Abstract
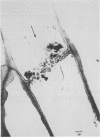
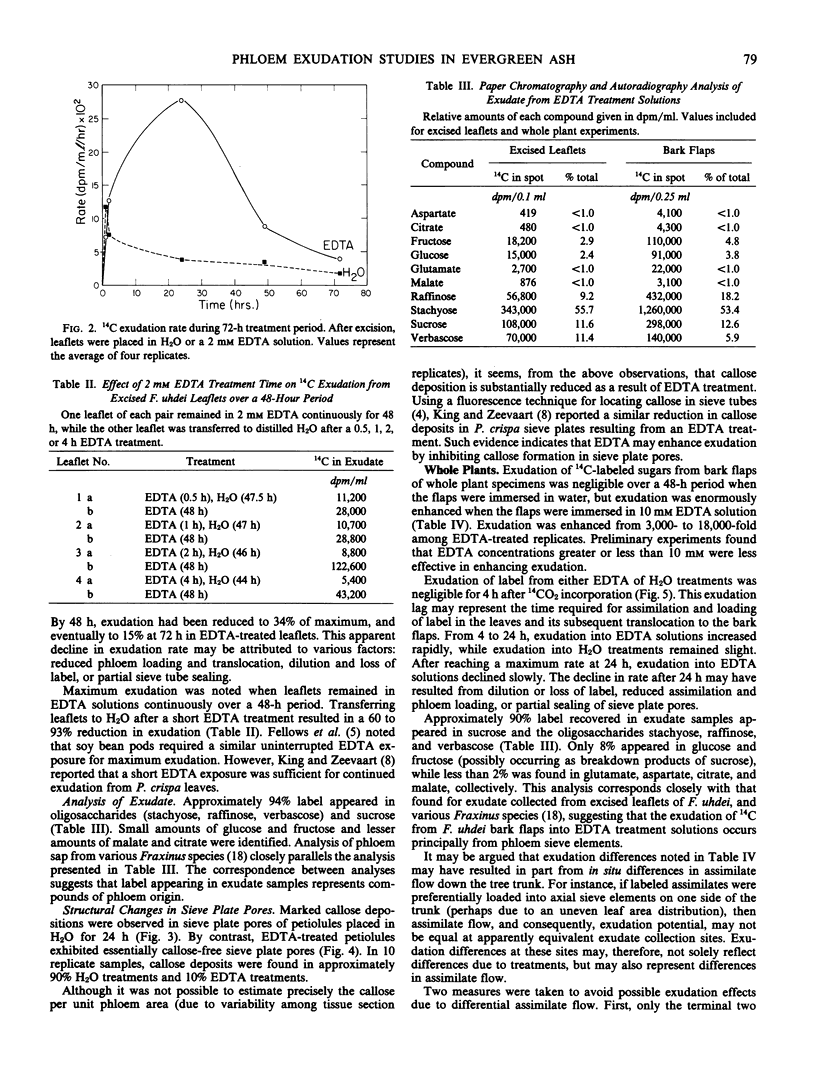
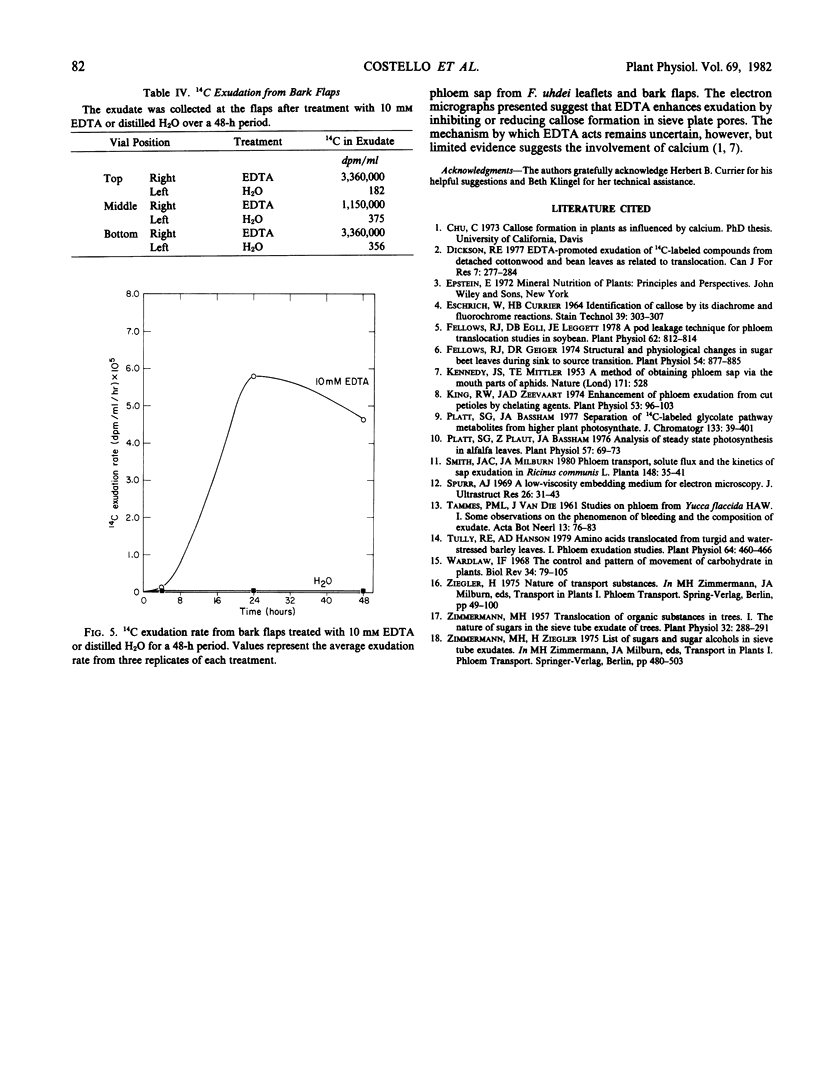
Ethylenediaminetetraacetic acid (EDTA) enhanced the exudation of 14C-labeled assimilates from excised leaflets and whole plant specimens of Fraxinus uhdei Wenz. A 2 millimolar EDTA concentration was found to be most effective in promoting exudation from excised leaflets, while 10 millimolar EDTA was most effective in whole plants experiments. Exudation rate reached a maximum after 24 hours in both experiments. The continuous presence of EDTA throughout the treatment period was required for maximum exudation from excised leaflets. Stachyose, raffinose, verbascose, and sucrose were the principal compounds found to occur in exudate samples. These compounds are typically transported in sieve elements of various Fraxinus species suggesting the exudate was of phloem origin. Electron microscope studies of petiolule sieve plate pores from excised leaflets showed substantially less callose appearing after treatment with EDTA than after H2O treatment. It is suggested that EDTA enhances phloem exudation by inhibiting or reducing callose formation in sieve plate pores. The exudation enhancement technique described for whole plant specimens is suggested as a useful means of collecting phloem sap and studying translocation in woody plants.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Fellows R. J., Egli D. B., Leggett J. E. A Pod Leakage Technique for Phloem Translocation Studies in Soybean (Glycine max [L.] Merr.). Plant Physiol. 1978 Nov;62(5):812–814. doi: 10.1104/pp.62.5.812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fellows R. J., Geiger D. R. Structural and Physiological Changes in Sugar Beet Leaves during Sink to Source Conversion. Plant Physiol. 1974 Dec;54(6):877–885. doi: 10.1104/pp.54.6.877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King R. W., Zeevaart J. A. Enhancement of Phloem exudation from cut petioles by chelating agents. Plant Physiol. 1974 Jan;53(1):96–103. doi: 10.1104/pp.53.1.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Platt S. G., Bassham J. A. Separation of 14 C-labeled glycolate pathway metabolites from higher plant photosynthate. J Chromatogr. 1977 Mar 21;133(2):396–401. doi: 10.1016/s0021-9673(00)83504-9. [DOI] [PubMed] [Google Scholar]
- Platt S. G., Plaut Z., Bassham J. A. Analysis of steady state photosynthesis in alfalfa leaves. Plant Physiol. 1976 Jan;57(1):69–73. doi: 10.1104/pp.57.1.69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res. 1969 Jan;26(1):31–43. doi: 10.1016/s0022-5320(69)90033-1. [DOI] [PubMed] [Google Scholar]
- Tully R. E., Hanson A. D. Amino Acids Translocated from Turgid and Water-stressed Barley Leaves: I. Phloem Exudation Studies. Plant Physiol. 1979 Sep;64(3):460–466. doi: 10.1104/pp.64.3.460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zimmermann M. H. Translocation of Organic Substances in Trees. I. The Nature of the Sugars in the Sieve Tube Exudate of Trees. Plant Physiol. 1957 Jul;32(4):288–291. doi: 10.1104/pp.32.4.288. [DOI] [PMC free article] [PubMed] [Google Scholar]