Figure 2. VEGFR2 is involved in VEGF-C-induced AKT but not ERK activation.
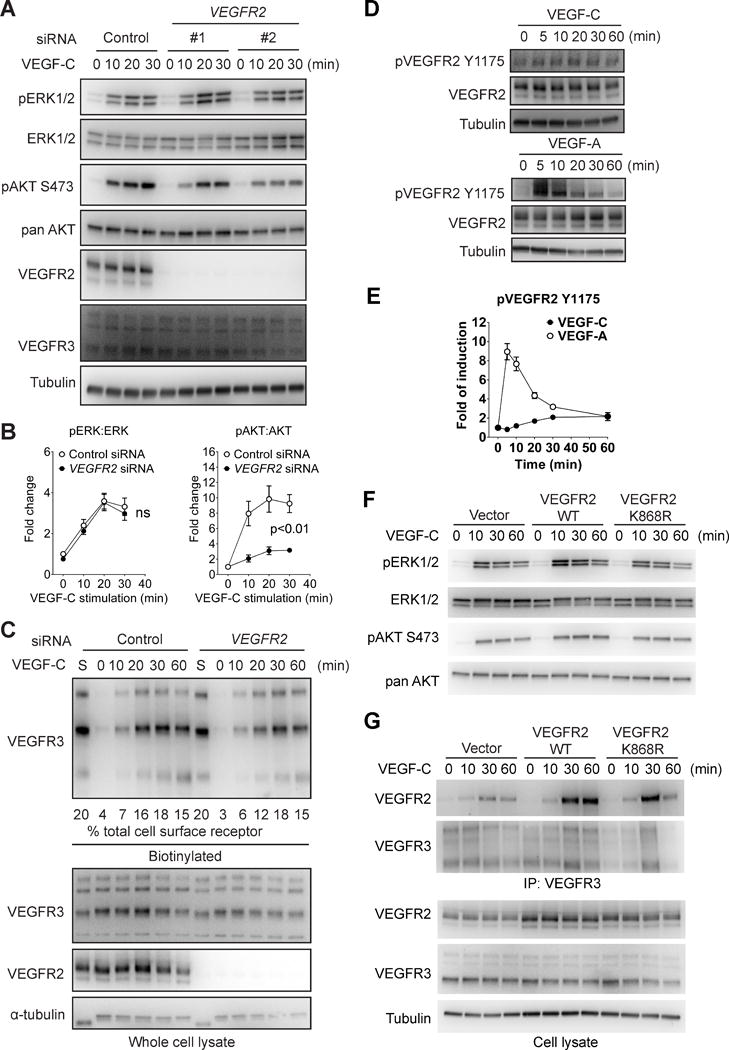
(A) HDLECs were transfected with two different siRNAs targeting human VEGFR2 or a negative control siRNA. The cells were then serum-starved and stimulated with 100 ng/ml VEGF-C. Activation of ERK1/2 and AKT was examined using Western blotting. (B) Quantitative analyses of ERK and AKT phosphorylation. Data represents Mean±SEM of three independent experiments with three distinct VEGFR2 siRNA sequences. Statistical analysis was performed using two way’s ANOVA. (C) HDLECs were biotin labeled as described in the Methods section and VEGF-C-induced VEGFR3 internalization in HDLECs treated with VEGFR2 or control siRNAs was determined by Western blotting of cell lysates at indicated time points (upper panel). Quantification was accomplished by deriving the percentage of internalized VEGFR3 to the amount of VEGFR3 present on the cell surface prior to VEGF-C treatment (lane “S”). Note that only 20% of lane S sample is loaded in the blot shown. Numbers under the blot refer to the % fraction of VEGFR3 internalized at various time points after VEGF-C stimulation. Note the absence of significant effect of VEGFR2 knockdown on VEGFR3 internalization. (D) Serum-starved HDLECs were stimulated with 100 ng/mL VEGF-C and VEGFR2 Y1175 phosphorylation was determined as shown. (E) Quantitative analyses of VEGFR2 Y1175 phosphorylation normalized to total VEGFR2 protein level. Data represents Mean±SEM of three independent experiments. (F) HDLECs infected with lentiviruses expressing either wild type VEGFR2, kinase-dead VEGFR2 mutant (K868R) or an empty control virus were stimulated with 100 ng/ml VEGF-C and activation of ERK and AKT was examined by Western blotting as indicated. (G) Upper panel: Western blot analysis of VEGFR3 antibody immunoprecipitate from HDLECs transduced with wild type or kinase-dead (K868R) VEGFR2 constructs or an empty virus control. Lower panel: Western blot analysis of total cell lysates corresponding to the upper panel.
