memorial Sloan-Kettering Cancer Center complication reporting system[16]. If the study provided medians and ranges instead of mean ± SD, we estimated the mean ± SD as described by Hozo et al[17]. RCTs were evaluated by the Jadad composite scale. High quality trials scored more than 2 out of a maximum possible score of 5. The quality of the non-randomized studies was assessed using the Newcastle-Ottawa Quality Assessment Scale (NOS). This scale varies from zero to 9 stars: studies with a score equal to or higher than 6 were considered methodologically sound.
Statistical analysis
Continuous variables were assessed using weighted mean difference (WMD), and dichotomous variables were analyzed using the RR. Statistical heterogeneity, which indicated between-study variance, was evaluated according to the Higgins I2 statistic[18]. To account for clinical heterogeneity, which refers to diversity relevant to clinical situations, we used the random-effects model based on DerSimonian and Laird’s method. Subgroup analysis of intraoperative outcomes, such as operation time, blood loss, and number of retrieved lymph nodes, was conducted for the number of TLG cases performed (40 cases were used as a cut-point), as the learning curve may have an impact on the operative outcomes. Potential publication bias was determined by conducting an informal visual inspection of funnel plots based on the complications. Data analyses were performed using Review Manager Version 5.1 (RevMan 5.1) software downloaded from the Cochrane Library. P < 0.05 was considered statistically significant.
RESULTS
Studies selected
The initial search strategy retrieved 1968 publications in English. After the titles and abstracts were reviewed, papers without a comparison of TLG and OG were excluded, which left 20 comparative studies, six[19-24] of which did not meet the inclusion criteria and were excluded. This left a total of one RCT and 13 observational studies[25-38], all of which were accessible in full-text format. A flow chart of the search strategies, which includes the reasons for excluding studies, is illustrated in Figure 1.
Figure 1.
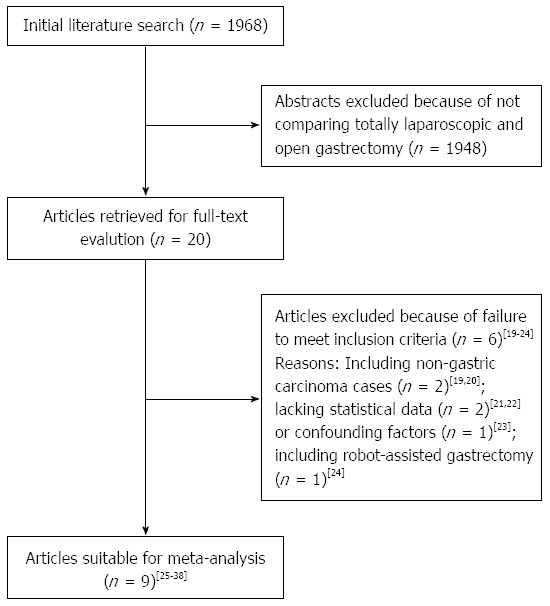
Flow chart of literature search strategies. Spelling of search in figure incorrect.
Study characteristics and quality
A total of 1532 patients were included in the analysis with 721 undergoing TLG (47.1%) and 811 undergoing OG (52.9%). These patients represented international studies and included data from 9 different countries or regions (5 from Italy, 1 from Belgium, 1 from France, 1 from the United States, 1 from Chile, 2 from South Korea, 1 from Japan, 1 from Taiwan and 1 from Hong Kong). In general, the quality of the included studies was satisfactory. The RCT received a Jadad score of 3. According to the NOS, three of the 13 observational studies received 7 stars, three articles received 8 stars, and the remaining seven received 9 stars. The characteristics and methodological quality assessment scores of the included studies are summarized in Table 1. The outcomes reported by the included studies are shown in Table 2.
Table 1.
Summary of studies included in the meta-analysis
| Ref. | Region | Study design | Year | Study period |
Sample size |
Level of lymphad-enectomy | Surgical extension | Reconstruction | Conversion |
Mortality |
Matched factors1 | Quality scores | ||
| TLG | OG | TLG | OG | |||||||||||
| Huscher et al[25] | Italy | RCT | 2005 | 1992-1996 | 30 | 29 | D1, D2 | D | B-II, R-Y | NR | 3.3% | 6.7% | - | 32 |
| Pugliese et al[26] | Italy | OCS (R) | 2007 | 2000-2005 | 48 | 99 | D2 | S, T | R-Y | 2.1% | 2.1% | 3.0% | - | 7 |
| Song et al[27] | South Korea | OCS (P) | 2008 | 2005-2006 | 20 | 20 | D1 + β, D2 | D | B-I, R-Y | 0 | 0 | 0 | - | 8 |
| Lee et al[28] | Taiwan | OCS (P) | 2008 | 1998-2005 | 34 | 34 | D2 | S | B-II | 2.9% | 0 | 0 | 1245 | 9 |
| Topal et al[29] | Belgium | OCS (R) | 2008 | 2003-2006 | 38 | 22 | D2 | T | R-Y | NR | 2.6% | 4.5% | - | 8 |
| Strong et al[30] | United States | OCS (P) | 2009 | 2005-2008 | 30 | 30 | D2 | S | B-II, R-Y | NR | 0 | 0 | 1245 | 9 |
| Wong et al[31] | Hong Kong | OCS (R) | 2009 | 2001-2006 | 18 | 41 | D1 + α/β, D2 | D | B-II, R-Y | 5.0% | 0 | 2.4% | - | 7 |
| Chouillard et al[32] | France | OCS (P) | 2010 | 2001-2007 | 51 | 79 | D2 | D, T | B-II, R-Y | 6.0% | 0 | 2.5% | - | 9 |
| Scatizzi et al[33] | Italy | OCS (P) | 2011 | 2006-2009 | 30 | 30 | D2 | D | R-Y | 6.7% | 0 | 0 | 12345 | 9 |
| Moisan et al[34] | Chile | OCS (R) | 2012 | 2005-2010 | 31 | 31 | D1 + α/β, D2 | S, T | B-II, R-Y | 3.2% | 0 | 0 | 1245 | 9 |
| Cianchi et al[35] | Italy | OCS (P) | 2012 | 2008-2012 | 41 | 41 | D1 + α/β, D2 | S, T | B-II, R-Y | 0 | 2.4% | 4.9% | 1245 | 8 |
| Siani et al[36] | Italy | OCS (R) | 2012 | 2003-2009 | 25 | 25 | D1 + α/β, D2 | T | R-Y | 0 | 0 | 0 | 145 | 9 |
| Shinohara et al[37] | Japan | OCS (P) | 2013 | 1998-2008 | 186 | 123 | D2 | D, P, T | B-I, R-Y | 2.2% | 1.1% | 0 | 12345 | 9 |
| Kim et al[38] | South Korea | OCS (P) | 2013 | 2011 | 139 | 207 | D2 | T | R-Y | 0 | 0 | 0 | - | 7 |
Jadad scores;
Factors matched between groups: 1: Age; 2: Sex; 3: Body mass index; 4: Stage; 5: Type of gastrectomy. RCT: Randomized controlled trial; OCS: Observational clinical study; P: Prospectively collected data; R: Retrospectively collected data; TLG: Totally laparoscopic gastrectomy; OG: Open gastrectomy; D: Distal gastrectomy; P: Proximal gastrectomy; T: Total gastrectomy; S: Subtotal gastrectomy; B-I: Billroth-I; B-II: Billroth-II; R-Y: Roux-en-Y; NR: Not reported.
Table 2.
Outcomes of included studies
| Ref. | Operation time | Blood loss | Transfusion | RLN | Proximal margin | Distal margin | Analgesic use | Ambulation | Flatus | Oral intake | Hospital stay | Morbidity | Mortality | Survival |
| Huscher et al[25] | Y | Y | Y | Y | Y | Y | Y | Y | ||||||
| Pugliese et al[26] | Y | Y | Y | Y1 | Y | Y | Y | Y | Y | Y | ||||
| Song et al[27] | Y | Y | Y | Y | Y | Y2 | Y | Y | Y | Y | Y | |||
| Lee et al[28] | Y | Y | Y | Y2 | Y | Y | Y | Y | ||||||
| Topal et al[29] | Y | Y | Y | Y | ||||||||||
| Strong et al[30] | Y | Y | Y | Y1 | Y | Y | Y | Y | ||||||
| Wong et al[31] | Y | Y | Y | Y | Y | Y | Y | |||||||
| Chouillard et al[32] | Y | Y | Y | Y | Y | Y | Y | Y | ||||||
| Scatizzi et al[33] | Y | Y | Y1 | Y | Y | Y | Y | Y | Y | Y | ||||
| Moisan et al[34] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | ||||
| Cianchi et al[35] | Y | Y | Y | Y | Y | Y | Y | Y | ||||||
| Siani et al[36] | Y | Y | Y | Y | Y | Y | Y | Y | ||||||
| Shinohara et al[37] | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y | ||||
| Kim et al[38] | Y | Y | Y | Y | Y2 | Y | Y | Y | Y | Y |
Reported days of analgesic use;
Reported days and amount of analgesic use. Y: Yes; RLN: Retrieved lymph nodes.
Intraoperative effects
All intraoperative outcomes are summarized in Table 3. Longer duration of operation was observed in the TLG group compared with the OG group (P < 0.001) (Figure 2A). Blood loss during surgery was decreased during the laparoscopic procedure (P < 0.001) (Figure 2B). The rate of transfusions was lower for TLG with a marginal difference between the groups (P = 0.09). No statistical difference was found between the two groups in terms of the number of harvested lymph nodes (P = 0.59) (Figure 2C). We also analyzed the retrieval of lymph nodes between the TLG and OG groups using the modified D2 lymphadenectomy, and no difference was noted (WMD = -1.33, 95%CI: -3.92-1.26, P = 0.31). The length of the proximal resection margin was similar in both groups (P = 1.00), as was the length of the distal resection margin (P = 0.28).
Table 3.
Pooled short-term outcomes
| Outcomes | No. of studies |
Sample size |
Heterogeneity | Overall effect size | 95%CI of overall effect | P value | |
| TLG | OG | (P value, I2) | |||||
| Operation time (min) | 14 | 721 | 811 | < 0.001, 97% | WMD = 58.04 | 37.77-78.32 | < 0.001 |
| Blood loss (mL) | 12 | 552 | 574 | < 0.001, 87% | WMD = -167.57 | -208.79-(-126.34) | < 0.001 |
| Transfusion | 3 | 268 | 233 | 0.11, 54% | RR = 0.49 | 0.21-1.11 | 0.09 |
| Retrieved lymph nodes | 13 | 683 | 789 | 0.04, 46% | WMD = -0.48 | -2.21-1.26 | 0.59 |
| Proximal margin (cm) | 2 | 159 | 227 | 0.03, 80% | WMD = 0.00 | -1.47-1.46 | 1.00 |
| Distal margin (cm) | 3 | 190 | 258 | 0.03, 70% | WMD = 0.94 | -0.76-(2.64) | 0.28 |
| Analgesics given (d) | 3 | 108 | 159 | 0.33, 11% | WMD = -1.79 | -2.37-(-1.21) | < 0.001 |
| Time to ambulation (d) | 3 | 264 | 252 | < 0.001, 93% | WMD = -0.91 | -1.65-(-0.16) | 0.02 |
| Time to first flatus (d) | 7 | 337 | 456 | < 0.001, 98% | WMD = -1.97 | -3.18-(-0.77) | 0.001 |
| Time to oral intake (d) | 8 | 525 | 580 | < 0.001, 96% | WMD = -2.39 | -3.34-(-1.45) | < 0.001 |
| Hospital stay (d) | 13 | 683 | 789 | < 0.001, 83% | WMD = -3.75 | -4.88-(-2.63) | < 0.001 |
| Overall complications | 14 | 721 | 811 | 0.74, 0% | RR = 0.71 | 0.58-0.86 | < 0.001 |
| Surgical complications | 12 | 635 | 690 | 0.76, 0% | RR = 0.75 | 0.57-0.98 | 0.03 |
| Medical complications | 11 | 615 | 670 | 0.97, 5% | RR = 0.57 | 0.38-0.86 | 0.008 |
| Mortality | 7 | 412 | 434 | 0.96, 0% | RR = 0.65 | 0.24-1.76 | 0.40 |
WMD: Weighted mean difference; RR: Risk ratio; TLG: Totally laparoscopic gastrectomy; OG: Open gastrectomy.
Figure 2.
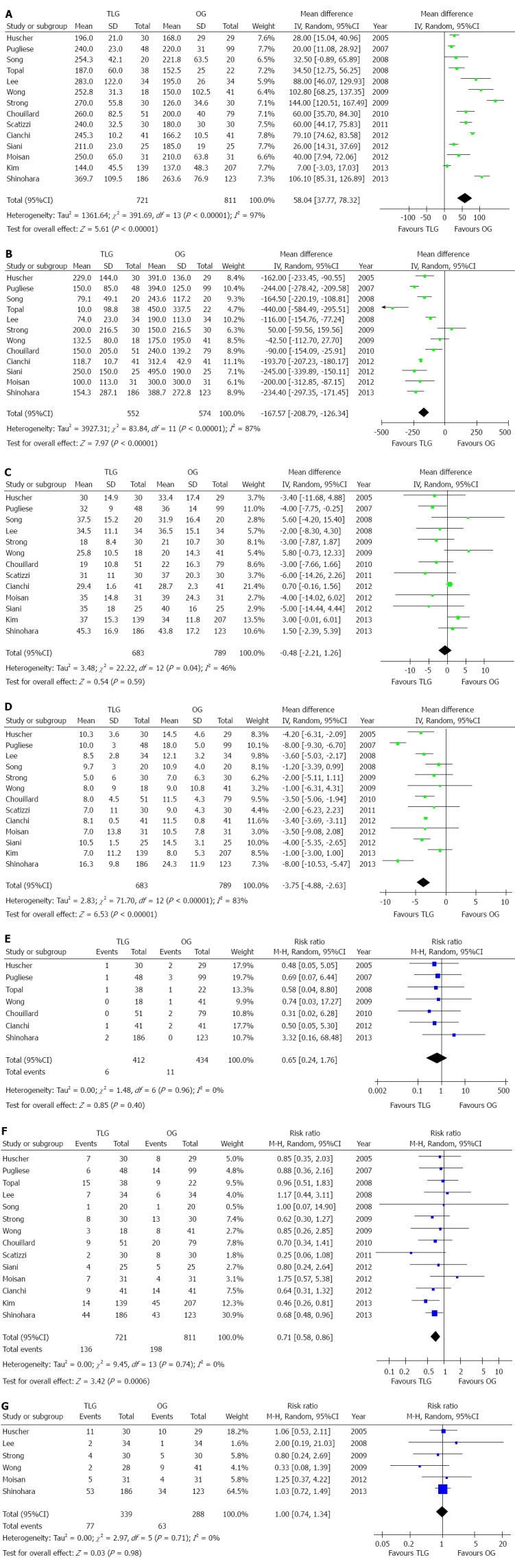
Meta-analysis of the pooled data. A: Operation time; B: Intraoperative blood loss; C: Retrieved lymph nodes; D: Postoperative hospital stay; E: Mortality; F: Overall complications; G: Recurrence.
Subgroup analysis of the learning curve
The overall effects of operation time, blood loss and retrieved lymph nodes were unchanged in the subgroups, although performing > 40 TLG cases resulted in a moderate reduction in operation time and blood loss. The outcomes of subgroup analyses are summarized in Table 4.
Table 4.
Subgroup analysis of learning curve using a cut-point of 40 totally laparoscopic gastrectomy cases
| Outcomes | No. of studies |
Sample size |
Heterogeneity | Overall effect size | 95%CI of overall effect | P value | |
| TLG | OG | (P value, I2) | |||||
| Operation time (min) | |||||||
| < 40 TLG cases | 9 | 256 | 262 | < 0.001, 92% | WMD = 60.54 | 35.64-85.45 | < 0.001 |
| > 40 TLG cases | 5 | 465 | 549 | < 0.001, 99% | WMD = 53.91 | 16.74-91.08 | 0.004 |
| Blood loss (mL) | |||||||
| < 40 TLG cases | 8 | 226 | 232 | < 0.001, 84% | WMD = -155.04 | -133.22 | < 0.001 |
| > 40 TLG cases | 4 | 326 | 342 | < 0.001, 85% | WMD = -194.81 | -95.98 | < 0.001 |
| Retrieved lymph nodes | |||||||
| < 40 TLG cases | 8 | 218 | 240 | 0.23, 25% | WMD = -1.41 | -6.11 | 0.37 |
| > 40 TLG cases | 5 | 465 | 549 | 0.06, 63% | WMD = 0.03 | -4.27 | 0.98 |
TLG: Totally laparoscopic gastrectomy; OG: Open gastrectomy; WMD: Weighted mean difference.
Postoperative clinical course
All postoperative outcomes are summarized in Table 3. Postoperative pain was evaluated by the number of days of analgesic use. Patients who underwent TLG received fewer analgesics (P < 0.001). The outcomes also favored TLG for first flatus day (P = 0.001) and first oral intake (P < 0.001), which indicated a quicker recovery of bowel function. Moreover, postoperative hospital stay was 3.75 d shorter for TLG patients (P < 0.001) (Figure 2D).
Mortality was described in seven studies, and there was no significant difference in postoperative mortality between the groups (P = 0.40) (Figure 2E). The rate of overall postoperative complications was lower in the TLG group (P < 0.001) (Figure 2F). Visual inspection of the funnel plot revealed symmetry, indicating no serious publication bias (Figure 3). After further analysis, surgical complications were also lower in the TLG group (P = 0.03). Wound problems such as infection and dehiscence occurred in 1.7% of TLG patients compared with 6.3% of OG patients (P < 0.001)[25,28,30,31,36-38]. Other surgical complications such as anastomotic leakage, intra-abdominal collections, bleeding, and anastomotic stricture were similar between the two groups (P > 0.05). In addition, TLG was associated with a significant reduction in medical complications (P = 0.008) with a possible contribution from pulmonary complications (TLG = 2.8%, OG = 4.8%, P = 0.003)[25-29,31,33-36,38]. The specific postoperative complications included in the studies are summarized in Table 5.
Figure 3.
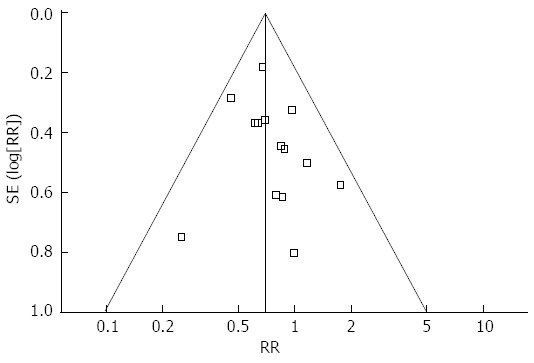
Funnel plots of the overall postoperative complications.
Table 5.
Systematic review of postoperative complications and reoperation n (%)
| Ref. | Group | n | Total events | Leakage | Stenosis | Pulmonary | Wound | Pancreatitis | Obstruction | Cardiovascular | Bleeding | Abscess | Other | Reoperation |
| Huscher et al[25] | TLG | 30 | 7 | 4 | 2 | 1 | NR | |||||||
| OG | 29 | 8 | 1 | 5 | 2 | NR | ||||||||
| Pugliese et al[26] | TLG | 48 | 61 | 2 | 3 | NR | ||||||||
| OG | 99 | 141 | 3 | NR | ||||||||||
| Song et al[27] | TLG | 20 | 1 | 1 | 0 | |||||||||
| OG | 20 | 1 | 1 | 0 | ||||||||||
| Lee et al[28] | TLG | 34 | 7 | 1 | 1 | 3 | 1 | 1 | 2 | |||||
| OG | 34 | 6 | 2 | 1 | 2 | 1 | 0 | |||||||
| Topal et al[29] | TLG | 38 | 151 | 2 | 1 | 1 | 3 | 6 | ||||||
| OG | 22 | 91 | 0 | |||||||||||
| Strong et al[30] | TLG | 30 | 8 | 1 | 1 | 1 | 3 | 1 | 1 | NR | ||||
| OG | 30 | 13 | 1 | 1 | 3 | 4 | 1 | 2 | 1 | NR | ||||
| Wong et al[31] | TLG | 18 | 3 | 1 | 1 | 1 | NR | |||||||
| OG | 41 | 8 | 3 | 2 | 1 | 1 | 1 | NR | ||||||
| Chouillard et al[32] | TLG | 51 | 91 | 2 | ||||||||||
| OG | 79 | 201 | 4 | |||||||||||
| Scatizzi et al[33] | TLG | 30 | 2 | 2 | 1 | |||||||||
| OG | 30 | 8 | 1 | 3 | 1 | 1 | 2 | 1 | ||||||
| Moisan et al[34] | TLG | 31 | 7 | 4 | 1 | 1 | 1 | 4 | ||||||
| OG | 31 | 4 | 2 | 2 | 4 | |||||||||
| Cianchi et al[35] | TLG | 41 | 9 | 2 | 1 | 1 | 2 | 1 | 1 | 1 | 3 | |||
| OG | 41 | 14 | 4 | 5 | 1 | 2 | 2 | 2 | ||||||
| Siani et al[36] | TLG | 25 | 4 | 1 | 1 | 2 | NR | |||||||
| OG | 25 | 5 | 2 | 2 | 1 | NR | ||||||||
| Shinohara et al[37] | TLG | 186 | 441 | 5 | 4 | 2 | 12 | 9 | 8 | |||||
| OG | 123 | 431 | 5 | 5 | 10 | 8 | 9 | 2 | ||||||
| Kim et al[38] | TLG | 139 | 14 | 1 | 2 | 2 | 1 | 3 | 3 | 2 | 5 | |||
| OG | 207 | 45 | 1 | 4 | 9 | 12 | 2 | 2 | 11 | 4 | NR | |||
| Total (%) | TLG | 721 | 136 | 24 (3.4) | 7 (1.9) | 13 (2.8) | 8 (1.7) | 17 (4.5) | 12 (4.0) | 2 (1.6) | 8 (2.3) | 11 (2.7) | 6 (2.0) | |
| OG | 811 | 198 | 27 (3.4) | 9 (2.4) | 28 (4.8) | 31 (6.3) | 9 (3.0) | 9 (2.4) | 5 (4.0) | 4 (0.9) | 23 (5.3) | 11 (3.0) | ||
| P value | 0.99 | 0.63 | 0.003 | < 0.001 | 0.32 | 0.23 | 0.45 | 0.11 | 0.06 | 0.42 |
1Some complications were not specified. NR: Not reported; TLG: Totally laparoscopic gastrectomy; OG: Open gastrectomy.
Recurrence and long-term survival rate
Six studies reported cancer recurrence[25,28,30,31,34,37]. The recurrence risk in the TLG group was 22.7% (77/339) and was 21.9% (63/288) in the OG group, however, the difference was not significant (RR = 1.00, 95%CI: 0.74-1.34, P = 0.98) (Figure 2G). Seven studies reported postoperative survival rate[25,31-34,36,37], all of which did not find a significant difference between the two groups. Although Strong et al[30] did not report specific survival rate, they also found no significant difference in the survival rate between the two groups after 36 mo of follow-up (P > 0.05). The systematic review of long-term survival outcomes is summarized in Table 6.
Table 6.
Systematic review of long-term survival
| Ref. | Group | Follow-up (mo) | Recurrence | Survival rate (%) |
| Huscher et al[25] | TLG | 60 (2-88) | 11 | 5-yr DFS: 57.3, 5-yr OS: 58.9 |
| OG | 55 (7-90) | 10 | 5-yr DFS: 54.8, 5-yr OS: 55.7 | |
| Lee et al[28] | TLG | NR1 | 2 | NR |
| OG | NR1 | 1 | NR | |
| Song et al[27] | TLG | 11 | 4 | NR2 |
| OG | 13.8 | 5 | NR2 | |
| Wong et al[31] | TLG | 32 (2-79) | 2 | 5-yr OS: 81.0 |
| OG | 27 (1-79) | 9 | 5-yr OS: 67.5 | |
| Chouillard et al[32] | TLG | 29 (12-74) | NR | OS, 1-yr: 88.4, 2-yr: 71.2, 3-y: 43.2 |
| OG | 33 (14-79) | NR | OS, 1-yr: 88.3, 2-yr: 54.8, 3-yr: 38.4 | |
| Scatizzi et al[33] | TLG | 18 (2-37) | NR | 42-mo OS: 70.9 |
| OG | 18 (7-42) | NR | 42-mo OS: 56.8 | |
| Moisan et al[34] | TLG | 28 | 5 | 3-yr DFS: 79.4, 3-yr OS: 82.3 |
| OG | 40 | 4 | 3-yr DFS: 83.4, 3-yr OS: 86.9 | |
| Siani et al[36] | TLG | 32.6 | NR | 5-yr DFS: 54.2, 5-yr OS: 55.7 |
| OG | 31.9 | NR | 5-yr DFS: 52.1, 5-yr OS: 52.9 | |
| Shinohara et al[37] | TLG | 48.8(25-58.5)a | 53 | 5-yr DFS: 65.8b, 5-yr OS: 68.1b |
| OG | 34 | 5-yr DFS: 62.0b, 5-yr OS: 63.7b |
All patients were followed up for at least 2 years;
DFS at 36 mo demonstrated no significant difference between the groups. Follow-up time was shown as median (range); a: Shown as interquartile range; b: Calculated by excluding stage IA and missed follow-up patients. DFS: Disease-free survival; OS: Overall survival; NR: Not reported.
DISCUSSION
The TLG technique was first conceptualized by Goh et al[39] in 1992, who reported two TLGs for the treatment of peptic ulcers. In 1996, Ballesta-Lopez first used this surgical technique for the treatment of gastric cancer and documented its feasibility and efficacy[40]. Although TLG has been in use for over 20 years, its development has been limited as successful reconstruction of the digestive tract has been difficult to achieve laparoscopically. However, continuous improvement and technical advances (in equipment as well as training) have made the application of TLG techniques in oncologic surgery for gastric cancer possible. Although RCTs are ideal for a meta-analysis, it is difficult to conduct a high-quality RCT to evaluate a new surgical intervention due to obstacles such as learning curve effects, ethical and cultural resistance, and urgent or unexpected events during surgery. For these reasons, the inclusion of non-RCTs is an appropriate strategy to extend the source of evidence. Therefore, our meta-analysis included existing observational studies with strict inclusion and exclusion criteria to evaluate the safety and efficacy of TLG.
Postoperative morbidity is usually used to estimate the feasibility and safety of a procedure. Morbidity was significantly lower in the TLG group than in the OG group, regardless of surgical or medical complications. Fewer surgical complications were primarily attributed to reduced wound complications (infections, dehiscence, etc.) in the TLG group. The scattered trocar incisions and contractible sample-extracting incision may explain the reduced wound infection seen in the TLG group. However, other surgical complications (such as leakage and intra-abdominal abscess) were not reduced in the TLG group. This is not surprising as the laparoscopic technique, although less invasive, results in the same organ and lymphatic resection as the open procedure. The significantly reduced medical complications could be explained by the reduced invasiveness of the laparoscopic technique and less postoperative pain. Postoperative pain was less serious in the TLG group than in the OG group as seen by the lower dosage or shorter duration of analgesic use. We also found that pulmonary complications occurred less often in the TLG group than in the OG group. One plausible hypothesis might be that the pain caused by a large incision as well as the use of tension sutures and abdominal bandages after laparotomy can make patients loath to cough, expectorate and perform exercise breathing effectively, thus the patients are prone to complications such as pulmonary infection. Our pooled analysis demonstrated that the postoperative hospital stay was 3.75 d shorter for TLG patients. The reduced use of analgesic drugs, shortened time of abdominal cavity exposure, less bowel manipulation, reduced inflammatory reactions, and earlier postoperative activities are considered to be the main reasons for earlier gastrointestinal recovery from laparoscopic surgery. However, the considerably longer postoperative stay may also have been related to local preferences and different health care systems, and some current series from referral centers report a median stay of more than 14 d after OG, which was considerably longer than that after TLG[25,26,36,37]. The current median length of stay at our hospital after TLG is 10 d, which is similar to most of the included studies[41]. Theoretically, decreased postoperative complication rates and faster recovery have the potential to allow eligible patients to receive postoperative chemotherapy in a timely manner, and therefore, possibly contribute to an improved oncologic outcome. However, these theoretical advantages are unproven.
Operative blood loss and the need for transfusions were lower in TLG cases shown in the pooled analysis. The reduced length of the incision wound and the application of energy-dividing devices, such as the Harmonic Scalpel and Ligasure, contributed to the reduction in blood loss. Another reason is that laparoscopy allows a magnified view of small vessels, particularly during dissection of the plane between the pancreas envelope and some major vessels such as the left gastric artery, common hepatic artery, coeliac trunk and splenic vessels. The most consistent finding in this meta-analysis was the longer operation time for TLG. The learning curve which is related to the surgeon’s experience, familiarity with instruments, and assistant compliance, can influence some of the outcomes studied, such as operation time and lymph node retrieval[42]. It has been suggested that experienced laparoscopic surgeons reach a plateau in operation time after about 40 operations[42,43]. As most of the studies in the present analysis did not explicitly describe the surgeon’s level of proficiency, a subgroup analysis was carried out by selecting studies in which the surgeon had performed more than 40 TLG operations as a surrogate marker of proficiency. This analysis demonstrated a moderate reduction in TLG operation time. Another reason for the prolonged operation time for TLG may be related to the reconstruction step, which is more difficult to complete during laparoscopy. To overcome these potential problems, various modified techniques have been reported. The most representative methods for distal gastrectomy are a delta-shaped anastomosis to perform a Billroth-I gastroduodenostomy and a linear stapler method to perform a side-to-side Billroth II gastrojejunostomy[41,44]. Two other intracorporeal reconstruction methods used in total gastrectomy are also representative; one using a transorally inserted anvil (OrVil™; Covidien) to perform an end-to-side esophagojejunostomy[45], the other using linear staplers to perform a side-to-side anastomosis[46]. These methods help to simplify the procedure of reconstruction and shorten the operation time.
Oncological outcome is a critical measure of success in laparoscopic surgery for malignant tumors. The number of retrieved lymph nodes and surgical resection margin are the major indicators of oncological surgical quality. The present meta-analysis showed that the number of retrieved lymph nodes with TLG was similar to that for OG, and significantly more than the 15 lymph nodes advocated by the Union for International Cancer Control (UICC). To reduce heterogeneity, a subgroup analysis of the extent of modified D2 lymphadenectomy was performed[26,28-30,32,33,37,38], which confirmed that similar numbers of lymph nodes were harvested. With regard to the length of the resection margin, only a limited number of studies reported on the proximal margin. There were no significant differences between the two groups overall. However, our previous meta-analysis demonstrated a reduced proximal margin in the LAG group compared with the OG group[4,15]. We argued that such a result may relate to the nature of LAG, where the specimen is resected and reconstruction is performed through a mini-laparotomy; and it is difficult to pull the proximal stomach using a narrow incision, which may influence the distance of the proximal margin. TLG avoids such difficulties and a longer proximal margin may be expected. The length of the distal resection margin was also similar between the two groups. Both proximal and distal margin lengths demonstrated that TLG is oncologically acceptable for proximal or distal located tumors. Cancer recurrence and long-term survival rate are two visually effective outcomes for evaluating surgical interventions in oncological therapy. The majority of recurrences occur during the first two years after surgery[47], therefore, we used two years as the qualification for NOS to assess the adequate follow-up period of each study. Based on these data, postoperative cancer recurrence and long-term survival rate in the TLG group were similar to those in the OG group. We extracted OS and DFS from data in all available articles. However, we did not analyze this due to the lack of survival data. Therefore, more studies of long-term outcomes are still required to assess the oncological adequacy of TLG.
This analysis has some limitations: (1) most of the studies included were non-randomized and retrospective in nature. Hence, these results are only an estimate of the true benefit of TLG for gastric cancer. We would like to emphasize the importance of RCTs for evaluating the potential short- and long-term benefits of TLG; (2) the laparoscopic cohorts from most, if not all of these institutions, represented initial experiences, and most of the studies had small sample sizes with fewer than 50 TLG procedures, which could have introduced a bias against the outcomes of TLG, because surgical parameters might be influenced by the surgeon’s learning curve; (3) the homogeneity test for the continuous variables showed the existence of significant heterogeneity among the included studies. Therefore, we used a random effects model to evaluate these parameters; and (4) some studies reported that TLG could further reduce trauma compared to LAG, especially in obese patients[48,49]. However, none of the included studies evaluated the effectiveness of TLG for very obese patients, which could undermine the strength of the analysis to some extent.
In conclusion, TLG is a safe, feasible approach for patients with gastric cancer. The results of TLG were favorable in terms of better cosmesis, less blood loss and faster recovery. However, more methodologically high-quality comparative studies are required to adequately evaluate the status of TLG.
COMMENTS
Background
Since it was first reported in 1994, laparoscopic-assisted gastrectomy (LAG) for gastric cancer has undergone rapid development and gained popularity in the past 20 years. During LAG, lymph node dissection is performed laparoscopically. However, resection of the stomach and anastomosis is performed with a direct view through a mini-laparotomy in the epigastrium. Totally laparoscopic gastrectomy (TLG) is considered to be incisionless, except for the trocar wounds, and is a laparoscopic approach for intracorporeal anastomosis without auxiliary incision. Although several meta-analyses and systematic reviews have been published on LAG, similar studies have not been conducted to assess the potential benefits and disadvantages of TLG.
Research frontiers
In order to accurately assess the current status of TLG, the authors strictly limited the inclusion criteria by focusing exclusively on TLG and carried out a comprehensive meta-analysis. This will contribute to a more systematic and objective evaluation of the safety of TLG in cancer treatment.
Innovations and breakthroughs
Based on this meta-analysis, TLG is a safe, feasible approach for patients with gastric cancer. The results of TLG were favorable in terms of better cosmesis, less blood loss and faster recovery, albeit with a longer operation time.
Applications
Despite a longer operation time, TLG can be performed safely in experienced surgical centers with a shorter hospital stay and fewer complications than open surgery. However, more methodologically high-quality comparative studies are required to adequately evaluate the status of TLG.
Peer review
This is a well written paper which will add a great deal to the literature on the subject. In the future, TLG will be rapidly developed in the field of abdominal minimally invasive surgery. Future research should compare LAG and TLG to verify the safety and feasibility of TLG.
Footnotes
Supported by Zhejiang Key Subject of Medical Science Foundation, grant No.11-CX-21
P- Reviewer: Ju JF, Nakayama Y S- Editor: Ma YJ L- Editor: Webster JR E- Editor: Ma S
References
- 1.Kitano S, Iso Y, Moriyama M, Sugimachi K. Laparoscopy-assisted Billroth I gastrectomy. Surg Laparosc Endosc. 1994;4:146–148. [PubMed] [Google Scholar]
- 2.Lee JH, Han HS, Lee JH. A prospective randomized study comparing open vs laparoscopy-assisted distal gastrectomy in early gastric cancer: early results. Surg Endosc. 2005;19:168–173. doi: 10.1007/s00464-004-8808-y. [DOI] [PubMed] [Google Scholar]
- 3.Kim YW, Baik YH, Yun YH, Nam BH, Kim DH, Choi IJ, Bae JM. Improved quality of life outcomes after laparoscopy-assisted distal gastrectomy for early gastric cancer: results of a prospective randomized clinical trial. Ann Surg. 2008;248:721–727. doi: 10.1097/SLA.0b013e318185e62e. [DOI] [PubMed] [Google Scholar]
- 4.Chen K, Xu XW, Mou YP, Pan Y, Zhou YC, Zhang RC, Wu D. Systematic review and meta-analysis of laparoscopic and open gastrectomy for advanced gastric cancer. World J Surg Oncol. 2013;11:182. doi: 10.1186/1477-7819-11-182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xu X, Chen K, Zhou W, Zhang R, Wang J, Wu D, Mou Y. Laparoscopic transgastric resection of gastric submucosal tumors located near the esophagogastric junction. J Gastrointest Surg. 2013;17:1570–1575. doi: 10.1007/s11605-013-2241-2. [DOI] [PubMed] [Google Scholar]
- 6.Shawki S, Bashankaev B, Denoya P, Seo C, Weiss EG, Wexner SD. What is the definition of “conversion” in laparoscopic colorectal surgery? Surg Endosc. 2009;23:2321–2326. doi: 10.1007/s00464-009-0329-2. [DOI] [PubMed] [Google Scholar]
- 7.Bamboat ZM, Strong VE. Minimally invasive surgery for gastric cancer. J Surg Oncol. 2013;107:271–276. doi: 10.1002/jso.23237. [DOI] [PubMed] [Google Scholar]
- 8.Kitano S, Shiraishi N, Fujii K, Yasuda K, Inomata M, Adachi Y. A randomized controlled trial comparing open vs laparoscopy-assisted distal gastrectomy for the treatment of early gastric cancer: an interim report. Surgery. 2002;131:S306–S311. doi: 10.1067/msy.2002.120115. [DOI] [PubMed] [Google Scholar]
- 9.Hayashi H, Ochiai T, Shimada H, Gunji Y. Prospective randomized study of open versus laparoscopy-assisted distal gastrectomy with extraperigastric lymph node dissection for early gastric cancer. Surg Endosc. 2005;19:1172–1176. doi: 10.1007/s00464-004-8207-4. [DOI] [PubMed] [Google Scholar]
- 10.Kim HH, Hyung WJ, Cho GS, Kim MC, Han SU, Kim W, Ryu SW, Lee HJ, Song KY. Morbidity and mortality of laparoscopic gastrectomy versus open gastrectomy for gastric cancer: an interim report--a phase III multicenter, prospective, randomized Trial (KLASS Trial) Ann Surg. 2010;251:417–420. doi: 10.1097/SLA.0b013e3181cc8f6b. [DOI] [PubMed] [Google Scholar]
- 11.Orsenigo E, Di Palo S, Tamburini A, Staudacher C. Laparoscopy-assisted gastrectomy versus open gastrectomy for gastric cancer: a monoinstitutional Western center experience. Surg Endosc. 2011;25:140–145. doi: 10.1007/s00464-010-1147-2. [DOI] [PubMed] [Google Scholar]
- 12.Yakoub D, Athanasiou T, Tekkis P, Hanna GB. Laparoscopic assisted distal gastrectomy for early gastric cancer: is it an alternative to the open approach? Surg Oncol. 2009;18:322–333. doi: 10.1016/j.suronc.2008.08.006. [DOI] [PubMed] [Google Scholar]
- 13.Ohtani H, Tamamori Y, Noguchi K, Azuma T, Fujimoto S, Oba H, Aoki T, Minami M, Hirakawa K. A meta-analysis of randomized controlled trials that compared laparoscopy-assisted and open distal gastrectomy for early gastric cancer. J Gastrointest Surg. 2010;14:958–964. doi: 10.1007/s11605-010-1195-x. [DOI] [PubMed] [Google Scholar]
- 14.Zeng YK, Yang ZL, Peng JS, Lin HS, Cai L. Laparoscopy-assisted versus open distal gastrectomy for early gastric cancer: evidence from randomized and nonrandomized clinical trials. Ann Surg. 2012;256:39–52. doi: 10.1097/SLA.0b013e3182583e2e. [DOI] [PubMed] [Google Scholar]
- 15.Chen K, Xu XW, Zhang RC, Pan Y, Wu D, Mou YP. Systematic review and meta-analysis of laparoscopy-assisted and open total gastrectomy for gastric cancer. World J Gastroenterol. 2013;19:5365–5376. doi: 10.3748/wjg.v19.i32.5365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Grobmyer SR, Pieracci FM, Allen PJ, Brennan MF, Jaques DP. Defining morbidity after pancreaticoduodenectomy: use of a prospective complication grading system. J Am Coll Surg. 2007;204:356–364. doi: 10.1016/j.jamcollsurg.2006.11.017. [DOI] [PubMed] [Google Scholar]
- 17.Hozo SP, Djulbegovic B, Hozo I. Estimating the mean and variance from the median, range, and the size of a sample. BMC Med Res Methodol. 2005;5:13. doi: 10.1186/1471-2288-5-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Weber KJ, Reyes CD, Gagner M, Divino CM. Comparison of laparoscopic and open gastrectomy for malignant disease. Surg Endosc. 2003;17:968–971. doi: 10.1007/s00464-002-8738-5. [DOI] [PubMed] [Google Scholar]
- 20.Dulucq JL, Wintringer P, Stabilini C, Solinas L, Perissat J, Mahajna A. Laparoscopic and open gastric resections for malignant lesions: a prospective comparative study. Surg Endosc. 2005;19:933–938. doi: 10.1007/s00464-004-2172-9. [DOI] [PubMed] [Google Scholar]
- 21.Tanimura S, Higashino M, Fukunaga Y, Takemura M, Tanaka Y, Fujiwara Y, Osugi H. Laparoscopic gastrectomy for gastric cancer: experience with more than 600 cases. Surg Endosc. 2008;22:1161–1164. doi: 10.1007/s00464-008-9786-2. [DOI] [PubMed] [Google Scholar]
- 22.Shim JH, Oh SI, Yoo HM, Jeon HM, Park CH, Song KY. Short-term outcomes of laparoscopic versus open total gastrectomy: a matched-cohort study. Am J Surg. 2013;206:346–351. doi: 10.1016/j.amjsurg.2012.11.011. [DOI] [PubMed] [Google Scholar]
- 23.Sarela AI. Entirely laparoscopic radical gastrectomy for adenocarcinoma: lymph node yield and resection margins. Surg Endosc. 2009;23:153–160. doi: 10.1007/s00464-008-0072-0. [DOI] [PubMed] [Google Scholar]
- 24.Guzman EA, Pigazzi A, Lee B, Soriano PA, Nelson RA, Benjamin Paz I, Trisal V, Kim J, Ellenhorn JD. Totally laparoscopic gastric resection with extended lymphadenectomy for gastric adenocarcinoma. Ann Surg Oncol. 2009;16:2218–2223. doi: 10.1245/s10434-009-0508-3. [DOI] [PubMed] [Google Scholar]
- 25.Huscher CG, Mingoli A, Sgarzini G, Sansonetti A, Di Paola M, Recher A, Ponzano C. Laparoscopic versus open subtotal gastrectomy for distal gastric cancer: five-year results of a randomized prospective trial. Ann Surg. 2005;241:232–237. doi: 10.1097/01.sla.0000151892.35922.f2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Pugliese R, Maggioni D, Sansonna F, Scandroglio I, Ferrari GC, Di Lernia S, Costanzi A, Pauna J, de Martini P. Total and subtotal laparoscopic gastrectomy for adenocarcinoma. Surg Endosc. 2007;21:21–27. doi: 10.1007/s00464-005-0409-x. [DOI] [PubMed] [Google Scholar]
- 27.Song KY, Park CH, Kang HC, Kim JJ, Park SM, Jun KH, Chin HM, Hur H. Is totally laparoscopic gastrectomy less invasive than laparoscopy-assisted gastrectomy?: prospective, multicenter study. J Gastrointest Surg. 2008;12:1015–1021. doi: 10.1007/s11605-008-0484-0. [DOI] [PubMed] [Google Scholar]
- 28.Lee WJ, Wang W, Chen TC, Chen JC, Ser KH. Totally laparoscopic radical BII gastrectomy for the treatment of gastric cancer: a comparison with open surgery. Surg Laparosc Endosc Percutan Tech. 2008;18:369–374. doi: 10.1097/SLE.0b013e31816fdd44. [DOI] [PubMed] [Google Scholar]
- 29.Topal B, Leys E, Ectors N, Aerts R, Penninckx F. Determinants of complications and adequacy of surgical resection in laparoscopic versus open total gastrectomy for adenocarcinoma. Surg Endosc. 2008;22:980–984. doi: 10.1007/s00464-007-9549-5. [DOI] [PubMed] [Google Scholar]
- 30.Strong VE, Devaud N, Allen PJ, Gonen M, Brennan MF, Coit D. Laparoscopic versus open subtotal gastrectomy for adenocarcinoma: a case-control study. Ann Surg Oncol. 2009;16:1507–1513. doi: 10.1245/s10434-009-0386-8. [DOI] [PubMed] [Google Scholar]
- 31.Wong SK, Tsui DK, Li MK. Laparoscopic distal gastrectomy for gastric cancer: initial experience on hand-assisted technique and totally laparoscopic technique. Surg Laparosc Endosc Percutan Tech. 2009;19:298–304. doi: 10.1097/SLE.0b013e3181b0613c. [DOI] [PubMed] [Google Scholar]
- 32.Chouillard E, Gumbs AA, Meyer F, Torcivia A, Helmy N, Toubal M, Karaa A. Laparoscopic versus open gastrectomy for adenocarcinoma: a prospective comparative analysis. Minerva Chir. 2010;65:243–250. [PubMed] [Google Scholar]
- 33.Scatizzi M, Kröning KC, Lenzi E, Moraldi L, Cantafio S, Feroci F. Laparoscopic versus open distal gastrectomy for locally advanced gastric cancer: a case-control study. Updates Surg. 2011;63:17–23. doi: 10.1007/s13304-011-0043-1. [DOI] [PubMed] [Google Scholar]
- 34.Moisan F, Norero E, Slako M, Varas J, Palominos G, Crovari F, Ibañez L, Pérez G, Pimentel F, Guzmán S, et al. Completely laparoscopic versus open gastrectomy for early and advanced gastric cancer: a matched cohort study. Surg Endosc. 2012;26:661–672. doi: 10.1007/s00464-011-1933-5. [DOI] [PubMed] [Google Scholar]
- 35.Cianchi F, Qirici E, Trallori G, Macrì G, Indennitate G, Ortolani M, Paoli B, Biagini MR, Galli A, Messerini L, et al. Totally laparoscopic versus open gastrectomy for gastric cancer: a matched cohort study. J Laparoendosc Adv Surg Tech A. 2013;23:117–122. doi: 10.1089/lap.2012.0310. [DOI] [PubMed] [Google Scholar]
- 36.Siani LM, Ferranti F, De Carlo A, Quintiliani A. Completely laparoscopic versus open total gastrectomy in stage I-III/C gastric cancer: safety, efficacy and five-year oncologic outcome. Minerva Chir. 2012;67:319–326. [PubMed] [Google Scholar]
- 37.Shinohara T, Satoh S, Kanaya S, Ishida Y, Taniguchi K, Isogaki J, Inaba K, Yanaga K, Uyama I. Laparoscopic versus open D2 gastrectomy for advanced gastric cancer: a retrospective cohort study. Surg Endosc. 2013;27:286–294. doi: 10.1007/s00464-012-2442-x. [DOI] [PubMed] [Google Scholar]
- 38.Kim HS, Kim BS, Lee IS, Lee S, Yook JH, Kim BS. Comparison of totally laparoscopic total gastrectomy and open total gastrectomy for gastric cancer. J Laparoendosc Adv Surg Tech A. 2013;23:323–331. doi: 10.1089/lap.2012.0389. [DOI] [PubMed] [Google Scholar]
- 39.Goh P, Tekant Y, Isaac J, Kum CK, Ngoi SS. The technique of laparoscopic Billroth II gastrectomy. Surg Laparosc Endosc. 1992;2:258–260. [PubMed] [Google Scholar]
- 40.Ballesta-Lopez C, Bastida-Vila X, Catarci M, Mato R, Ruggiero R. Laparoscopic Billroth II distal subtotal gastrectomy with gastric stump suspension for gastric malignancies. Am J Surg. 1996;171:289–292. doi: 10.1016/S0002-9610(97)89571-2. [DOI] [PubMed] [Google Scholar]
- 41.Chen K, Xu X, Mou Y, Pan Y, Zhang R, Zhou Y, Wu D, Huang C. Totally laparoscopic distal gastrectomy with D2 lymphadenectomy and Billroth II gastrojejunostomy for gastric cancer: short- and medium-term results of 139 consecutive cases from a single institution. Int J Med Sci. 2013;10:1462–1470. doi: 10.7150/ijms.6632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kim MC, Jung GJ, Kim HH. Learning curve of laparoscopy-assisted distal gastrectomy with systemic lymphadenectomy for early gastric cancer. World J Gastroenterol. 2005;11:7508–7511. doi: 10.3748/wjg.v11.i47.7508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yoo CH, Kim HO, Hwang SI, Son BH, Shin JH, Kim H. Short-term outcomes of laparoscopic-assisted distal gastrectomy for gastric cancer during a surgeon’s learning curve period. Surg Endosc. 2009;23:2250–2257. doi: 10.1007/s00464-008-0315-0. [DOI] [PubMed] [Google Scholar]
- 44.Kanaya S, Gomi T, Momoi H, Tamaki N, Isobe H, Katayama T, Wada Y, Ohtoshi M. Delta-shaped anastomosis in totally laparoscopic Billroth I gastrectomy: new technique of intraabdominal gastroduodenostomy. J Am Coll Surg. 2002;195:284–287. doi: 10.1016/s1072-7515(02)01239-5. [DOI] [PubMed] [Google Scholar]
- 45.Jeong O, Park YK. Intracorporeal circular stapling esophagojejunostomy using the transorally inserted anvil (OrVil) after laparoscopic total gastrectomy. Surg Endosc. 2009;23:2624–2630. doi: 10.1007/s00464-009-0461-z. [DOI] [PubMed] [Google Scholar]
- 46.Wang ZQ, Cai ZM, Chen J, Lei X, Luo HX, Yu PW. A modified method of laparoscopic side-to-side esophagojejunal anastomosis: report of 14 cases. Surg Endosc. 2008;22:2091–2094. doi: 10.1007/s00464-008-9744-z. [DOI] [PubMed] [Google Scholar]
- 47.McCulloch P. The role of surgery in patients with advanced gastric cancer. Best Pract Res Clin Gastroenterol. 2006;20:767–787. doi: 10.1016/j.bpg.2006.03.006. [DOI] [PubMed] [Google Scholar]
- 48.Kim BS, Yook JH, Choi YB, Kim KC, Kim MG, Kim TH, Kawada H, Kim BS. Comparison of early outcomes of intracorporeal and extracorporeal gastroduodenostomy after laparoscopic distal gastrectomy for gastric cancer. J Laparoendosc Adv Surg Tech A. 2011;21:387–391. doi: 10.1089/lap.2010.0515. [DOI] [PubMed] [Google Scholar]
- 49.Sugimoto M, Kinoshita T, Shibasaki H, Kato Y, Gotohda N, Takahashi S, Konishi M. Short-term outcome of total laparoscopic distal gastrectomy for overweight and obese patients with gastric cancer. Surg Endosc. 2013;27:4291–4296. doi: 10.1007/s00464-013-3045-x. [DOI] [PubMed] [Google Scholar]


