Figure 1.
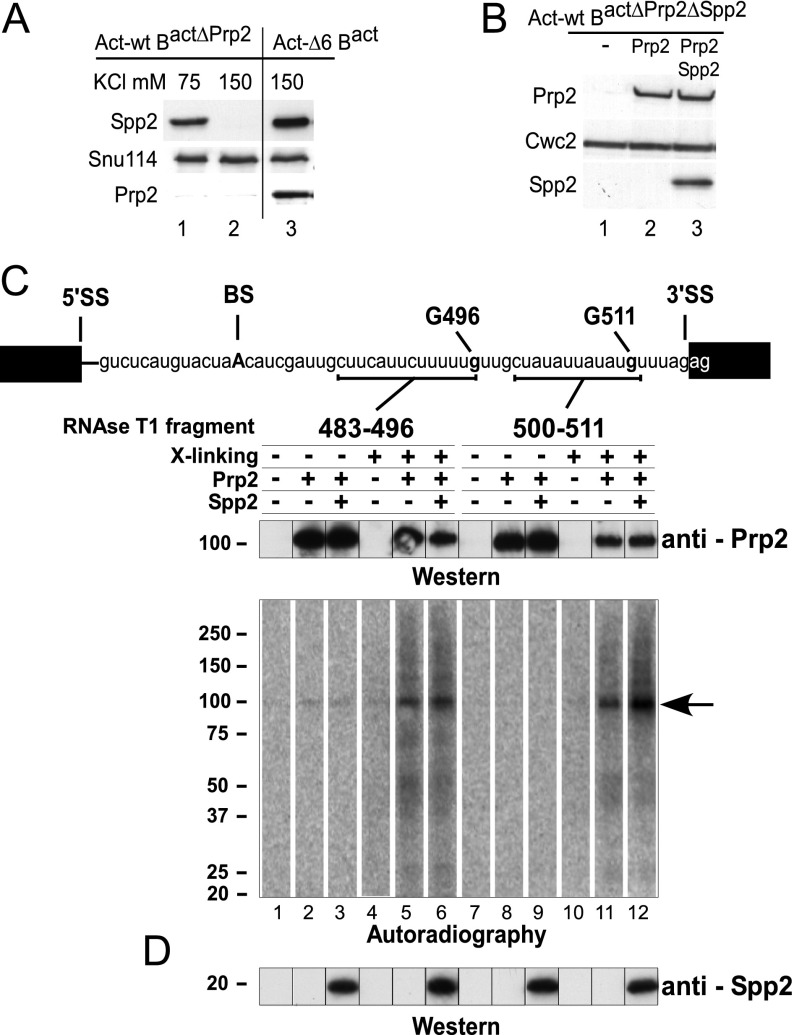
Prp2 and Spp2 bind to the spliceosome independently. (A) Western blot analysis of Spp2 association with the spliceosome. Spliceosomes were assembled on wild-type (lanes 1,2) or truncated (ActΔ6) actin pre-mRNAs (which leads to stalling of the spliceosome assembly at the Bact stage) (lane 3) in heat-treated prp2-1 extract (lanes 1,2) or wild-type extract (lane 3). Spliceosomes were affinity-purified in parallel in the presence of 75 mM (lane 1) or 150 mM (lanes 2,3) KCl. Immunoblotting was performed with rabbit polyclonal antibodies against GST-Spp2, Prp2, and Snu114 as indicated. (B) Prp2 and Spp2 association with the spliceosome at 150 mM KCl. The Bact ΔPrp2 ΔSpp2 spliceosomes were incubated with buffer (lane 1), Prp2 (lane 2), or Prp2 and Spp2 (lane 3). Unbound proteins were removed by washing with buffer containing 150 mM KCl, and spliceosomes were fractionated by glycerol gradient centrifugation at 150 mM KCl. Immunoblotting was performed with rabbit polyclonal antibodies against Prp2, Spp2, and Cwc2. (C) Cross-linking of Prp2 to pre-mRNA in purified spliceosomes. Two distinct site-specifically labeled pre-mRNAs were created, each carrying a single 32P-labeled phosphate 5′ at nucleotide G 496 or G 511. The theoretical RNA fragments remaining after digestion with RNase T1 are indicated by a bar below the sequence. Bact ΔPrp2 ΔSpp2 spliceosomes were assembled on site-specifically modified actin pre-mRNAs and purified at 150 mM KCl. After incubating with Prp2 or Prp2 plus Spp2, the complexes were UV cross-linked and then digested with RNase T1. Proteins cross-linked to 32P-labeled RNA were precipitated with antibodies against Prp2 and analyzed by SDS PAGE. Cross-linked 32P-labeled proteins were visualized by Western blot analysis (top panel) and autoradiography (bottom panel) as described above. The arrow indicates the 32P-labeled RNA cross-linked to Prp2. (D) Bact spliceosomes before cross-linking were probed with antibodies against Spp2.