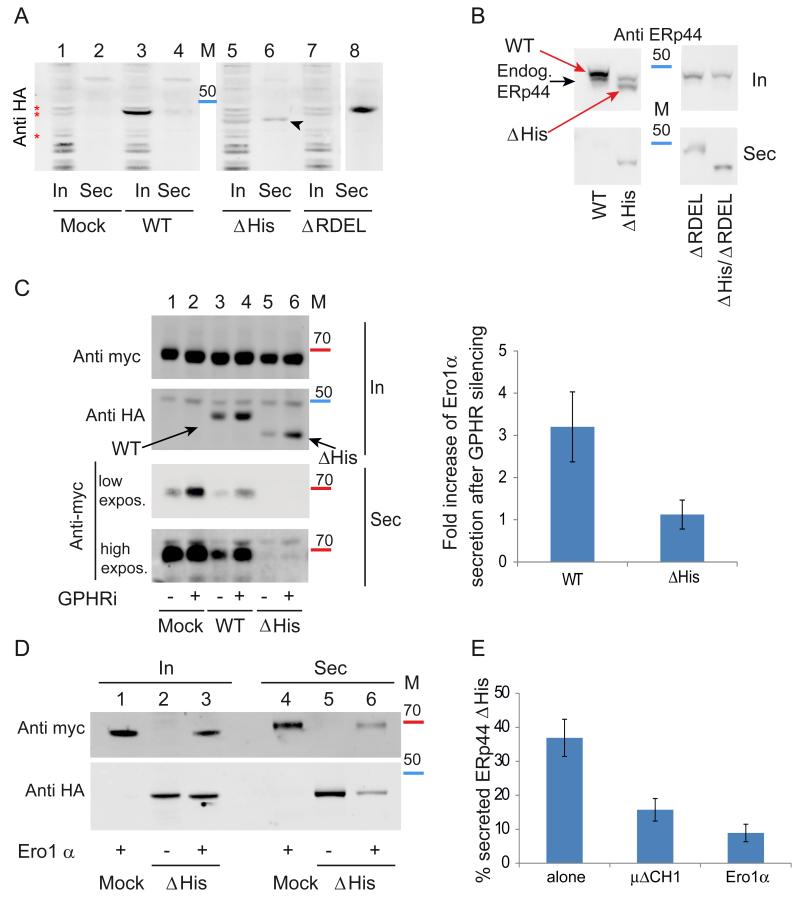
Figure 2. The histidine-rich loop regulates accessibility of the active site and RDEL motif in vivo.
A) ERp44 mutants lacking the histidine-rich loop are secreted. The indicated HeLa transfectants were cultured for 4 hours in fresh medium, and aliquots from lysates (In) and supernatants (Sec) analyzed under reducing conditions by western blotting with anti-HA antibodies. The first two lanes (Mock) show cells transfected with an empty plasmid. The arrowhead points at secreted ΔHis ERp44. Red asterisks point at background bands recognized by anti-HA antibodies. Albeit not elegant, these unwanted bands provide god loading controls. In all Western blot analyses, molecular weight markers were used (see also Fig. 5): the red and blue bands indicate the migration of the 70 and 50 kDa markers.
B) Residual retention of ERp44 ΔHis is due to KDELR activity. Aliquots from lysates (In) and spent media (Sec) were collected from HeLa transfectants (treated as in A) and analyzed under reducing conditions by WB with monoclonal anti-ERp44 antibodies (36C9) that recognize also endogenous ERp44. The black and red arrows point to endogenous and HA-tagged over-expressed ERp44 molecules, respectively. Note that deletion of the C-terminal RDEL allows complete ΔHis secretion, while no endogenous ERp44 is detectable extracellularly.
C) ERp44 ΔHis is hyperactive in retaining Ero1α in a pH-independent manner. HeLa cells were co-transfected with ERp44 variants and Ero1α as indicated and the pH gradient in the ESC was altered by GPHR silencing (− lanes show cells treated with irrelevant siRNA) as described previously (Vavassori et al., 2013). Aliquots from lysates (In) and culture media (Sec) were collected and analyzed by WB by sequential staining of the same filter with anti-HA and anti-myc antibodies to detect ERp44 or Ero1α respectively (as indicated on the left hand margin). Note that less Ero1 is secreted out of cells expressing the ΔHis mutant although its intracellular level is lower than that of wt ERp44. A higher exposure of the secreted samples is shown to detect the small amounts of Ero1α secreted out of cells expressing the ΔHis mutant. Intracellular and secreted proteins were quantified by densitometric analyses, and the mean fold of induction of Ero1α secretion upon GPHR silencing calculated relative to controls (right panel). Data represent the average of ≥ 8 experiments like the one shown in the left panel ± s.d.
D) Client-induced retention of ΔHis. Aliquots of lysates (In) and supernatants (Sec) of the indicated HeLa transfectants were analyzed as above. Note that much less ERp44 ΔHis is secreted in the presence of Ero1α, and vice versa.
E) Secretion of ERp44 ΔHis is inhibited by client proteins (Ero1α and μΔCH1) Secretion of ΔHis in the presence or absence of over-expressed Ero1α or μΔCH1 was analyzed as above. To quantify secretion, we calculated the ratios between the ΔHis bands detected extra- and intracellularly after 4 hours cultivation in fresh medium. To facilitate the comparison amongst different transfectants, the values obtained for wt ERp44 and ΔRDEL were arbitrarily assumed as zero and 100, respectively. Histograms show the average ≥3 experiments +/− s.d.