Figure 8.
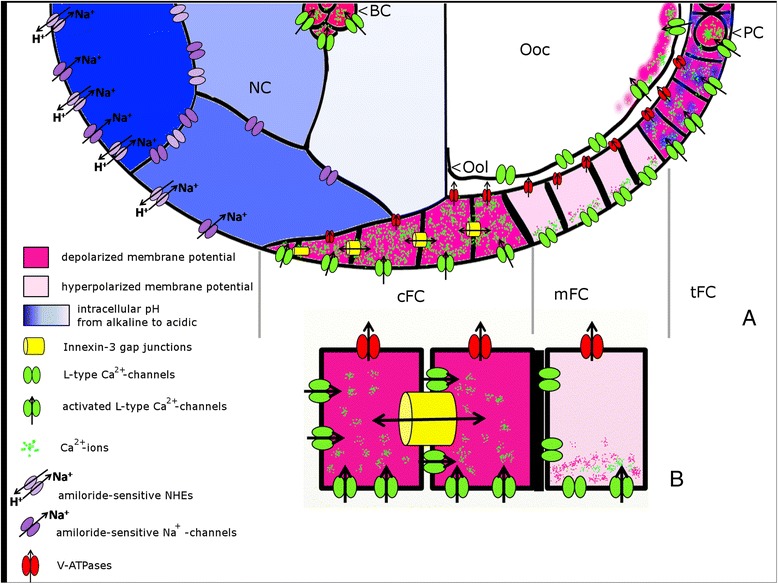
Summary of V mem -, pH i - and ion-transport patterns analysed in the present study. S9-follicles (for example) are characterized through complex patterns of Vmem, pHi and distribution or activity of ion-transport mechanisms (for abbreviations, see Figure 1). A: Within NC, an anteroposterior pHi-gradient, the anterior NC being more alkaline (dark blue) and the posterior NC being more acidic (light blue), was observed. A corresponding pattern was found for amiloride-sensitive Na+-transporters. Presumably, the pHi-grandient is either generated by asymmetrically distributed NHE, with higher concentrations in anterior NC, or the pHi-gradient influences the activity of amiloride-sensitive Na+-channels and NHE. The FC-epithelium is subdivided into cFC, mFC, tFC, PC and BC (the sFC are not shown). L-type Ca2+-channels as well as V-ATPases are equally distributed in the membranes of cFC, mFC and tFC. V-ATPase concentration is higher on the ventral side. Depolarized regions (cFC, tFC, PC and BC) are characterized through higher Ca2+-channel activity. Since inwardly directed Ca2+-currents through L-type Ca2+-channels depend on Vmem, we propose a mechanism by which alterations of Vmem can be translated into cellular responses through voltage-dependent Ca2+-currents. Within each mFC, we found an apicobasal potential gradient (the basal side being more depolarized), as well as an apicobasal gradient of Ca2+-channel activity. Innexin-3-containing gap junctions, which are enriched in lateral membranes of cFC, are supposed to regulate Vmem-, pHi- and ion distributions within cFC and to establish a communication border to the neighboring mFC. This border between cFC and mFC, the apicobasal Vmem-gradient within mFC, the specialized communication via innexin-3-containing gap junctions, and the supposed voltage-gated Ca2+-signaling are summarized in more detail in B. Despite a growing number of new results, there still exist various missing links between the bioelectric patterns [5-7], the ion-transport patterns [8,9,26,41,65-67] and the distributions of gap-junction proteins [12,46,50] described previously in Drosophila ovarian follicles.
