Figure 8.
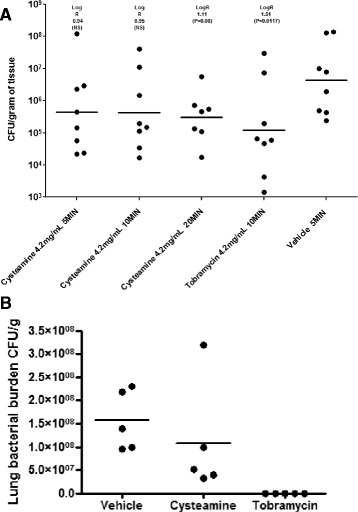
Antimicrobial activity of cysteamine following nebulisation (A) and intra-tracheal dosing (B) in a mouse acute lung infection model. (A) The clinical strain P. aeruginosa EUPPA103 was administered at ~6.5 × 104 CFU/mouse by intranasal injection under temporary inhaled anesthesia. Mice were placed within sealed nebulisation chamber and exposed to cysteamine at 4.2 mg/ml for 5, 10 or 20 minutes (total 1 dose) or tobramycin at 4.2 mg/ml in aqueous solution for 10 minutes via aerosol delivery system 1 hour post-infection. Experimental endpoint was lung tissue burden 25 h post-infection. Vehicle was sterile physiological water. The lower limit of detection was approximately 50 cfu/g of tissue. (B) The clinical strain P. aeruginosa ATCC27853 was administered at 3 × 104 - 1 × 105 cfu/40 μl/mouse by intranasal injection under temporary inhaled anesthesia. Mice were given two doses (5 mg/kg each) of cysteamine or tobramycin delivered intratracheally 10 min and 6 h after infection. Experimental endpoint was lung tissue burden 26 h post-infection. Vehicle was sterile physiological water. The lower limit of detection was approximately 50 cfu/g of tissue.
