Abstract
Polygonum minus (Polygonaceae), generally known as ‘kesum’ in Malaysia is among the most commonly used food additive, flavoring agent and traditionally used to treat stomach and body aches. Raw or cooked leaves of P. minus are used in digestive disorders in the form of a decoction and the oil is used for dandruff. The pharmacological studies on P. minus have demonstrated antioxidant, in vitro LDL oxidation inhibition, antiulcer activity, analgesic activity, anti-inflammatory activity, in vitro antiplatelet aggregation activity, antimicrobial activity, digestive enhancing property and cytotoxic activity. The spectroscopic studies of essential oil of P. minus showed the presence of about 69 compounds, which are responsible for the aroma. The phytochemical studies showed presence of flavonoids and essential oils. This review is an effort to update the botanical, phytochemical, pharmacological and toxicological data of the plant P. minus.
Keywords: Antioxidant, antiulcer, phytoconstituents, polygonum
INTRODUCTION
Every plant on earth is useful to mankind either directly or indirectly. Many plants have created history in the treatment of diseases. The classical examples include opium for pain management, digitalis for cardiac failure and cinchona for malaria. With the recognition of the role of antioxidants in many diseases, there was a worldwide search for natural antioxidants. Plants containing these principles are nowadays screened for a variety of pharmacological properties. The plant Polygonum minus is not an exception in this context as it is reported to have a high content of antioxidants and hence has gained great attention. In Malaysia it is known as “kesum” and also commonly known as “laksa leaves.” In Malaysia, it is a part of several types of fresh vegetables that are consumed raw, as “ulam” or equivalent to salad in other countries. These include “selom” (Oenanthe javanica; Apiaceae) and “pegaga” (Centella asiatica; Mackinlayaceae). Most of these herbs are believed to be associated with antioxidant activities and have many beneficial effects.[1] Kesum has many claims with regards to its traditional uses. Raw or cooked leaves and seeds of P. minus are used in digestive disorders in the form of a decoction and the oil is used in dandruff. But it is more commonly used as a flavoring agent. This review is focused on the botanical, phytochemical, pharmacological and toxicological aspects of P. minus.
PLANT PROFILE
Synonym: Persicaria minor (Huds.)
Kingdom: Plantae
Superdivision: Spermatophyta
Division: Magnoliophyta
Class: Magnoliopsida
Order: Polygonales
Family: Polygonaceae
Genus: Polygonum
Species: Polygonum minus Huds.
Polygonum minus is generally known as phakphai in Thailand, Kleiner Knöterich in German and Chakhong-machain Manipuri (India). P. minus originated from Southeast Asian countries namely Malaysia, Thailand, Vietnam and Indonesia. It grows wild in damp areas near the ditches, river banks and lakes. It survives well on cool and hilly areas.[2] The plant also grows in India and Sri Lanka, up to 800-900 meters above sea level.
Morphology
This plant [Figure 1] achieves a height of 1.0 m on the lowlands and 1.5 m on the hilly areas. It is a slender twining shrub, frequently climbing up on tall trees. The leaves are long and lanceolate measuring about 5-7 cm long and 0.5-2.0 cm wide. The dark green leaves are aromatic and arranged alternately on a stem. The anatomy and micromorphology of the leaves of P. minus have been described by Bunawan et al.[3] The stem is cylindrical, green and slightly reddish having short internodes with nodes that are easily rooted.[2] However one more type of kesum that is vertical-growing is also available. For commercial production, the vertical type is more suitable for efficient field maintenance and harvesting.[2]
Figure 1.

Morphology of the Polygonum minus
Phytochemistry
P. minus has not has been subjected to much phytochemical studies as there are only a few reports available on its chemical constituents. To date there are reports on identification and isolation of flavonoids and essential oil.
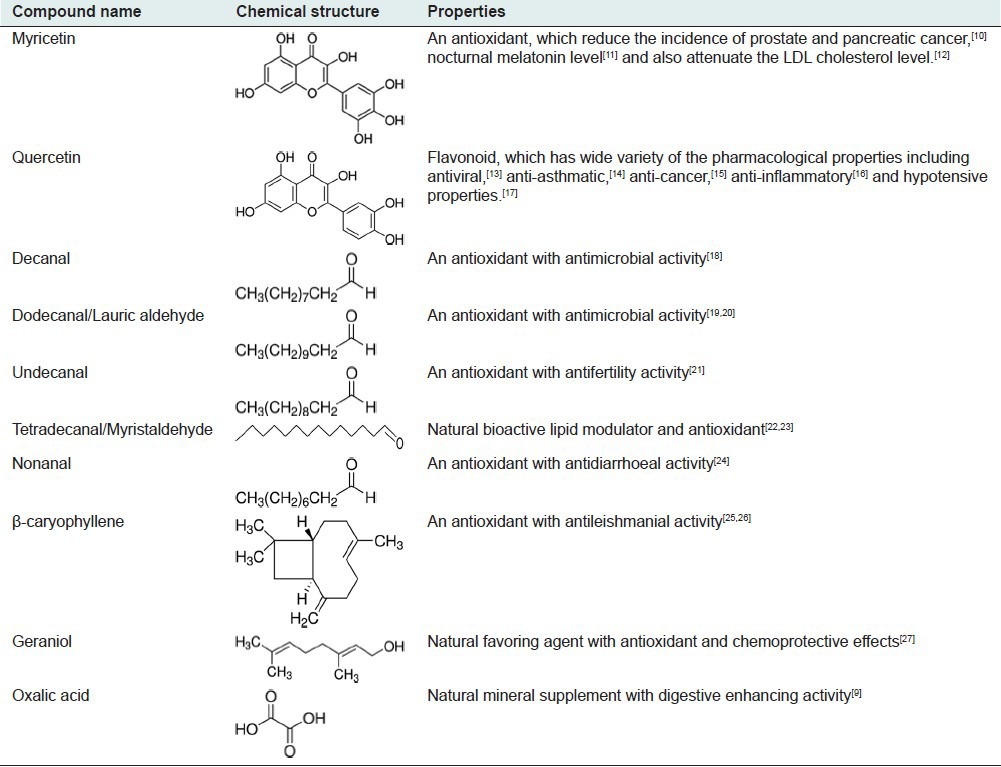
The flavonoids reported in are flavonols: Myricetin [3,5,7-trihydroxy-2-(3,4,5-trihydroxyphenyl)- 4-chromenone] and quercetin [2-(3,4-dihydroxyphenyl) -3,5,7-trihydroxy-4 H-chromen-4-one]; a methyl flavonol [6,7-4’,5’-dimethylenedioxy-3,5,3’- trimethoxyflavone] and a flavone [6,7-methylenedioxy- 5,3’,4’,5’- tetramethoxyflavone].[4,5]
Kesum has high levels of essential oil (72.54%), which contains aliphatic aldehydes (87%).[4] Decanal (24.36%) and dodecanal (48.18%) are major aliphatic aldehydes present in Kesum, others include 1-decanol (2.49%), 1-dodecanol (2.44%), undecanal (1.77%), tetradecanal (1.42%), 1-undecanol (1.41%), nonanal (0.86%), 1-nonanol (0.76%), and β-caryophyllene (0.18%).[6] The essential oil in the leaves of the plant, which is responsible for the aroma is reported to contain 69 compounds that have been identified by gas chromatography and mass spectroscopy.[6] The compounds found in the essential oil include geraniol [(trans)-3,7-dimethyl-2,6-octadien-1-ol] and geranial/citral (3,7-dimethylocta-2,6-dienal).[7] These compounds are responsible for the fragrance and are found to be antimicrobial, antioxidant, immunostimulant and anti-carcinogenic.[8] Vimala et al., reported the presence of oxalic acid in leaves and this may be responsible for the digestive enhancement activity.[9] Pharmacological properties of the selected phytoconstituents of P. minus are listed in Table 1.[9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27]
Table 1.
Pharmacological properties of selected phytoconstituents of P. minus
Pharmacological effects
Pre-clinical studies
Antioxidant capacity
Antioxidant activity of the P. minus was reported by many researchers using DPPH radical scavenging, TBARS and FRAP assay. The high polyphenol content, vitamin C and β carotene was suggested to be responsible for the antioxidant activity of P. minus.[6,28] The aqueous, methanol and ethanol extracts have been reported to possess high antioxidant potential, comparable with synthetic antioxidant such as butylhydroxytoluene (BHT) and gallic acid.[29,30,31,32,33,34] The Total Phenolic Content (TPC) of the methanol and ethanol extracts of the leaves of P. minus were reported to be 31.38 mg and 21.06 mg Gallic Acid Equivalent (GAE) per g of the extract, respectively. The aqueous extract of leaves of P. minus showed a significant antioxidant property and high TPC (2800.6 mg/100 g GAE).[9] TPC of the aqueous, methanol, and ethanol extracts of the whole plant of P. minus were reported to be 55.5 mg, 122.1 mg and 207 mg GAE per g, respectively.[31,35,36] The total ascorbic acid content was found to be 0.54 mg/g FW (fresh weight) and the flavonoid content was found to be 2.46 mg/G DW (dry weight).[32]
Role in LDL oxidation
The inhibitory action of P. minus on LDL oxidation induced by collagen was reported by Saputri and Jantan, 2011.[36] The LDL oxidation activity of the various concentrations of methanol extract of P. minus was measured against copper mediated oxidation in isolated human LDL. It was further reported that this inhibition was dose dependent and an increase in the concentration of the extract showed higher inhibition.[36] This study confirms the correlation between high phenolic content and inhibition of LDL oxidation.
Anti-platelet aggregation activity
The inhibitory potential of the methanol extract of P. minus on human platelet aggregation was reported by Saputri and Jantan, 2011.[36] The inhibitory potential was measured against various platelet aggregation inducers, namely arachidonic acid (AA), adenosine diphosphate (ADP) and collagen. It was reported that this extract was not much effective against AA and ADP induced platelet aggregation as the % of inhibition was 25.6 ± 0.8 and 24.0 ± 2.1 respectively.[36]
Gastro-protective activity
The aqueous extract of whole plant of P. minus was assessed for its ulcer prevention properties against an ethanol induction model.[37] The results showed significant inhibition of ulcerated areas in rats pretreated with P. minus and this effect was dose dependent and the extract (250 mg/kg) was equally effective as omeprazole (20 mg/kg). The gastro-protective effect of the bioactive fraction (EA: MeOH = 1:1) of P. minus leaves may be due to alteration inmucus production, hexoseamine levels and PGE2 synthesis in rodents.[38] Christapher et al., also studied the antiulcer effect of methanol and aqueous extracts of leaves of P. minus in rodents using pyloric ligation model. The methanol extract of P. minus has significant gastro-protective (antiulcer) effect at the dose of 200 mg/kg BW whereas aqueous extracts of P. minus did not showed any gastro-protective in Wistar rats.[39]
Antimicrobial activity
The plant extracts were screened against various bacteria, viruses and fungi. The antibacterial activity was mainly done on Helicobacter pylori that that causes duodenal ulcer. It has been reported that petroleum ether, methanol and chloroform extract of P. minus produced high inhibition zones against H. pylori, however no inhibition zone was observed with aqueous extract.[40] Musa et al., reported the antibacterial activity of P. minus against 10 isolated pathogenic fish bacteria.[41] Further, it has been demonstrated that P. minus was slightly more effective than control in preventing microbial growth in refrigerated duck meatballs.[42] The antiviral activity of ethanol extract of P. minus was tested against Herpes simplex virus type-1(HSV-1) and Vesicular stomatitis virus (VSV), where it has shown strong antiviral activity against HSV-1and weak activity against VSV.[28]
Johnny et al., studied the antifungal activities of the 15 selected Malaysian plants including P. minus against Colletotrichum gloeosporioides isolated from mango. Different extracting solvents were used and methanol extract of P. minus showed effective inhibition of radical growth as compared to chloroform and acetone.[43] Traditionally, P. minus is mixed with a little kerosene and applied as a paste on the skin to get rid of the fungal infections.[44]
Digestive enhancing activity
Vimala et al., demonstrated the presence of oxalic acid in P. minus at 200 nm by High-Performance Liquid Chromatography (HPLC) method. Oxalic acid is a proven digestive aid and this rationalizes the traditional use of kesum as a digestive system stimulant.[9,45]
Cytotoxicity and genotoxicity
Many species of the genus Polygonum viz. P. bistorta, P. avicular and P. multiflorum have been reported to be cytotoxic against a variety of cancer cell lines.[46,47,48] P. avicular has been reported to be effective against cervical cancer cell line. Likewise P. minus also demonstrated cytotoxic effect against Hela cells (CD50:0.1 mg/ml). However when tested against normal lung fibroblast cell line (Hs888Lu), both ethanol and aqueous extracts did not show any effective inhibition.[31] Wan-Ibrahim evaluated 20 Malaysian medicinal plants, which includes P. minus for genotoxic effects on human lymphocytes. Concentrations up to 2000 μg/mL were tested and at the highest concentration P. minus caused moderate DNA damage, i.e. the strand breaks was 31.4%.[49]
Immunomodulatory effect
George et al., studied immunomodulatory property of aqueous extract of P. minus on Swiss albino mice using in vivo carbon clearance assay and reported the dose (200 and 400 mg/kg) dependent increase in phagocytic index. This study showed that the phagocytic index of 400 mg/kg body weight is comparable to standard drug Levamisole at 2.5 mg/kg of body weight.[50]
Antiproliferative activity
Ghazali et al., studied the antiproliferative effect of various solvent extracts of P. minus using in vitro MTT assay against HepG2, WRL68, HeLA, HCT 116, MCF-7 and Chang cell lines. In this study ethanol extract showed lowest IC50 of 32.25 ± 3.72 μg/ml towards HepG2 cell lines with minimum toxicity in WRL68 normal embryonic liver cells whereas methanol extract showed moderate antiproliferative activity against HCT 116 cell lines (IC50 = 56.23 ± 3.2 μg/ml).[51] This study also demonstrate direct correlation between antioxidant capacity and antiproliferative effect of a fraction of ethyl acetate extract.
Anti-inflammatory and analgesic activity
Christapher et al., studied the anti-inflammatory and analgesic activity effect of methanol and aqueous extracts of leaves of P. minus in rodents. The study demonstrated that the aqueous extract possesses analgesic activity in formalin test, acetic acid-induced writhing, tail immersion test and anti-inflammatory activity in carrageenan induced inflammation whereas the methanolic extract showed analgesic activity but no anti-inflammatory activity.[39]
Cognitive enhancing effect
George et al., studied the effect of water extract of P. minus on the learning and memory and reported that the extract has the activity against scopolamine induced cognition deficit in Barnes maze rat model.[52]
Clinical studies
Sexual performance and well-being
Jay et al., conducted a clinical trial on Eurycoma longifolia (200 mg daily) in combination with water extract of P. minus (100 mg daily). Randomized, double blind, and placebo- controlled parallel design study was conducted on healthy male volunteers of age 45-65 years. The study showed that the combination is more effective than placebo in enhancing sexual performance in healthy volunteers.[53]
Clinical safety data
Toxicity to liver and kidney is the major concern of consumption of natural product. P. minus was found to be safe to liver and kidney even for prolonged use (100 mg daily/12 weeks). The basic laboratory values for liver and kidney including albumin, AST, ALT, alkaline phosphatase, bilirubin, Blood Urea Nitrogen (BUN), creatinine, and calculated Glomerular Filtration Rate (GFR) did not show any significant change in the study period.[53]
Toxicity data
Choudhary et al., studied the acute and sub-acute toxic effects of aqueous extract of P. minus on wistar rats for 28 days oral administration and reported that the no-observed adverse- effect level (NOAEL) is more than 1000 mg/kg of the body weight.[54] This study may suggests the safety of raw leaf consumption along with other food in the south east Asia region.
CONCLUSION
Plants containing antioxidant principles are nowadays, extensively screened for various pharmacological activities. P. minus is abundantly available in Southeast Asia and possesses high antioxidant activity. This plant has been long used in traditional medicine and Malay cuisine. The pharmacological activities rationalize the traditional claims about this plant. However the scientific data about this plant and its identified phytoconstituents are limited with respect to its pharmacological activities, pharmacokinetics and clinical trials. Exploration of pharmacological properties, bioactivity- guided isolation of active principles of P. minus and studies on their structure-activity relationships, mechanisms of actions, pharmacokinetics and toxicity are required for the development of P. minus as a successful therapeutic agent. Furthermore, clinical studies also can be conducted with standardized extracts or fractions.
ACKNOWLEDGMENTS
This work was supported in part by AIMST University Internal Grant (AURGC/18/FOP/2013).
Footnotes
Source of Support: Nil
Conflict of Interest: None declared.
REFERENCES
- 1.Kähkönen MP, Hopia AI, Vuorela HJ, Rauha JP, Pihlaja K, Kujala TS, et al. Antioxidant activity of plant extracts containing phenolic compounds. J Agric Food Chem. 1999;47:3954–62. doi: 10.1021/jf990146l. [DOI] [PubMed] [Google Scholar]
- 2.Abd. Rahman Azmil I, Ahmad AW, Omar T. Kesum (Polygonum minus Huds). Penanaman tumbuhan ubatan and beraroma. In: Musa Y, Muhammad Ghawas M, Mansor P, editors. Serdang: MARDI; 2005. pp. 102–7. [Google Scholar]
- 3.Bunawan H, Talip N, Noor NM. Foliar anatomy and micromorphology of Polygonum minus Huds. and their taxonomic implications. Aust J Crop Sci. 2011;5:123–7. [Google Scholar]
- 4.Yaacob KB. Essential oil of Polygonum minus Huds. J Essent Oil Res. 1990;4:167–72. [Google Scholar]
- 5.Imelda F, Faridah DN, Kusumaningrum HD. Bacterial inhibition and cell leakage by extract of Polygonum minus Huds. Leaves. Int Food Res J. 2014;21:553–60. [Google Scholar]
- 6.Baharum SN, Bunawan H, Ghani MA, Mustapha WA, Noor NM. Analysis of the chemical composition of the essential oil of Polygonum minus Huds. Using two-dimensional gas chromatography-time-of-flight mass spectrometry (GC-TOF MS) Molecules. 2010;15:7006–15. doi: 10.3390/molecules15107006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Maarof ND, Ali ZM, Nor ND, Hassan M. Isolation of NADP + geraniol dehydrogenase from Polygonum minus. National biotechnology seminar, Kannur, Kerala, India. 2010. [Last accessed on 2014 Aug 8]. Available in http://www.inbiosis.ukm.my/inbiosis/media/Dr-Maizom-National-Biotech-Symposium. pdf .
- 8.Verlet N. Commercial aspects. In: Hay RK, Waterman PG, editors. Volatile oil crops: Their biology, biochemistry and production. Essex, UK: Longman Scientific and Technical; 1993. pp. 137–74. [Google Scholar]
- 9.Vimala S, Rohana S, Rashih AA, Juliza M. Antioxidant evaluation in Malaysian medicinal plant: Persicaria minor (Huds.) leaf. Sci J Med Clin Trials. 2011;1:9–16. [Google Scholar]
- 10.Nöthlings U, Murphy SP, Wilkens LR, Henderson BE, Kolonel LN. Flavonols and pancreatic cancer risk: The multiethnic cohort study. Am J Epidemiol. 2007;166:924–31. doi: 10.1093/aje/kwm172. [DOI] [PubMed] [Google Scholar]
- 11.Knekt P, Kumpulainen J, Järvinen R, Rissanen H, Heliövaara M, Reunanen A, et al. Flavonoid intake and risk of chronic diseases. Am J Clin Nutr. 2002;76:560–8. doi: 10.1093/ajcn/76.3.560. [DOI] [PubMed] [Google Scholar]
- 12.Ding Y, LI Y. Minireview: Therapeutic potential of myricetin in diabetes mellitus. Food Sci Human Wellness. 2012;1:19–25. [Google Scholar]
- 13.Yu YB, Miyashiro H, Nakamura N, Hattori M, Park JC. Effects of triterpenoids and flavonoids isolated from Alnus firma on HIV-1 viral enzymes. Arch Pharm Res. 2007;30:820–6. doi: 10.1007/BF02978831. [DOI] [PubMed] [Google Scholar]
- 14.Park HJ, Lee CM, Jung ID, Lee JS, Jeong YI, Chang JH, et al. Quercetin regulates Th1/Th2 balance in a murine model of asthma. Int Immunopharmacol. 2009;9:261–7. doi: 10.1016/j.intimp.2008.10.021. [DOI] [PubMed] [Google Scholar]
- 15.Murakami A, Ashida H, Terao J. Multitargeted cancer prevention by quercetin. Cancer Lett. 2008;8:315–25. doi: 10.1016/j.canlet.2008.03.046. [DOI] [PubMed] [Google Scholar]
- 16.Stewart LK, Soileau JL, Ribnicky D, Wang ZQ, Raskin I, Poulev A, et al. Quercetin transiently increases energy expenditure but persistently decreases circulating markers of inflammation in C57BL/6J mice fed a high-fat diet. Metabolism. 2008;57:S39–46. doi: 10.1016/j.metabol.2008.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Edwards RL, Lyon T, Litwin SE, Rabovsky A, Symons JD, Jalili T. Quercetin reduces blood pressure in hypertensive subjects. J Nutr. 2007;137:2405–11. doi: 10.1093/jn/137.11.2405. [DOI] [PubMed] [Google Scholar]
- 18.Liu K, Chen Q, Liu Y, Zhou X, Wang X. Isolation and biological activities of decanal, linalool, valencene, and octanal from sweet orange oil. J Food Sci. 2012;77:C1156–61. doi: 10.1111/j.1750-3841.2012.02924.x. [DOI] [PubMed] [Google Scholar]
- 19.Kubo I, Fujita K, Kubo A, Nihei K, Ogura T. Antibacterial activity of coriander volatile compounds against Salmonella choleraesuis. J Agric Food Chem. 2004;52:3329–32. doi: 10.1021/jf0354186. [DOI] [PubMed] [Google Scholar]
- 20.Hajhashemi V, Rabbani M, Ghanadi A, Davari E. Evaluation of antianxiety and sedative effects of essential oil of Ducrosia anethifolia in mice. Clinics (Sao Paulo) 2010;65:1037–42. doi: 10.1590/S1807-59322010001000020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rai M, Carpinella MC. Naturally occurring bioactive compounds. The Netherlands: Elsevier; 2006. Advance on phytomedicine (volume 3) [Google Scholar]
- 22.Tang EL, Rajarajeswaran J, Fung SY, Kanthimathi MS. Antioxidant activity of Coriandrum sativum and protection against DNA damage and cancer cell migration. BMC Complement Altern Med. 2001;13:347. doi: 10.1186/1472-6882-13-347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bushnev AS, Baillie MT, Holt JJ, Menaldino DS, Merrill AH, Jr, Liotta DC. An efficient asymmetric synthesis of enigmols (1-deoxy-5-hydroxysphingoid bases), an important class of bioactive lipid modulators. ARKIVOC 2010. 2010:263–77. [Google Scholar]
- 24.Zavala-Sánchez MA, Pérez-Gutiérrez S, Perez-González C, Sánchez-Saldivar D, Arias-García L. Antidiarrhoeal activity of nonanal, an aldehyde isolated from Artemisia ludoviciana. Pharm Biol. 2002;40:263–8. [Google Scholar]
- 25.Soares DC, Portella NA, Ramos MF, Siani AC, Saraiva EM. Trans- β -Caryophyllene: An effective antileishmanial compound found in commercial copaiba oil (Copaifera spp.) Evid Based Complement Alternat Med 2013. 2013:7613–23. doi: 10.1155/2013/761323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kim C, Cho SK, Kim KD, Nam D, Chung WS, Jang HJ, et al. β-Caryophyllene oxide potentiates TNFα-induced apoptosis and inhibits invasion through down-modulation of NF-kB-regulated gene products. Apoptosis. 2014;19:708–18. doi: 10.1007/s10495-013-0957-9. [DOI] [PubMed] [Google Scholar]
- 27.Carnesecchi S, Schneider Y, Ceraline J, Duranton B, Gosse F, Seiler N, et al. Geraniol, a component of plant essential oils, inhibits growth and polyamine biosynthesis in human colon cancer cells. J Pharmacol ExpTher. 2001;298:197–200. [PubMed] [Google Scholar]
- 28.Ali AM, Mackeen MM, El-Sharkawy SH, Abdul Hamid J, Ismail NH, Ahmad F, et al. Antiviral and cytotoxic activities of some plants used in Malaysian indigenous medicine. Pertanika J Trop Agric Sci. 1996;19:129–36. [Google Scholar]
- 29.Faujan N, Abdullah N, Sani A, Babji A. Antioxidative activities of water extracts of some Malaysian herbs. ASEAN Food J. 2007;14:61–8. [Google Scholar]
- 30.Huda-Faujan N, Noriham A, Norrakiah A, Babji A. Antioxidant activity of plants methanolic extracts containing phenolic compounds. Afr J Biotechnol. 2010;8:484–9. [Google Scholar]
- 31.Qader SW, Abdulla MA, Chua LS, Najim N, Zain MM, Hamdan S. Antioxidant, total phenolic content and cytotoxicity evaluation of selected Malaysian plants. Molecules. 2011;16:3433–43. doi: 10.3390/molecules16043433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sumazian Y, Syahid A, Hakim M, Maziah M. Antioxidant activities, flavonoids, ascorbic acid and phenolic content of Malaysian vegetables. J Med Plant Res. 2010;4:881–90. [Google Scholar]
- 33.Gorinstein S, Vargas O, Jaramillo N, Salas I, Ayala A, Arancibia-Avila P, et al. The total polyphenols and the antioxidant potentials of some selected cereals and pseudocereals. Eur Food Res Technol. 2007;225:321–8. [Google Scholar]
- 34.Almey AA, Khan CA, Zahir IS, Suleiman KM, Aisyah MR, Rahim KK. Total phenolic content and primary antioxidant activity of methanolic and ethanolic extracts of aromatic plants’ leaves. Int Food Res J. 2010;17:1077–84. [Google Scholar]
- 35.Qader SW, Abdulla MA, Chua LS, Najim N, Zain MM, Hamdan S. Antioxidant, total phenolic content and cytotoxicity evaluation of selected Malaysian plants. Molecules. 2011;16:3433–43. doi: 10.3390/molecules16043433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Saputri FC, Jantan I. Effects of selected medicinal plants on human low-density lipoprotein oxidation, 2, 2-diphenyl-1-picrylhydrazyl (DPPH) radicals and human platelet aggregation. J Med Plant Res. 2011;5:6182–91. [Google Scholar]
- 37.Wasman S, Mahmood A, Salehhuddin H, Zahra A, Salmah I. Cytoprotective activities of Polygonum minus aqueous leaf extract on ethanol-induced gastric ulcer in rats. J Med Plants Res. 2010;4:2658–65. [Google Scholar]
- 38.Qader SW, Abdulla MA, Chua LS, Sirat HM, Hamdan S. Pharmacological mechanisms underlying gastroprotective activities of the fractions obtained from Polygonum minus in Sprague Dawley rats. Int J Mol Sci. 2012;13:1481–96. doi: 10.3390/ijms13021481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Christapher PV, Xin TY, Kiun CF, Leng LC, Fu NG, Yuan GL, et al. Evaluation of analgesic, anti-inflammatory, antipyretic and antiulcer effect of aqueous and methanol extracts of leaves of Polygonum minus Huds.(Polygonaceae) in rodents. Arch Med Health Sci (In press) [Google Scholar]
- 40.Uyub AM, Nwachukwu IN, Azlan AA, Fariza SS. In vitro antibacterial activity and cytotoxicity of selected medicinal plant extracts from Penang Island Malaysia on metronidazole-resistant Helicobacter pylori and some pathogenic bacteria. Ethnobot Res Appl. 2010;8:95–106. [Google Scholar]
- 41.Musa N, Wei LS, Seng CT, Wee W, Leong LK. Potential of edible plants as remedies of systemic bacterial disease infection in cultured fish. Glob J Pharmacol. 2008;2:31–6. [Google Scholar]
- 42.Faridah A. Effects of herbal marinades on the shelf. life of chilled chicken Satay. Thesis submitted to Universiti Teknologi Mara, Malaysia. 2008. [Last accessed on 2014 Jan 9]. Available from: http://www.eprints.uitm.edu.my/id/eprint/1037 or http://www.eprints.uitm.edu.my/1037/
- 43.Johnny L, Yusuf UK, Nulit R. Antifungal activity of selected plant leaves crude extracts against a pepper anthracnose fungus, Colletotrichum capsici (Sydow) butler and bisby (Ascomycota: Phyllachorales) Afr J Biotechnol. 2011;10:4157–65. [Google Scholar]
- 44.Ong H, Nordiana M. Malay ethno-medico botany in Machang, Kelantan, Malaysia. Fitoterapia. 1999;70:502–13. [Google Scholar]
- 45.Eastwood MA, Nyhlin H. Beeturia and colonic oxalic acid. QJM. 1995;88:711–7. [PubMed] [Google Scholar]
- 46.Manoharan KP, Yang D, Hsu A, Huat BT. Evaluation of Polygonum bistorta for anticancer potential using selected cancer cell lines. Med Chem. 2007;3:121–6. doi: 10.2174/157340607780059495. [DOI] [PubMed] [Google Scholar]
- 47.Mohammad R, Hossein B, Davood F, Farnaz T, Ali F, Yuse R. The apoptotic and cytotoxic effects of Polygonum avicular extract on Hela-S cervical cancer cell line. Afr J Biochem Res. 2011;5:373–8. [Google Scholar]
- 48.Liu LP, Liao ZP, Yin D, Li WD, Liu D, Li Q, et al. The protective effects of Polygonum multiflorum stilbene glycoside preconditioning in an ischemia/reperfusion model of HUVECs. Acta Pharmacol Sin. 2010;31:405–12. doi: 10.1038/aps.2010.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wan-Ibrahim W, Sidik K, Kuppusamy U. A high antioxidant level in edible plants is associated with genotoxic properties. Food Chem. 2010;122:1139–44. [Google Scholar]
- 50.Annie G, Sasikala C, Yogendra C, Praveen B, Murthy S. Immunomodulatory activity of an aqueous extract of Polygonum minus. Huds on Swiss albino mice using carbon clearance assay. Asian Pac J Trop Dis. 2014;4:398–400. [Google Scholar]
- 51.Ghazali MA, Al-Naqeb G, Selvarajan KK, Hasan MH, Adam A. Apoptosis induction by Polygonum minus is related to antioxidant capacity, alterations in expression of apoptotic-related genes, and S-Phase cell cycle arrest in HepG2 cell line. Biomed Res Int 2014. 2014:13. doi: 10.1155/2014/539607. Article ID 539607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.George A, Ng CP, O’Callaghan M, Jensen GS, Wong HJ. In vitro and ex-vivo cellular antioxidant protection and cognitive enhancing effects of an extract of Polygonum minus Huds (Lineminus™) demonstrated in a Barnes Maze animal model for memory and learning. BMC Complement Altern Med. 2014;14:161. doi: 10.1186/1472-6882-14-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Udani JK, George AA, Musthapa M, Pakdaman MN, Abas A. effects of a proprietary freeze-dried water extract of Eurycoma longifolia (Physta) and Polygonum minus on sexual performance and well-being in men: A randomized, double-blind, placebo-controlled study. Evid Based Complement Alternat Med 2014. 2014:179529. doi: 10.1155/2014/179529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Ming YK, Noraisyah BZ, Vandana KC, Yogendra KC. Acute and sub-acute oral toxicity of Polygonum minus aqueous extract (BIOTROPICS®PM101) in wistar rats. Int J Pharm Pharm Sci. 2013;5:120–4. [Google Scholar]


