Abstract
The A- type of glucose 6-phosphate dehydrogenase (EC 1.1.1.49) has been isolated from human erythrocytes deficient in this enzyme. The specific activity of the purified protein is similar to that previously reported for the enzyme isolated from normal, nondeficient erythrocytes. During the purification procedure, a portion of the A- enzyme converts spontaneously, from the native “fraction I”, to a “fraction II” having different kinetic and chromatographic properties. The conversion of fraction I to II can be reproduced freely by treatment with iodosobenzoate, and fraction II can be converted back to fraction I by treatment with dithioglycol. We suggest that fraction II is an enzyme species in which one or more sulfhydryl groups have been oxidized to disulfide(s). The tendency to oxidation appears to be a property specific to the A- variant and may represent the basis for its rapid rate of inactivation and consequent deficiency in vivo.
Keywords: protein structure, sulfhydryl groups, enzyme inactivation, biochemical genetics
Full text
PDF
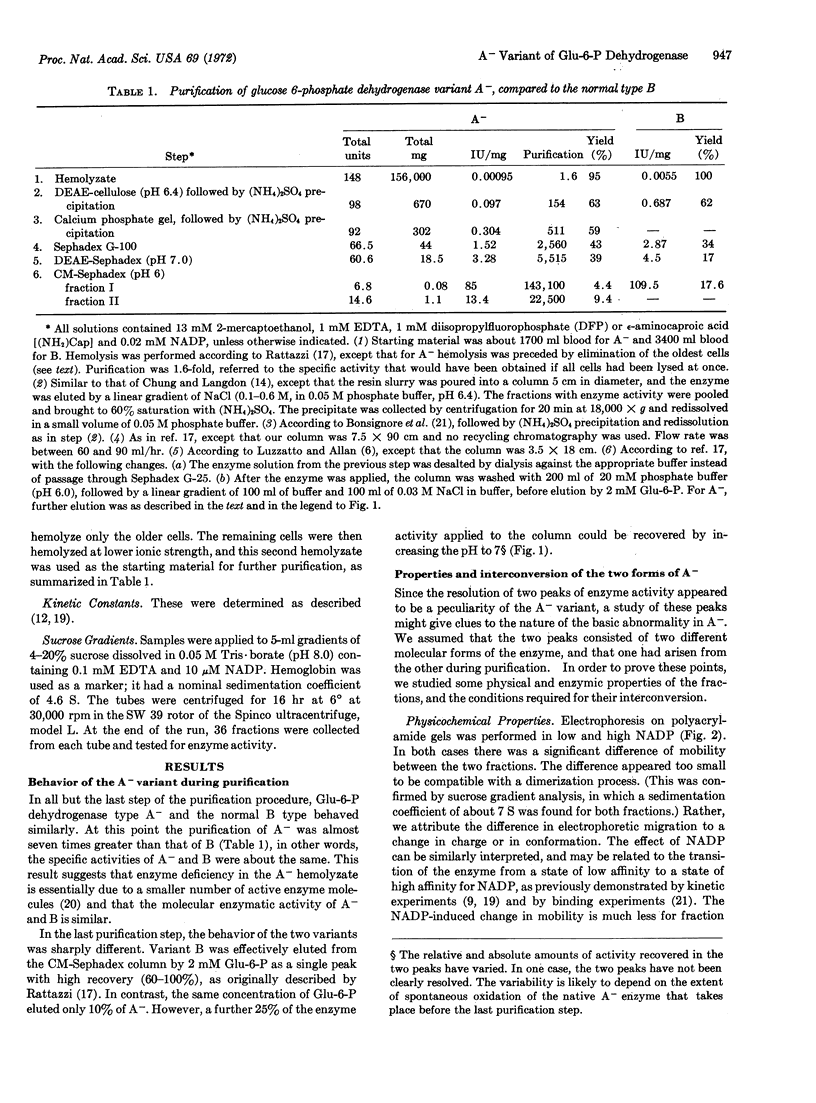

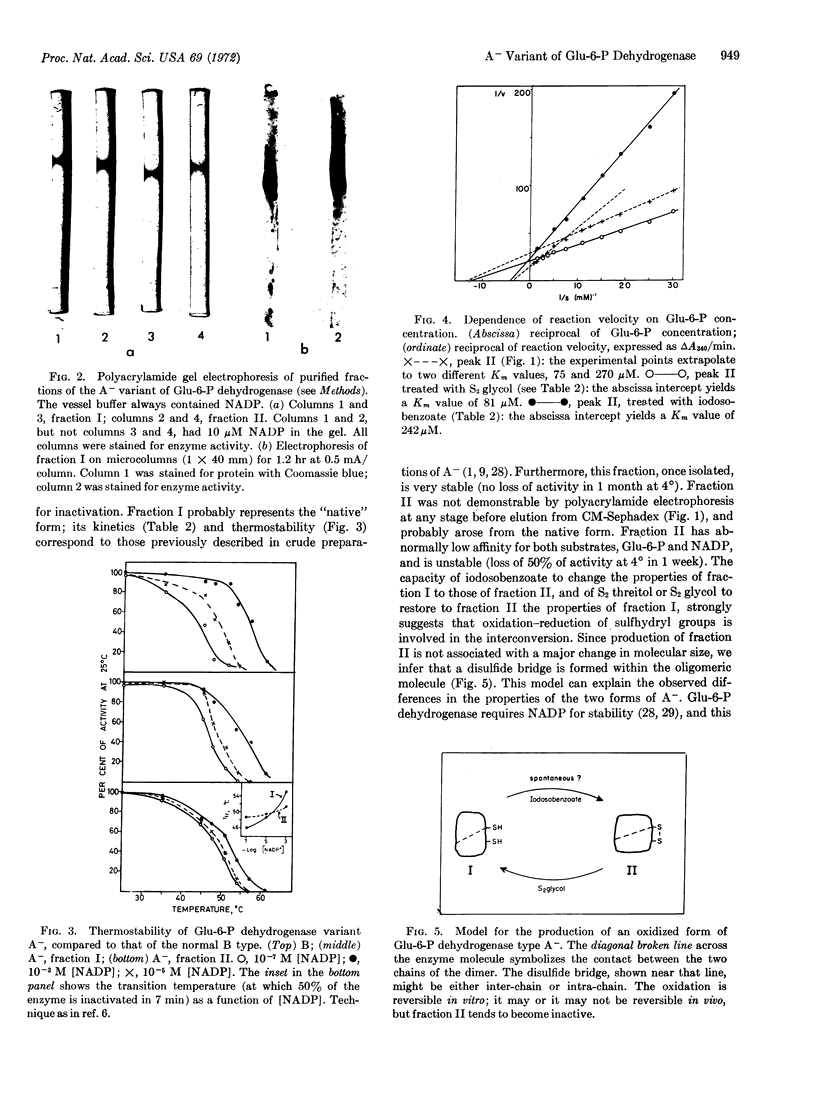

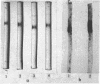
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Afolayan A., Luzzatto L. Genetic variants of human erythrocyte glucose 6-phosphate dehydrogenase. I. Regulation of activity by oxidized and reduced nicotinamide-adenine dinucleotide phosphate. Biochemistry. 1971 Feb 2;10(3):415–419. doi: 10.1021/bi00779a009. [DOI] [PubMed] [Google Scholar]
- BOYER S. H., PORTER I. H., WEILBACHER R. G. Electrophoretic heterogeneity of glucose-6-phosphate dehydrogenase and its relationship to enzyme deficiency in man. Proc Natl Acad Sci U S A. 1962 Oct 15;48:1868–1876. doi: 10.1073/pnas.48.10.1868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonsignore A., Lorenzoni I., Cancedda R., De Flora A. Distinctive patterns of NADP binding to dimeric and tetrameric glucose 6-phosphate dehydrogenase from human red cells. Biochem Biophys Res Commun. 1970 Apr 8;39(1):142–148. doi: 10.1016/0006-291x(70)90769-2. [DOI] [PubMed] [Google Scholar]
- Bonsignore A., Lorenzoni I., Cancedda R., Nicolini A., Damiani G., De Flora A. Purification of glucose 6-phosphate dehydrogenase from human erythrocytes. Ital J Biochem. 1970 May-Jun;19(3):165–177. [PubMed] [Google Scholar]
- CHUNG A. E., LANGDON R. G. Human erythrocyte glucose 6-phosphate dehydrogenase. I. Isolation and properties of the enzyme. J Biol Chem. 1963 Jul;238:2309–2316. [PubMed] [Google Scholar]
- CLELAND W. W. DITHIOTHREITOL, A NEW PROTECTIVE REAGENT FOR SH GROUPS. Biochemistry. 1964 Apr;3:480–482. doi: 10.1021/bi00892a002. [DOI] [PubMed] [Google Scholar]
- Cohen P., Rosemeyer M. A. Human glucose-6-phosphate dehydrogenase: purification of the erythrocyte enzyme and the influence of ions on its activity. Eur J Biochem. 1969 Mar;8(1):1–7. doi: 10.1111/j.1432-1033.1969.tb00487.x. [DOI] [PubMed] [Google Scholar]
- De Flora A., Lorenzoni I., Mangiarotti M. A., Dina D., Bonsignore A. Electrophoretic behaviour of human erythrocyte glucose 6-phosphate dehydrogenase during purification. Biochem Biophys Res Commun. 1968 May 10;31(3):501–507. doi: 10.1016/0006-291x(68)90505-6. [DOI] [PubMed] [Google Scholar]
- KIRKMAN H. N., HENDRICKSON E. M. Glucose 6-phosphate dehydrogenase from human erythrocytes. II. Subactive states of the enzyme from normal persons. J Biol Chem. 1962 Jul;237:2371–2376. [PubMed] [Google Scholar]
- Kirkman H. N., Riley H. D., Crowell B. B. DIFFERENT ENZYMIC EXPRESSIONS OF MUTANTS OF HUMAN GLUCOSE-6-PHOSPHATE DEHYDROGENASE. Proc Natl Acad Sci U S A. 1960 Jul;46(7):938–944. doi: 10.1073/pnas.46.7.938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luzzatto L., Afolayan A. Enzymic properties of different types of human erythrocyte glucose-6-phosphate dehydrogenase, with characterization of two new genetic variants. J Clin Invest. 1968 Aug;47(8):1833–1842. doi: 10.1172/JCI105873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luzzatto L., Allan N. C. Different properties of glucose 6-phosphate dehydrogenase from human erythrocytes with normal and abnormal enzyme levels. Biochem Biophys Res Commun. 1965 Dec 21;21(6):547–554. doi: 10.1016/0006-291x(65)90520-6. [DOI] [PubMed] [Google Scholar]
- Luzzatto L., Okoye V. C. Resolution of genetic variants of human erythrocyte glucose 6-phosphate dehydrogenase by thin-layer chromatography. Biochem Biophys Res Commun. 1967 Dec 15;29(5):705–709. doi: 10.1016/0006-291x(67)90274-4. [DOI] [PubMed] [Google Scholar]
- Luzzatto L. Regulation of the activity of glucose-6-phosphate dehydrogenase by NADP+ and NADPH. Biochim Biophys Acta. 1967 Sep 12;146(1):18–25. doi: 10.1016/0005-2744(67)90069-1. [DOI] [PubMed] [Google Scholar]
- MARKS P. A., GROSS R. T. Erythrocyte glucose-6-phosphate dehydrogenase deficiency: evidence of differences between Negroes and Caucasians with respect to this genetically determined trait. J Clin Invest. 1959 Dec;38:2253–2262. doi: 10.1172/JCI104006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MARKS P. A., SZEINBERG A., BANKS J. Erythrocyte glucose 6-phosphate dehydrogenase of normal and mutant human subjects: properties of the purified enzymes. J Biol Chem. 1961 Jan;236:10–17. [PubMed] [Google Scholar]
- Piomelli S., Corash L. M., Davenport D. D., Miraglia J., Amorosi E. L. In vivo lability of glucose-6-phosphate dehydrogenase in GdA- and GdMediterranean deficiency. J Clin Invest. 1968 Apr;47(4):940–948. doi: 10.1172/JCI105786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell R. D., Brewer G. J., DeGowin R. L., Carson P. E. Effects of glucose-6-phosphate dehydrogenase deficiency upon the host and upon host-drug-malaria parasite interactions. Mil Med. 1966 Sep;131(9 Suppl):1039–1056. [PubMed] [Google Scholar]
- Rattazzi M. C. Isolation and purification of human erythrocyte glucose-6-phosphate dehydrogenase from small amounts of blood. Biochim Biophys Acta. 1969 May;181(1):1–11. doi: 10.1016/0005-2795(69)90221-9. [DOI] [PubMed] [Google Scholar]
- SEGAL H. L., BOYER P. D. The role of sulfhydryl groups in the activity of D-glyceraldehyde 3-phosphate dehydrogenase. J Biol Chem. 1953 Sep;204(1):265–281. [PubMed] [Google Scholar]
- Yoshida A. A single amino Acid substitution (asparagine to aspartic Acid) between normal (b+) and the common negro variant (a+) of human glucose-6-phosphate dehydrogenase. Proc Natl Acad Sci U S A. 1967 Mar;57(3):835–840. doi: 10.1073/pnas.57.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoshida A. Amino acid substitution (histidine to tyrosine) in a glucose-6-phosphate dehydrogenase variant (G6PD Hektoen) associated with over-production. J Mol Biol. 1970 Sep 28;52(3):483–490. doi: 10.1016/0022-2836(70)90414-6. [DOI] [PubMed] [Google Scholar]
- Yoshida A. Glucose 6-phosphate dehydrogenase of human erythrocytes. I. Purification and characterization of normal (B+) enzyme. J Biol Chem. 1966 Nov 10;241(21):4966–4976. [PubMed] [Google Scholar]
- Yoshida A., Stamatoyannopoulos G., Motulsky A. G. Negro variant of glucose-6-phosphate dehydrogenase deficiency (A-) in man. Science. 1967 Jan 6;155(3758):97–99. doi: 10.1126/science.155.3758.97. [DOI] [PubMed] [Google Scholar]