Alterations in single muscle fibre size (hypertrophy or atrophy) occur by regulating protein turnover (synthesis/degradation rates) and myonuclear content within each cell. Skeletal myofibres are multinucleated (syncytial) cells and postmitotic myonuclei cannot divide to create new nuclei; consequently, myonuclear addition depends on the action of a relatively small pool of muscle stem cells (i.e. satellite cells). While satellite cell activation (and myonuclear addition) coincides with hypertrophy in most models, satellite cell independent hypertrophy has been shown in response to non-physiological conditions (i.e. extreme atrophy, Jackson et al. 2012, or satellite cell ablation, McCarthy et al. 2011). Healthy muscle hypertrophy, regrowth (following atrophy), and regeneration (following injury) are probably distinct processes, with some conditions requiring myonuclear addition via satellite cells and others achieving satellite cell-independent growth.
As a heterogeneous tissue, muscle contains multiple fibre types with distinctive functional and metabolic profiles; human skeletal muscle commonly expresses three myosin heavy chain (MyHC) fibre types (I, IIa and IIx) as well as intermediate ‘hybrids’ (I/IIa, IIa/IIx, I/IIa/IIx). Fibre type-specific responses to the same stimulus often vary widely and intrinsic mechanisms regulating hypertrophy probably differ among fibre types. A recent article published in The Journal of Physiology investigated the effects of aerobic exercise training on MyHC fibre type-specific hypertrophy and satellite cell activation in healthy adult skeletal muscle (Fry et al. 2014). While most mechanistic hypertrophy research in humans utilizes robust resistance training programmes to stimulate growth, this study stood out by using a modest and practical 12 week aerobic training programme. Pre- and post-training vastus lateralis muscle biopsies were taken from 23 volunteers (6 men and 17 women) to investigate MyHC fibre type distribution, fibre size (cross-sectional area, CSA), and satellite cell and myonuclear content using immunofluorescence histochemical analyses. Briefly, the researchers found that aerobic training: (1) increased MyHC IIa and decreased MyHC IIa/IIx (hybrid) fibre proportions, (2) increased MyHC I and IIa fibre CSA, and (3) increased satellite cell and myonuclear content in MyHC I (but not II) fibres in healthy adults. MyHC fibre type distribution and size changes have been shown following aerobic training, but this is the first study to document a phenomenon where hypertrophy occurred in both fibre types (MyHC I and II) with satellite cell activation and myonuclear addition only in MyHC I fibres. The remainder of this commentary will focus on the differential role of myonuclear addition as a hypertrophy mechanism in slow (MyHC I) vs. fast (MyHC II) muscle fibres as highlighted in the investigation by Fry and colleagues.
The ‘myonuclear domain’ represents the sarcoplasmic volume regulated by a single nucleus in multinucleated skeletal muscle fibres. During initial hypertrophy, muscle cell growth may occur without myonuclear addition via enhanced transcriptional and translational responses leading to elevated protein accretion and myonuclear domain expansion. However, there may be a ‘myonuclear domain ceiling’, or theoretical point at which myonuclear addition must take place for sustained fibre hypertrophy (Petrella et al. 2006) as the capacity of genetic machinery and transport distances must be adequate to maintain the larger cell's needs. Significant increases in myonuclei per fibre have been reported when the myofibre size increases >26% (Petrella et al. 2006). In agreement with the myonuclear domain theory, Fry et al. showed modest (yet significant) MyHC IIa fibre growth (12% increase in CSA) accompanied by no significant myonuclear addition (3% increase). In contrast, MyHC I fibres grew (albeit slightly less than IIa fibres), but this hypertrophy was accompanied by significant myonuclear addition (10% increase). At this point I would like to note that by using only an anti-MyHC I antibody to fibre type, myonuclear content was compared between fibres that contained the MyHC I (slow) isoform vs. those that expressed the IIa and/or IIx (fast) isoform. This may produce misclassifications in fibre types (e.g. MyHC I/IIa could be counted as MyHC I fibres). Hybrid fibre types make up a large proportion of human muscle and their physiology is under-researched due to limitations and confusion of identification. Future investigations should utilize methods to measure myonuclear content using antibodies specific to MyHC I, IIa and IIx isoforms to provide complete profiles of the fibre type continuum. Notwithstanding this potential limitation, the threefold increase in myonuclear addition in MyHC I vs. II fibres in this investigation by Fry et al. provides great insight into the potentially divergent hypertrophy mechanisms between fibre types.
MyHC I fibres contain significantly more myonuclei (i.e. smaller myonuclear domains) compared to MyHC IIa fibres regardless of fibre size (Liu et al. 2009). A greater abundance of myonuclei allows for more efficient transport of molecular components needed for rapid manufacturing of new proteins and is probably necessary in MyHC I fibres due to their faster protein synthesis rates (and greater mRNA transcription demand) compared to fast fibre types. The elevated myonuclear content in MyHC I fibres following aerobic training shown by Fry et al. supports the idea that myonuclear domain size is more tightly regulated in MyHC I vs. II fibres. From a practical perspective this makes sense because MyHC I fibres are used during the normal activities of daily living and probably require more frequent repair of damaged proteins compared to the less frequently activated MyHC II fibres.
As mentioned previously, the necessity of satellite cell activation (and myonuclear addition) during hypertrophy has been challenged (McCarthy et al. 2011; Jackson et al. 2012), and the uncertainty of their role in human muscle probably stems from variances in methods (e.g. growth stimulus and histochemical procedures) and populations being studied. Participants in the investigation by Fry et al. consisted of both males and females of various ages (26–68 years) and fitness levels (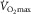 19.4–53.7 ml kg−1 min−1), but no age or sex effect in fibre CSA or satellite cell content changes was reported. Participant heterogeneity can be viewed as a strength given that the results may be applicable to large proportions of the population. However, as mechanisms responsible for fibre type-specific hypertrophy (i.e. satellite cell activation and myonuclear addition) are uncertain (even in highly controlled animal models), future human investigations may consider using more homogeneous participants.
19.4–53.7 ml kg−1 min−1), but no age or sex effect in fibre CSA or satellite cell content changes was reported. Participant heterogeneity can be viewed as a strength given that the results may be applicable to large proportions of the population. However, as mechanisms responsible for fibre type-specific hypertrophy (i.e. satellite cell activation and myonuclear addition) are uncertain (even in highly controlled animal models), future human investigations may consider using more homogeneous participants.
Several variables have been proposed to determine the obligatory nature of myonuclear addition in muscle hypertrophy, including the type of growth stimuli, magnitude and duration of hypertrophy response, age, species, developmental phase, and sample acquisition timing. This investigation by Fry and colleagues adds ‘MyHC fibre type’ to this list; the authors show that slow (MyHC I) and fast (MyHC II) skeletal muscle fibres have the capacity to undergo two modes of hypertrophy in response to the same stimulus, either with or without myonuclear addition via satellite cell activation. The magnitude of the contribution of myonuclear domain regulation to muscle fibre hypertrophy remains to be established, but it may take a more central role in MyHC I fibres. Future research should build upon knowledge presented in the manuscript by Fry et al. to continue elucidating mechanisms responsible for MyHC specific muscle fibre size regulation.
Acknowledgments
I would like to thank Dr Andrew J. Galpin, Mr Kevin A. Murach and Ms Mandy M. Ross for their insightful comments and edits during the preparation of this manuscript.
Additional information
Competing interests
None declared.
Funding
None declared.
References
- Fry CS, Noehren B, Mula J, Ubele MF, Westgate PM, Kern PA. Peterson CA. Fibre type-specific satellite cell response to aerobic training in sedentary adults. J Physiol. 2014;592:2625–2635. doi: 10.1113/jphysiol.2014.271288. &. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jackson JR, Mula J, Kirby TJ, Fry CS, Lee JD, Ubele MF, Campbell KS, McCarthy JJ, Peterson CA. Dupont-Versteegden EE. Satellite cell depletion does not inhibit adult skeletal muscle regrowth following unloading-induced atrophy. Am J Physiol Cell Physiol. 2012;303:C854–C861. doi: 10.1152/ajpcell.00207.2012. &. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J-X, Höglund A-S, Karlsson P, Lindblad J, Qaisar R, Aare S, Bengtsson E. Larsson L. Myonuclear domain size and myosin isoform expression in muscle fibres from mammals representing a 100 000-fold difference in body size. Exp Physiol. 2009;94:117–129. doi: 10.1113/expphysiol.2008.043877. &. [DOI] [PubMed] [Google Scholar]
- McCarthy JJ, Mula J, Miyazaki M, Erfani R, Garrison K, Farooqui AB, Srikuea R, Lawson BA, Grimes B, Keller C, Van Zant G, Campbell KS, Esser KA, Dupont-Versteegden EE. Peterson CA. Effective fiber hypertrophy in satellite cell-depleted skeletal muscle. Development. 2011;138:3657–3666. doi: 10.1242/dev.068858. &. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petrella JK, Kim JS, Cross JM, Kosek DJ. Bamman MM. Efficacy of myonuclear addition may explain differential myofiber growth among resistance-trained young and older men and women. Am J Physiol Endocrinol Metab. 2006;291:E937–E946. doi: 10.1152/ajpendo.00190.2006. &. [DOI] [PubMed] [Google Scholar]


