Abstract
• Background and Aims The uptake, translocation and redistribution of the heavy metals zinc, manganese, nickel, cobalt and cadmium are relevant for plant nutrition as well as for the quality of harvested plant products. The long-distance transport of these heavy metals within the root system and the release to the shoot in young wheat (Triticum aestivum ‘Arina’) plants were investigated.
• Methods After the application of 65Zn, 54Mn, 63Ni, 57Co and 109Cd for 24 h to one seminal root (the other seminal roots being excised) of 54-h-old wheat seedlings, the labelled plants were incubated for several days in hydroponic culture on a medium without radionuclides.
• Key Results The content of 65Zn decreased quickly in the labelled part of the root. After the transfer of 65Zn from the roots to the shoot, a further redistribution in the phloem from older to younger leaves was observed. In contrast to 65Zn, 109Cd was released more slowly from the roots to the leaves and was subsequently redistributed in the phloem to the youngest leaves only at trace levels. The content of 63Ni decreased quickly in the labelled part of the root, moving to the newly formed parts of the root system and also accumulating transiently in the expanding leaves. The 54Mn content decreased quickly in the labelled part of the root and increased simultaneously in leaf 1. A strong retention in the labelled part of the root was observed after supplying 57Co.
• Conclusions The dynamics of redistribution of 65Zn, 54Mn, 63Ni, 57Co and 109Cd differed considerably. The rapid redistribution of 63Ni from older to younger leaves throughout the experiment indicated a high mobility in the phloem, while 54Mn was mobile only in the xylem and 57Co was retained in the labelled root without being loaded into the xylem.
Keywords: Heavy metals, phloem, redistribution, root system, transport, Triticum aestivum, xylem
INTRODUCTION
Heavy metals are components of the biosphere, occurring naturally in soils and plants, but, as a consequence of industrialization, heavy metals from various sources such as fossil fuel combustion, sewage sludge, industrial waste and fertilizer, contaminate the environment. Plants growing on polluted soils may contain elevated levels of heavy metals (Gallego et al., 2002; Zornoza et al., 2002). Heavy metal ions such as zinc, manganese and nickel are essential micronutrients for plants but, when present in excess, these, and also non-essential heavy metals such as cadmium, can accumulate in plant parts used for human or animal nutrition to undesirably high contents. At even higher levels, they can become toxic to the plant (Williams et al., 2000).
Primary transport in the xylem, retranslocation in the phloem, and transfer from the xylem to the phloem are important processes for the redistribution of an element within a plant (Marschner, 1995). Transport in the xylem is directed from the roots to above-ground plant parts in the transpiration stream, whereas phloem transport takes place from sources to sinks and is more selective (Marschner, 1995). Solutes may be selectively transferred from the xylem to the phloem and channelled to sinks (Feller, 1989; Herren and Feller 1994, 1997a, b).
The distribution of heavy metals within plants varies among the elements. The mobility of the micronutrient zinc within the plant is generally considered to be intermediate between the highly mobile elements such as potassium or phosphorus and the immobile element calcium (Marschner, 1995), but it can be readily transported in the phloem (e.g in wheat; Herren and Feller, 1994, 1996; Pearson et al., 1995; Haslett et al., 2001). The redistribution of zinc may depend on plant age as well as on the zinc contents of the source organs (Herren and Feller, 1996). Cadmium is found in all soils in at least trace quantities (Page et al., 1981). As cadmium and zinc are chemically very similar (Chesworth, 1991), they may be taken up and transported in plants by similar pathways (Grant et al., 1998). The uptake of cadmium by plants is governed by a number of factors such as pH, temperature, aeration, total cadmium concentration in the soil and the presence of other micro- and macronutrients (McLaughlin et al., 1999). Most of the plants studied have retained >50% of the absorbed cadmium in the roots (Obata and Umebayashi, 1993), and the long-distance translocation of cadmium may depend on the availability of other elements (Herren and Feller, 1997a). The micronutrient manganese is, in general, considered to be an element with a rather low phloem mobility, although redistribution of manganese in the phloem may depend on the plant species and the developmental stage (Herren and Feller, 1994). The heavy metals nickel and cobalt are considered to have an intermediate mobility in the phloem (Marschner, 1995), although Zeller and Feller (1998) reported that nickel has a slightly greater phloem mobility than cobalt in wheat. The distribution of cobalt in plants is species-dependent (Moreno-Caselles et al., 1997a, b).
In view of these differences, the aim of the work reported here was to identify the relative mobilities of zinc, manganese, nickel, cobalt and cadmium in the root system of wheat and to compare the transfer of these heavy metals from the labelled root to newly formed roots, to the shoot and to the culture medium.
MATERIALS AND METHODS
Germination and labelling
Grains of wheat (Triticum aestivum L. ‘Arina’) were germinated on wet paper (tissue paper with the nutrient solution mentioned below) in a plastic dish in darkness at a temperature of 21–25 °C. The plastic dish was incubated at a slope of 30°. The grains were placed with the crease turned towards the paper and the embryo oriented downwards on the slope to achieve a homogenous and straight growth of the roots. Two and a half days later, three seminal roots and the shoot were visible. The labelling procedure was based on that reported by Minder and Feller (2003). To ensure that the radionuclides were taken up by only one root, the other two seminal roots were excised with scissors (this procedure caused no major effects on the further development of the plant). The remaining seminal root of the young plants (which was named main root at this time and throughout the experiment) was put into a 300-μL tube (3·5 cm long) containing 300 μL of radiolabelled nutrient solution. The nutrient solution contained 1·5 mm KH2PO4, 0·76 mm MgSO4, 0·34 mm Ca(NO3)2, 0·22 mm KNO3, 7·5 μm Fe (added as Sequestren), 0·25 μm MnCl2, 1·23 μm H3BO3, 0·04 μm ZnSO4, 0·05 μm Na2MoO4, 0·012 μm Ni(NO3)2, 0·025 μm CuSO4 [nutrient solution described in Hildbrand et al. (1994); diluted one to four]. This nutrient medium was used before, during and after (washing and further incubation) the labelling phase throughout the experiment. Co and Cd are not nutrients and were not added separately but were introduced as contaminants of the salts mentioned above to the nutrient medium in a final concentration of 2·1 nm for Co and 0·4 nm for Cd as determined by ICP-MS.
For the analysis of the whole plants by gamma spectrometry or autoradiography, 65Zn (2117 kBq L−1) and 109Cd (4151 kBq L−1) were applied separately. For the experiment investigating the accumulation of 65Zn and 109Cd in the root tips, 65Zn (635·9 kBq L−1) and 109Cd (2474 kBq L−1) were applied as a mixture. 54Mn (66·1 kBq L−1), 63Ni (6835 kBq L−1) and 57Co (32·8 kBq L−1) were applied as a mixture for the analysis with gamma spectrometry or with the liquid scintillation counter. For autoradiography, 54Mn (155·9 kBq L−1), 63Ni (12720 kBq L−1) and 57Co (79·3 kBq L−1) were applied separately. The specific activities of the radionuclide stock solutions were 7·5 MBq µmol−1 for 65Zn, 207·9–4158·8 MBq µmol−1 for 109Cd, >21·4 MBq µmol−1 for 54Mn, 2·2–21·7 MBq µmol−1 for 63Ni and 8·8–18·5 MBq nmol−1 for 57Co. For labelling, the tubes with the plants were placed in a dark container at high humidity. After labelling for 24 h, the seedlings were taken from the tubes, dipped three times sequentially in 100 mL of nutrient solution to remove adsorbed radioactive solutes. From this step to the end of the experiment, the plants were grown and incubated in nutrient solution lacking radionuclides.
The main root of each seedling was then placed in a 300-μL tube containing 300 μL nutrient solution with 0·1 % Congo red for 1 h (in darkness). (Although Congo red stained the root tissues permanently, it had no effect on the subsequent development of the wheat.). Staining with Congo red permitted the part of the main root present during the labelling phase to be discriminated from the more apical part of the main root deriving from later elongation. After labelling, the seedlings were dipped three times sequentially in 100 mL of nutrient solution to remove excess dye. They were then incubated for 1 h in darkness in 200-mL beakers (12 plants per beaker with the root placed in small holes in the cover) containing nutrient solution. Excess dye could diffuse from the root apoplast into the medium (day 0). The nutrient solution was replaced and the seedlings were incubated 24 h in darkness at high humidity to achieve a size suitable for transfer to culture pots (day 1). Beside the main root, one or two additional seminal roots were present after this incubation.
Incubation of plants
Four labelled plants and two identically treated but initially unlabelled control plants were grown together hydroponically in culture pots containing 1 L continuously aerated nutrient solution (without radionuclides). The plants were grown with a photoperiod of 14 h (100 µmol photons m−2 s−1 from four Philips TLD 36W/25 and two Osram Fluora L 36W/77 fluorescent lamps, measured 20 cm above the culture pot). Room temperature was 25 °C during the day and 21 °C at night. The nutrient solution was changed on the 12th day of the experiment and then every 8 d.
Analysis of the plants and the media
To investigate the dynamics of 65Zn, 109Cd, 54Mn, 63Ni and 57Co in wheat, the plants were harvested at different times (0, 1, 4, 8, 12, 20, 28 and 50 d after labelling with radionuclides) and then dissected into the labelled and stained part of the main root, the apical part of the main root including its lateral roots (unstained), the lateral roots branching from the labelled part of the main root, the other roots (other seminal and adventitious nodal roots), the grain, the scutellum (including scutellum, small shoot axis and shoot apex), the coleoptile, leaf 1 (oldest leaf) to leaf 8 (youngest leaf, expanding at the end of the experiment). No tillers were produced during the experiment. The older leaves (leaves 1, 2 and 3 and sometimes leaf 4) were senescent at the end of the experiment (day 50). For the experiment on the accumulation of 65Zn and 109Cd in the root tips, the labelled root and the two newly formed seminal roots were dissected into the root cap, segment 1 (the first segment of 2 mm immediately behind the root cap) to segment 5 (the fifth segment behind the root cap). The different parts of the plants were placed in plastic tubes and held at room temperature for drying. The radioactivity was measured using a gamma counter (1480 Wizard 3″; Wallac Oy, Turku, Finland). A 20-mL sample of the remaining nutrient solution from each culture pot was also analysed in the gamma counter.
For 63Ni measurements, the plant parts were transferred after gamma counting into glass tubes and ashed at 550 °C for 8 h. The ash was dissolved in 1 mL 20 mm citric acid. After mixing, 200 μL were transferred to Ready CapsTM (Beckman Instruments, USA) and dried at <65 °C for 4 h. The radioactivity was then detected in a liquid scintillation counter (beta counter, Betamatic V; Kontron Instruments) and corrected for interferences deriving from other radionuclides (54Mn and 57Co) present in the samples as quantified previously by gamma spectrometry. This correction for the quantification of 63Ni in the presence of 54Mn and 57Co was necessary since 54Mn and 57Co also contributed to the overall measurement in the beta counter.
The experiments were repeated three times (two for counting with the gamma and beta counters and one for autoradiography) with essentially the same results as shown in Table 1. Four replicate plants from the same pot were sampled and analysed separately (pseudoreplicates). The contents of 65Zn, 109Cd, 54Mn, 57Co (gamma counting) and 63Ni (beta counting) are expressed in counts per minute (cpm) per plant part.
Table 1.
Comparison of the radionuclide distribution at the end (day 50) of two separate experiments
| Radionuclide content (% of whole plant) |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plant parts |
65Zn |
109Cd |
54Mn |
63Ni |
57Co |
|||||
| Experiment 1 | ||||||||||
| Leaf 8 | 1·8 ± 0·6ab | 0·2 ± 0·1b | 5·2 ± 2·2a | 1·0 ± 0·3ab | 1·4 ± 0·8ab | |||||
| Leaf 7 | 13·7 ± 0·8b | 2·3 ± 0·1c | 2·6 ± 1·7c | 35·1 ± 4·8a | 0·9 ± 0·4c | |||||
| Leaf 6 | 15·4 ± 1·9a | 3·9 ± 0·2bc | 4·0 ± 2·3bc | 7·4 ± 1·3b | 0·1 ± 0·1c | |||||
| Leaf 5 | 9·9 ± 0·4a | 5·3 ± 0·7ab | 5·0 ± 2·9ab | 0·9 ± 0·3b | 0·3 ± 0·2b | |||||
| Leaf 4 | 5·7 ± 0·5b | 10·6 ± 0·8a | 1·4 ± 1·1c | 0·8 ± 0·5c | 0·9 ± 0·3c | |||||
| Leaf 3 | 6·6 ± 0·5b | 13·0 ± 0·7a | 6·0 ± 3·0b | 1·4 ± 0·6bc | 0·0 ± 0·0c | |||||
| Leaf 2 | 6·9 ± 0·1bc | 15·8 ± 1·5a | 11·4 ± 2·7ab | 1·4 ± 0·3c | 2·8 ± 1·1c | |||||
| Leaf 1 | 11·8 ± 1·0b | 24·3 ± 0·3a | 30·4 ± 4·1a | 1·4 ± 0·5c | 2·8 ± 1·0c | |||||
| Coleoptile | 0·5 ± 0·1b | 1·7 ± 0·5ab | 11·2 ± 4·9a | 2·9 ± 0·9ab | 3·8 ± 1·9ab | |||||
| Scutellum | 17·7 ± 1·0a | 3·6 ± 0·3b | 7·0 ± 4·7b | 3·6 ± 0·4b | 5·7 ± 0·9b | |||||
| Grain | 0·9 ± 0·3a | 2·1 ± 0·6a | 2·1 ± 1·4a | 5·8 ± 1·5a | 4·6 ± 1·7a | |||||
| Labelled part of main root | 0·1 ± 0·0b | 9·2 ± 0·4b | 2·9 ± 2·0b | 5·1 ± 2·7b | 57·7 ± 3·8a | |||||
| Lateral roots of labelled part | 0·2 ± 0·1ab | 0·1 ± 0·1b | 2·2 ± 1·6ab | 3·6 ± 0·7a | 3·1 ± 0·7ab | |||||
| Apical part of main root | 2·2 ± 0·5b | 2·6 ± 0·3b | 4·6 ± 2·7b | 12·4 ± 1·6a | 11·4 ± 1·2a | |||||
| Other roots | 6·6 ± 0·4b | 5·3 ± 0·4b | 4·0 ± 2·5b | 17·2 ± 1·0a | 4·5 ± 1·4b | |||||
| Experiment 2 | ||||||||||
| Leaf 8 | 0·0 ± 0·0a | 0·1 ± 0·1a | 2·8 ± 1·9a | 0·0 ± 0·0a | 1·0 ± 0·9a | |||||
| Leaf 7 | 6·0 ± 2·3b | 1·3 ± 0·0b | 1·5 ± 0·4b | 39·1 ± 4·0a | 1·0 ± 1·0b | |||||
| Leaf 6 | 12·0 ± 1·0ab | 3·3 ± 0·3ab | 2·7 ± 1·3b | 16·2 ± 6·3a | 2·3 ± 1·0b | |||||
| Leaf 5 | 11·4 ± 1·9a | 4·8 ± 0·3b | 4·3 ± 1·7b | 0·0 ± 0·0b | 2·3 ± 1·3b | |||||
| Leaf 4 | 6·4 ± 0·6ab | 9·0 ± 0·8a | 3·2 ± 1·1bc | 0·5 ± 0·5c | 2·3 ± 0·7c | |||||
| Leaf 3 | 7·5 ± 0·6b | 14·5 ± 1·0a | 7·6 ± 1·7b | 0·5 ± 0·5c | 1·8 ± 1·1c | |||||
| Leaf 2 | 12·0 ± 1·8b | 16·6 ± 1·9b | 25·4 ± 2·2a | 0·7 ± 0·7c | 1·3 ± 0·7c | |||||
| Leaf 1 | 18·2 ± 2·1a | 23·6 ± 4·3a | 33·3 ± 6·2a | 0·9 ± 0·9b | 1·4 ± 0·8b | |||||
| Coleoptile | 3·0 ± 1·7a | 4·2 ± 1·1a | 8·0 ± 4·5a | 1·2 ± 1·2a | 1·7 ± 0·8a | |||||
| Scutellum | 12·7 ± 1·4a | 3·1 ± 0·5b | 5·2 ± 1·4b | 1·5 ± 1·4b | 1·8 ± 1·2b | |||||
| Grain | 0·0 ± 0·0a | 1·1 ± 0·1a | 0·2 ± 0·2a | 1·1 ± 1·1a | 4·4 ± 1·8a | |||||
| Labelled part of main root | 2·1 ± 1·0b | 6·4 ± 0·2b | 1·7 ± 1·1b | 0·4 ± 0·4b | 56·4 ± 4·0a | |||||
| Lateral roots of labelled part | 1·9 ± 0·6a | 0·6 ± 0·1a | 0·9 ± 0·4a | 0·0 ± 0·0a | 1·7 ± 0·8a | |||||
| Apical part of main root | 0·9 ± 0·7b | 4·8 ± 0·5ab | 1·8 ± 0·7b | 11·3 ± 4·3ab | 14·3 ± 3·4a | |||||
| Other roots | 5·9 ± 1·2b | 6·6 ± 0·3b | 1·4 ± 0·8b | 26·6 ± 3·5a | 6·3 ± 2·7b | |||||
The radionuclide contents are presented as percentages of the total content in the plant.
Experiment 1 refers to results presented in Figs 2 and 3, whereas Experiment 2 refers to another series.
54Mn, 63Ni and 57Co were applied in both experiments as a mixture allowing the analyses in the same samples (always four plants). For experiment 1, 65Zn (four plants) and 109Cd (four plants) were applied separately. In experiment 2, 65Zn and 109Cd were applied as a mixture (three plants only).
Means ± standard error are shown.
a,b,cValues in the same row (same plant part) followed by the same letter are not significantly different at the P = 0·01 level.
The release of radionuclides from the roots into the medium was measured by analysing samples of the nutrient solution. The transfer of the released radionuclides to unlabelled plants was quantified by analysing two identically treated but initially unlabelled control plants growing together with four labelled plants on the same pot.
For autoradiography, the plants (labelled with only one radionuclide) were harvested, immediately arranged on paper, protected with baking paper and three sheets of typewriter paper and dried by placing a metal plate heated to 220 °C on the top for 10–20 s. Then the plants were exposed to X-ray film (Fuji medical X-ray film, super RX) for 3 months.
Statistical analyses
In Table 1, significant differences were tested with analysis of variance and Student's t-test (Statistix for Windows, version 1·0, Analytical Software, Tallahassee FL, USA; rejection level 0·01, n = 3 or 4).
RESULTS
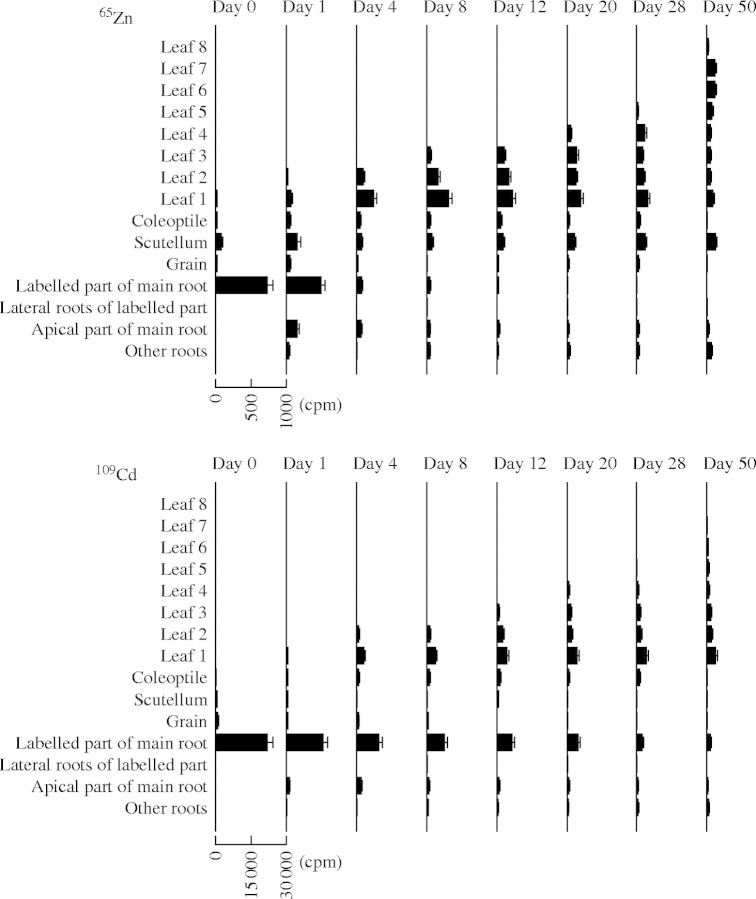
The five radionuclides, 65Zn, 109Cd, 54Mn, 63Ni and 57Co, applied to the young main root were redistributed differently within the root system (Fig. 1). The content of 65Zn decreased in the labelled part of the main root and increased simultaneously in the leaves (Fig. 2). After 4 d, only 15 % of the initial 65Zn remained in this part of the root system. On day 1, there was a marked increase of the content of 65Zn in the apical part of the main root and afterwards it decreased. In the lateral roots branching from the labelled part of the main root, the content of 65Zn was very low. 65Zn increased in the other roots to a low level at the beginning of the experiment and then remained relatively constant. Most of the 65Zn was transferred from the labelled part of the main root to the shoot.
Fig. 1.
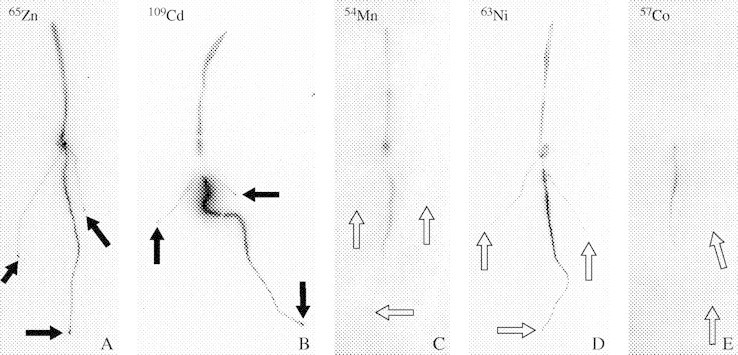
Distribution of 65Zn, 109Cd, 54Mn, 63Ni and 57Co in young wheat plants 2 d after labelling the main root (autoradiographs). The root tips of plants supplied with 65Zn and 109Cd were strongly labelled (black arrows), whereas no such accumulation was observed in the tips after supplying 54Mn, 63Ni or 57Co (open arrows).
Fig. 2.
Dynamics of 65Zn and 109Cd in wheat plants. The plants were dissected into the labelled part of the main root, the lateral roots branching from the labelled part of the main root, the apical part of the main root, the other roots, the grain, the scutellum (including the scutellum, the small shoot axis and the shoot apex), the coleoptile and leaf 1 (oldest leaf) to leaf 8 (youngest leaf). Means and standard errors of four replicates are shown for the radionuclide contents (cpm) in the different plant parts.
The content of 109Cd decreased more slowly in the labelled part of the main root than for 65Zn (Fig. 2). Four days after supplying 109Cd, 50 % of the initial radionuclide remained in this part of the root system. The content of 109Cd was very low in the lateral roots branching from this part. The content of 109Cd in the apical part of the main root increased and then remained relatively constant.
The content of 54Mn decreased quickly in the labelled part of the main root (Fig. 3), so that, after 4 d, only 10% of the initial 54Mn remained. This result indicated a rapid release of the 54Mn to the xylem. As observed for 65Zn and 109Cd, very little of the original 54Mn remained in the root system, most being transported to the shoot.
Fig. 3.
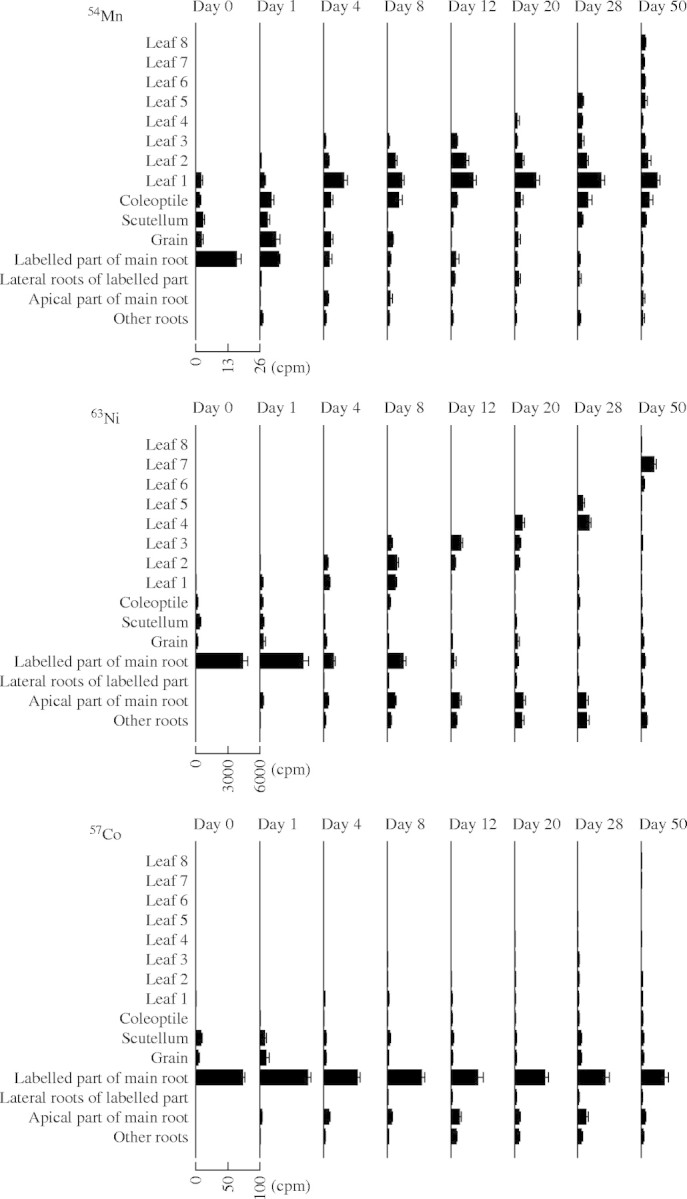
Dynamics of 54Mn, 63Ni and 57Co in wheat plants. The plants were dissected into the labelled part of the main root, the lateral roots branching from the labelled part of the main root, the apical part of the main root, the other roots, the grain, the scutellum (including the scutellum, the small shoot axis and the shoot apex), the coleoptile and leaf 1 (oldest leaf) to leaf 8 (youngest leaf). Means and standard errors of four replicates are shown for the radionuclide contents (cpm) in the different plant parts.
A considerable portion of the applied 63Ni was redistributed within the root system (apical part of the main root, and other roots) (Fig. 3). The content of this radionuclide decreased quickly in the labelled part of the root system and only 20% of the initial 63Ni remained 4 d after labelling. In the lateral roots of the labelled part of the main root, the quantity of 63Ni was very low, but the percentage of 63Ni in the other parts of the root system was higher than for the other heavy metals investigated. This radionuclide accumulated in the apical part of the main root and in the other roots.
A strong retention in the labelled part of the main root was observed after supplying 57Co (Fig. 3). After 4 d, 80% of this radionuclide remained in this part of the root system and about 50% was retained there throughout the experiment (50 d). Some 57Co moved to the apical part of the main root, but the 57Co content in the other parts of the root system remained very low.
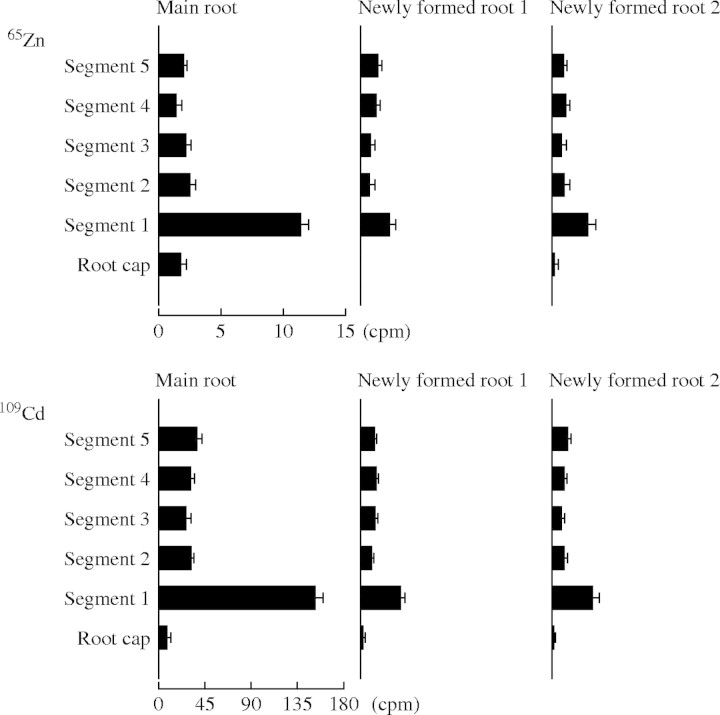
On autoradiographs using 65Zn or 109Cd, but not with 54Mn, 63Ni and 57Co, spots of radioactivity were observed at the tip of the main root and the two newly formed seminal roots, indicating that this local accumulation was rather specific for 65Zn and 109Cd (Fig. 1). A high level of 65Zn was detected in the first segment of 2 mm immediately behind the root cap. The content of 65Zn was low in the root cap and in the four other segments of 2 mm behind the first segment. This fact was observed for the main root and also for the two newly formed seminal roots (Fig. 4). A similar accumulation in the first segment immediately behind the root cap in the main root and in the two newly formed seminal roots was also detected for 109Cd.
Fig. 4.
Distribution of 65Zn and 109Cd in the wheat roots 1 d after labelling. The main root and the two newly formed roots were dissected into the root cap, segment 1 (the first segment of 2 mm immediately behind the root cap) to segment 5 (the fifth segment of 2 mm behind the root cap). Means and standard errors of eight replicates are shown for the radionuclide contents (cpm) in the different root parts.
The root tips of the plants supplied with 65Zn and 109Cd were strongly labelled for some days after labelling. Figure 5 shows plants 4 d after labelling with 109Cd, showing an accumulation of 109Cd at the tips of the main root, the newly formed seminal roots and the lateral roots branching from the labelled part of the main root. No similar accumulation of 109Cd was observed at the tips of the roots of the identically treated but initially unlabelled control plant. The control plants were not exposed to radioactivity but were grown in the same pot as the labelled plants after the labelling period. The reason for doing this was to examine the extent of re-uptake of radioactivity that was previously released into the solution. These results lead to the conclusion that the accumulation of the radionuclides in the root tips must have been caused by an internal transport, probably in the phloem, rather than caused by an uptake of radionuclides, released from the nutrient solution, into the root tips. The content of 109Cd was higher in the other parts of the roots of the control plant, while less radionuclide was found in root parts close to the grain. The quantity of 109Cd in the shoot of the control plant was rather low.
Fig. 5.
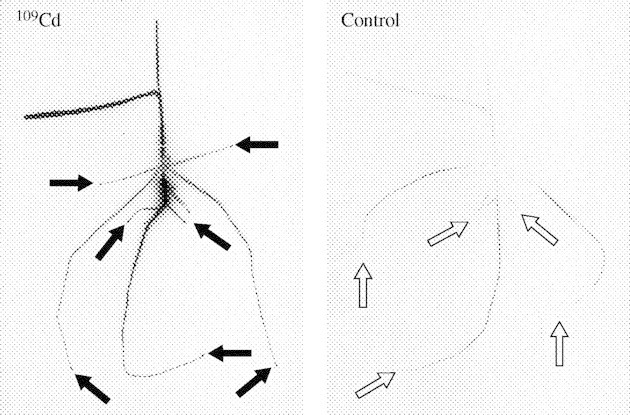
Distribution of 109Cd in young wheat plants 4 d after labelling the main root (autoradiographs). The labelled plant is shown at the left side and the identically treated but initially unlabelled control plant is shown at the right side. The root tips of the plant supplied with 109Cd were strongly labelled (black arrows), while no such accumulation was observed in the tips of the control plant (open arrows).
The dynamics of the five radionuclides differed in the shoot of young wheat plants (Figs 1–3). The transfer of 65Zn from the root system to the shoot was very fast: the content of 65Zn in each leaf increased during its expansion and declined again in a later phase of leaf development. The quantity of 65Zn in the oldest leaves decreased to a low level. Some 65Zn reached the scutellum (including scutellum, short shoot axis and shoot apex) during the first days after its application to the main root and was present there throughout the experiment. In contrast to 65Zn, 109Cd moved slowly into the shoot. The 109Cd content increased in each leaf and afterwards remained relatively constant. The content of 54Mn decreased quickly in the root system and increased simultaneously in leaf 1. 54Mn also moved into leaf 2 but the content in this leaf was lower than in leaf 1, the content of 54Mn in the other leaves was low, and no major redistribution within the shoot was observed. In contrast to the other radionuclides investigated, the content of 54Mn in the coleoptile was relatively high compared with the contents in leaves. The content of 63Ni in the leaves present increased rapidly after its application to the main root, and then it was retranslocated from those leaves to newly expanding leaves. Up the end of the experiment (50 d), 63Ni continued to move from older to younger leaves and to accumulate in the youngest leaf. In contrast, only a very small quantity of 57Co moved to the upper part of the plant.
Table 1 provides a comparison of the radionuclide distribution at the end of the experiment (day 50) of two separate series of experiments. Although the quantities of radionuclides in the plant parts are not exactly the same, the general tendencies described above are very similar in both series of experiments. Differences in the application of 65Zn and 109Cd (separately in experiment 1, as a mixture in experiment 2) caused no major changes in the distribution of these radionuclides.
The labelled plants released some radionuclides in the nutrient solution (Table 2). The re-uptake of the released radionuclides was analysed in two identically treated but initially unlabelled control plants growing together with four labelled plants on the same pot. At day 50, the two unlabelled control plants contained between 1·84 % (109Cd) and 8·46 % (54Mn) of the total radiolabel in the pot, indicating that the release from labelled plants and the re-uptake depended on the element investigated.
Table 2.
Transfer of 65Zn, 109Cd, 54Mn, 63Ni and 57Co from the labelled to the unlabelled plants in the same culture pot
| Time after labelling (d) | Contents in unlabelled plants (% of initial label) |
||||||||
|---|---|---|---|---|---|---|---|---|---|
|
65Zn |
109Cd |
54Mn |
63Ni |
57Co |
|||||
| 4 | 0·45 | 0·68 | 2·45 | 0·71 | 1·64 | ||||
| 8 | 1·82 | 1·33 | 5·30 | 1·15 | 2·05 | ||||
| 12 | 2·49 | 1·28 | 7·92 | 2·19 | 2·95 | ||||
| 20 | 3·53 | 1·17 | 4·30 | 2·46 | 3·66 | ||||
| 28 | 4·31 | 0·94 | 9·78 | 3·81 | 2·83 | ||||
| 50 | 3·87 | 1·84 | 8·46 | 3·25 | 3·69 | ||||
After 1 d, each culture pot contained four labelled and two initially unlabelled plants. The content in the two unlabelled plants is expressed as a percentage of the total content in the pot (including labelled plants, unlabelled plants and nutrient medium).
DISCUSSION
The presence in the other parts of the root system of the five radionuclides 65Zn, 109Cd, 54Mn, 63Ni and 57Co soon after labelling (Figs 1–3), the peak of the 65Zn content in the apical part of the main root at day 1 (Fig. 2) and the accumulation of 65Zn and of 109Cd in the root tips of the labelled plants (Fig. 4) but not in the root tips of the control plants (Fig. 5) lead to the conclusion that the radionuclides were translocated in the phloem from the labelled part of the main root into the other parts of the root system. Indeed, the first segment of 2 mm immediately behind the root cap contained three to six times the quantities of 65Zn and 109Cd in the subsequent segments (Fig. 4). This finding indicates that 65Zn and 109Cd accumulated in the segment containing the apical meristem. If these spots had been present only in the main root, this could be explained by the hypothesis that the radioactivity was present in the meristem after labelling and moved with the growth of the root, but the spots were also observed in the tips of the newly formed roots. This fact indicates that 109Cd and 65Zn were translocated upwards in the xylem, then transferred from the xylem into the phloem and transported in the phloem to the root tips. A similar accumulation of 65Zn in the root tips has been shown previously by Haslett et al. (2001). It has been suggested that the accumulation of 65Zn in this part of the root might be due to a high demand for Zn in the rapidly growing tissue of wheat (Haslett et al., 2001). Moreover, the fact that the spots of radioactivity were not present in the root tips of the control plants led to the conclusion that the redistribution of the radionuclides from the labelled part of the main root to the other parts of the root system was not caused by an efflux from the labelled root into the nutrient medium and an uptake from the medium into the apical segment of the other roots. This accumulation in the tips must have been due to an internal translocation presumably in the phloem. This result is in agreement with the conclusions of Welch et al. (1999), from split root experiments, that Cd can move from one part of the root system of wheat into another part. They reported that Cd movements between different roots must occur in the phloem, either directly through phloem connections between root segments or secondarily by remobilization of Cd from the shoot (Welch et al., 1999). Haslett et al. (2001) concluded that xylem-to-phloem transfer of Zn might occur in the crown where roots meet the stem. However, in the present experiments, the different radionuclides from the labelled root part may also have been transported in the bundles up to the leaves followed by a transfer to the phloem for downward transport to the roots.
A strong retention of 57Co in the labelled part of the main root was evident (Fig. 3), but it remains to be elucidated where (on the tissue level and on the subcellular level) and in which form (soluble or insoluble) 57Co is retained in the root. It is possible that a proportion of the 57Co ions were bound to negatively charged sites on the surface of the cell walls (Wang and Evangelou, 1995). Cd was transported more slowly than Zn, Mn or Ni from the labelled part of the main root to the shoot indicating that Cd was transiently retained in this part of the root system. Cd may accumulate in the vacuole of root cells and may be transported across the tonoplast by several mechanisms, e.g. by a Cd2+/H+-antiport system or a phytochelatin-Cd transporter (Hart et al., 1998). Phytochelatins have been shown to be involved in the detoxification of heavy metals by chelation and subsequent sequestration into the vacuoles. Gong et al. (2003) reported that root-to-shoot transport of phytochelatins occurs and that phytochelatins provide a major mechanism for the long-distance transport of Cd2+ in Arabidopsis. A vacuolar compartmentation of Cd would tend to limit symplastic movement of Cd, resulting in a greater retention of this heavy metal in the root. Variation in the retention of Cd in the roots (caused by a deposition inside the cells or by binding to cell walls) in different plant species or in different genotypes of a given species has been reported (Cakmak et al., 2000). The results presented here are very similar to results obtained from comparable experiments with plants grown on soil (data not shown), reinforcing the hypothesis that heavy metals were transported in the phloem from the labelled part of the main root into the other parts of the root system.
Although 65Zn, 109Cd, 54Mn and 63Ni can move more or less rapidly in the phloem within the root system, a large quantity of these radionuclides (but not of 57Co) moved toward the leaves, indicating that these heavy metals were transported in the transpiration stream in the xylem (Figs 2 and 3). Evidence for this was provided by Salt et al. (1995) who showed that abscisic-induced stomatal closure drastically reduced Cd accumulation in shoots of Indian mustard. The retranslocation of the five radionuclides also differed considerably. There was a redistribution of 65Zn via the phloem from older to younger leaves. Xylem and phloem may contribute to the transport within the shoot, but the export from a leaf must occur via the phloem (Haslett et al., 2001). In contrast to 65Zn, 109Cd was far less effectively loaded into the phloem and transported from older to younger leaves. Reid et al. (2003) reported that Cd transported in the xylem to the leaves of potato plants could be readily remobilized in the phloem, whereas a minor percentage of 109Cd and 54Mn applied to a leaf of young wheat plants was exported in the phloem to younger leaves and roots (Riesen and Feller, 2005). The latter study, and the results presented here showing that 54Mn was transported rapidly in the xylem from the labelled root to the shoot and accumulated in the leaf 1, support the hypothesis that 54Mn was not redistributed via the phloem within the root system or within the shoot. This result is consistent with the evidence that Mn reaches the developing wheat grain mainly in the xylem, whereas Zn can be transported to the maturing ear in the phloem (Herren and Feller, 1994). The low 109Cd and 54Mn content in the youngest wheat leaves (Figs 2 and 3) might derive partially from slow release from the root system and partially from export out of older leaves in the case of 109Cd, or from the radionuclide content in the nutrient solution (Table 2) in the case of 54Mn. As for 65Zn, the results for 63Ni indicate a rapid redistribution in the phloem in the root system as well as in the shoot (Fig. 3), but, in contrast to 65Zn, 63Ni completely disappeared from the oldest leaf and appeared in the youngest leaves. 57Co was strongly retained in the labelled part of the main root and not transferred to the shoot, indicating that this radionuclide was not loaded into the xylem (Fig. 3). However, a minor percentage of the applied 57Co was transferred to the apical part of the labelled root and to other roots, indicating that this element is to some extent mobile in the phloem. It has been observed previously that Co was transferred less effectively than Ni from the xylem to the phloem and that 57Co has a lower phloem mobility compared with 63Ni (Zeller and Feller, 1998, 1999; Riesen and Feller, 2005). The phloem sap contains various types of ligands, such as metal-binding proteins, nicotianamine and citrate, facilitating the transport of micronutrient cations from source to sink organs (Welch, 1995; Krüger et al., 2001), although little is known about the mechanisms involved in the loading of the heavy metals Zn, Cd, Mn, Ni and Co into the phloem and the chemical forms transported in the phloem.
A low percentage of the radionuclides applied to the young main root was released from the labelled plants into the nutrient solution (Table 2). There are three possible mechanisms for this release: (1) intact cells may transfer ions into the nutrient medium across the plasma membrane or release ions by leakage; (2) senescing root cells may release ions when their membrane becomes disrupted; or (3) ions bound to cell wall components may be released.
In conclusion, in young wheat plants, Ni was the most mobile of the heavy metals investigated (Zn, Cd, Mn, Ni and Co). It was readily redistributed within the roots and within the shoot, since the initially applied 63Ni was always detected in the youngest plant parts. Zn was less mobile than Ni, although the initially applied 65Zn was detected in mature and in growing plant parts. 109Cd was far less effectively redistributed than 65Zn or 63Ni, while 54Mn was rapidly transported via the xylem to the shoot and was essentially immobile in the phloem. Co represents an interesting element in this context, since it was to some extent redistributed within the root system but not or only very slowly released to the shoot, indicating a poor loading into the xylem vessels. The mechanisms causing the different transport velocities and distribution patterns remain to be elucidated.
Acknowledgments
This project was funded by the National Centre of Competence in Research (NCCR) Plant Survival, research programme of the Swiss National Science Foundation. We thank Dr Virginie Matera for ICP-MS analyses.
LITERATURE CITED
- Cakmak I, Welch RM, Hart J, Norvell WA, Oztürk L, Kochian LV. 2000. Uptake and retranslocation of leaf-applied cadmium (109Cd) in diploid, tetraploid and hexaploid wheats. Journal of Experimental Botany 51: 221–226. [DOI] [PubMed] [Google Scholar]
- Chesworth W. 1991. Geochemistry of micronutrients. In: Mortvedt JJ, Cox FR, Shuman LM, Welch RM, eds. Micronutrients in agriculture, 2nd edn. Madison, WI: Soil Science Society of America, 1–30. [Google Scholar]
- Feller U. 1989. Transfer of rubidium from the xylem to the phloem in wheat internodes. Journal of Plant Physiology 133: 764–767. [Google Scholar]
- Gallego S, Benavides M, Tomaro M. 2002. Involvement of an antioxidant defence system in the adaptive response to heavy metal ions in Helianthus annuus L. cells. Plant Growth Regulation 36: 267–273. [Google Scholar]
- Gong J-M, Lee DA, Schroeder JI. 2003. Long-distance root-to-shoot transport of phytochelatins and cadmium in Arabidopsis Proceedings of the National Academy of Sciences of the USA 100: 10118–10123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grant CA, Buckley WT, Bailey LD, Selles F. 1998. Cadmium accumulation in crops. Canadian Journal of Plant Science 78: 1–17. [Google Scholar]
- Hart JJ, Welch RM, Norvell WA, Sullivan LA, Kochian LV. 1998. Characterization of cadmium binding, uptake, and translocation in intact seedlings of bread and durum wheat cultivars. Plant Physiology 116: 1413–1420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haslett BS, Reid RJ, Rengel Z. 2001. Zinc mobility in wheat: uptake and distribution of zinc applied to leaves or roots. Annals of Botany 87: 379–386. [Google Scholar]
- Herren T, Feller U. 1994. Transfer of zinc from xylem to phloem in the peduncle of wheat. Journal of Plant Nutrition 17: 1587–1598. [Google Scholar]
- Herren T, Feller U. 1996. Effect of locally increased zinc contents on zinc transport from the flag leaf lamina to the maturing grains of wheat. Journal of Plant Nutrition 19: 379–387. [Google Scholar]
- Herren T, Feller U. 1997. Transport of cadmium via xylem and phloem in maturing wheat shoots: comparison with the translocation of zinc, strontium and rubidium. Annals of Botany 80: 623–628. [Google Scholar]
- Herren T, Feller U. 1997. Influence of increased zinc levels on phloem transport in wheat shoots. Journal of Plant Physiology 150: 228–231. [Google Scholar]
- Hildbrand M, Fischer A, Feller U. 1994. Protein catabolism in bean leaf discs: accumulation of a soluble fragment of ribulose-1,5-bisphosphate carboxylase/oxygenase under oxygen deficiency. Journal of Experimental Botany 45: 1197–1204. [Google Scholar]
- Krüger C, Hell R, Stephan UW. 2001. A metal-binding LEA protein trafficks micronutrients in the phloem of Ricinus communis L. In: Horst WJ, Schenk MK, Bürkert A, Claassen N, Flessa H, Frommer WB, et al, eds. Plant nutrition—food security and sustainability of agro-ecosystems through basic and applied research. Kluwer Academic Publishers: Dordrecht, 194–195. [Google Scholar]
- McLaughlin MJ, Parker DR, Clarke JM. 1999. Metals and micronutrients—food safety issues. Field Crops Research 60: 143–163. [Google Scholar]
- Marschner H. 1995.Mineral nutrition of higher plants, 2nd edn. London: Academic Press. [Google Scholar]
- Minder S, Feller U. 2003. Dynamics of sodium-22 and cesium-134 in young wheat plants. Journal of Plant Nutrition 26: 731–744. [Google Scholar]
- Moreno-Caselles J, Pérez-Espinosa A, Pérez-Murcia MD, Moral R, Gómez I. 1997. Effect of increased cobalt treatments on cobalt concentration and growth of tomato plants. Journal of Plant Nutrition 20: 805–811. [Google Scholar]
- Moreno-Caselles J, Pérez-Espinosa A, Pérez-Murcia MD, Moral R, Gómez I. 1997. Cobalt-induced stress in tomato plants: effect on yield, chlorophyll content, and nutrient evolution. Journal of Plant Nutrition 20: 1231–1237. [Google Scholar]
- Obata H, Umebayashi M. 1993. Production of SH compounds in higher plants of different tolerance to Cd. Plant and Soil 155/156: 533–536. [Google Scholar]
- Page AL, Bingham FT, Chang AC. 1981. Cadmium. In: Lepp NW, eds. Effect of heavy metal pollution on plants. Vol. 1. Effects of trace metals on plant function. Englewood, NJ: Prentice-Hall, 77–109. [Google Scholar]
- Pearson JN, Rengel Z, Jenner CF, Graham RD. 1995. Transport of zinc and manganese to developing wheat grains. Physiologia Plantarum 95: 449–455. [Google Scholar]
- Reid RJ, Dunbar KR, McLaughlin MJ. 2003. Cadmium loading into potato tubers: the roles of the periderm, xylem and phloem. Plant, Cell and Environment 26: 201–206. [Google Scholar]
- Riesen O, Feller U. 2005. Redistribution of nickel, cobalt, manganese, zinc and cadmium via the phloem in young and in maturing wheat. Journal of Plant Nutrition 28: 421–430. [Google Scholar]
- Salt DE, Prince RC, Pickering IJ, Raskin I. 1995. Mechanisms of cadmium mobility and accumulation in Indian mustard. Plant Physiology 109: 1427–1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Evangelou VP. 1995. Metal tolerance aspects of plant cell wall and vacuole. In: Pessarakli M, ed. Handbook of plant and crop physiology. New York, NY: Marcel Dekker, 695–717. [Google Scholar]
- Welch RM. 1995. Micronutrient nutrition of plants. Critical Reviews in Plant Sciences 14: 49–82. [Google Scholar]
- Welch RM, Hart JJ, Norvell WA, Sullivan LA, Kochian LV. 1999. Effects of nutrient solution zinc activity on net uptake, translocation, and root export of cadmium and zinc by separated sections of intact durum wheat (Triticum turgidum L. var. durum) seedling roots. Plant and Soil 208: 243–250. [Google Scholar]
- Williams LE, Pittman JK, Hall JL. 2000. Emerging mechanisms for heavy metal transport in plants. Biochimica et Biophysica Acta 1465: 104–126. [DOI] [PubMed] [Google Scholar]
- Zeller S, Feller U. 1998. Redistribution of cobalt and nickel in detached wheat shoots: effects of steam-girdling and of cobalt and nickel supply. Biologia Plantarum 41: 427–434. [Google Scholar]
- Zeller S, Feller U. 1999. Long-distance transport of cobalt and nickel in maturing wheat. European Journal of Agronomy 10: 91–98. [Google Scholar]
- Zornoza P, Vázquez S, Esteban E, Fernández-Pascual M, Carpena R. 2002. Cadmium-stress in nodulated white lupin: strategies to avoid toxicity. Plant Physiology and Biochemistry 40: 1003–1009. [Google Scholar]