Abstract
Our preliminary studies confirmed that an active principle region of Buyang Huanwu decoction, comprising alkaloid, polysaccharide, aglycon, glucoside and volatile oil, can induce bone marrow mesenchymal stem cell differentiation into neurons. Mitogen-activated protein kinase signaling was identified as one of the key pathways underlying this differentiation process. The present study shows phosphorylated extracellular signal-regulated protein kinase and phosphorylated p38 protein expression was increased after differentiation. Cellular signaling pathway blocking agents, PD98059 and SB203580, inhibited extracellular signal-regulated protein kinase and p38 in mitogen-activated protein kinase signaling pathways respectively. mRNA and protein expression of the neuronal marker, neuron specific enolase, and neural stem cell marker, nestin, were decreased in bone marrow mesenchymal stem cells after treatment with the active principle region of Buyang Huanwu decoction. Experimental findings indicate that, extracellular signal-regulated protein kinase and p38 in mitogen-activated protein kinase signaling pathways participate in bone marrow mesenchymal stem cell differentiation into neuron-like cells, induced by the active principle region of Buyang Huanwu decoction.
Keywords: Buyang Huanwu decoction, bone marrow mesenchymal stem cells, extracellular signal-regulated protein kinase, mitogen-activated protein kinase signaling pathway, neuron specific enolase, nestin, cell signal transduction pathway, neural regeneration
Abbreviations
apr-BYHWD, active principle region of Buyang Huanwu decoction; BMSCs, bone marrow mesenchymal stem cells; MAPK, mitogen-activated protein kinase; ERK, extracellular signal-regulated protein kinase
INTRODUCTION
Buyang Huanwu decoction (BYHWD) is used to treat clinical sequelae following ischemic stroke and modern pharmacological research has demonstrated it to function in a multi-cause, multi-effect, multiple target manner. BYHWD acts to regulate the immune system, preventing inflammation and modulating lipid metabolism[1], dilating cerebral blood vessels[2], improving microcirculation and blood rheology[3], acting as an anti-coagulant and inhibiting thrombosis, reducing anti-free radicals and eliciting a neuroprotective mechanism[4]. Administration of BYHWD and transplantation of bone marrow mesenchymal stem cells (BMSCs) can enhance the survival rate, homing rate and differentiation rate of BMSCs[5,6,7,8]. Due to the complexity of traditional Chinese medicine prescriptions, we determined the median lethal dose of BYHWD using a sequential method and defined the optimal compatibility dosage using orthogonal design in a preliminary study. The active principle region of BYHWD (apr-BYHWD) consists of five components: alkaloid, polysaccharide, aglycon, glycoside and volatile oil[9]. We found the apr-BYHWD can induce BMSC differentiation into neuron-like cells in vitro, with a superior effect compared with BYHWD.
Many signal pathways contribute to the differentiation and development of neural cells, including Notch[10], Wnt[11], cAMP-PKA[12,13] and mitogen-activated protein kinase (MAPK) signaling pathways. In a study by Ma et al [14], neural differentiation of BMSCs was primarily achieved through the MAPK signaling pathway. To investigate further the signal transduction mechanism underlying apr-BYHWD induction of BMSCs neuronal differentiation, this study aimed to explore expression levels of phosphorylated extracellular signal-regulated protein kinase (ERK) and phosphorylated p38 protein in MAPK/ERK kinase (MEK)-ERK and p38MAPK pathways. Furthermore, the mRNA and protein expression levels of the neural cell marker, neuron specific enolase (NSE) and the neural stem cell marker, nestin were studied during the process of apr-BYHWD-induced BMSC differentiation, after MEK-ERK and p38MAPK pathways were blocked. This would provide us with a greater understanding of the role of p38MAPK and MEK-ERK pathways in the neural differentiation of BMSCs induced by apr-BYHWD.
RESULTS
Morphology of cultured primary BMSCs
Culture-expanded primary BMSCs were round with varying sizes, as observed using optical microscopy. Suspended cells concentrated together and began to adhere by 4–6 hours, with the majority of cells adhered by 24 hours. Non-adherent cells were removed at 48 hours after culture medium was replenished. Cells grew in clusters which gradually merged by 3-4 days. Cells had a short spindle or acicular morphology with visible nuclei. By days 7-10, cells had covered the bottom of the culture flask. After passage, cells were completely adherent within 24 hours. BMSCs at passage 3 were larger than the primary cells and the majority exhibited a spindle or fibrillar arrangement. Cell cytoplasm and nuclei were clear (Figure 1).
Figure 1.
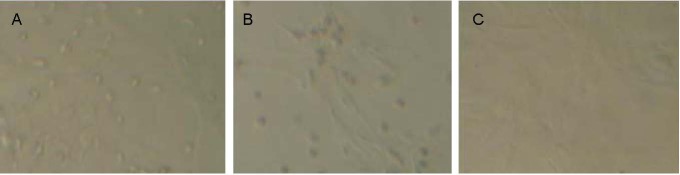
Rat bone marrow mesenchymal stem cells at primary, second and third passages (inverted phase contrast microscopy, × 100).
(A) Primary cells were round.
(B) Passage 2 cells formed colonies.
(C) Passage 3 cells had a fusiform or fibrillar arrangement.
Identification of BMSCs surface antigens
Flow cytometry was used to determine the cell surface antigen profile of BMSCs. Analysis showed expression of CD90 in more than 98% of BMSCs, while expression levels of CD11b and CD45 were less than 2% (Figure 2). This indicated the BMSC population has good uniformity and high purity.
Figure 2.
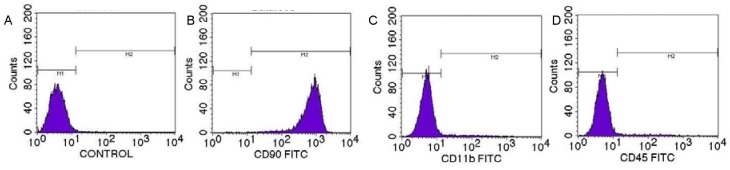
CD11b, CD45 and CD90 expression in rat bone marrow mesenchymal stem cells detected by flow cytometry.
(A) Blank control cells. (B-D) FITC labeled CD90, CD11 and CD45 positive cells respectively. FITC: Fluorescein isothiocyanate.
Phosphorylated protein ERK and p38 expression in BMSCs after induction with apr-BYHWD
BMSCs at passage 4 were divided into three groups: (1) apr-BYHWD group: BMSCs were incubated with culture medium containing 0.039 g/L apr-BYHWD; (2) negative control group: rat BMSCs were incubated with culture medium containing equivalent Dulbecco's Modified Eagle's Medium (DMEM); (3) normal control group: rat BMSCs were incubated with culture medium containing equivalent 10% fetal bovine serum.
Cells in the apr-BYHWD group were induced for 28 days. ERK protein levels were significantly higher in the apr-BYHWD group at day 28 compared with the blank control and negative control groups (P < 0.05; Table 1, Figure 3). At days 7, 14 and 28, p38 phosphorylated protein levels were significantly increased in the apr-BYHWD group compared with the blank control and negative control groups (P < 0.05; Table 1, Figure 4).
Table 1.
Western blot analysis of pERK and p-p38 protein expression in bone marrow mesenchymal stem cells induced by Buyang Huanwu decoction (BYHWD)

Figure 3.
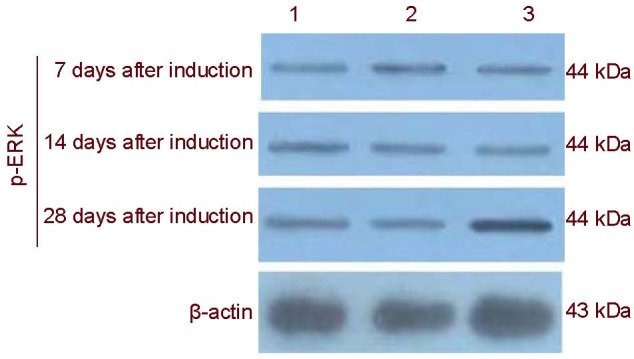
Electropherogram of phosphorylated extracellular signal-regulated protein kinase (p-ERK) protein expression in rat bone marrow mesenchymal stem cells induced with Buyang Huanwu decoction (BYHWD) detected by western blot analysis.
1: Blank control group; 2: negative control group; 3: apr-BYHWD group. In the apr-BYHWD group, phosphorylated extracellular signal-regulated protein kinase protein expression peaked at day 28. apr-BYHWD: Active principle region of BYHWD.
Figure 4.

Electropherogram of p38 phosphorylated protein (p-p38) expression in rat bone marrow mesenchymal stem cells induced with Buyang Huanwu decoction (BYHWD) detected by western blot analysis.
1: Blank control group; 2: negative control group; 3: apr-BYHWD group. In the apr-BYHWD group, p38 phosphorylated protein expression peaked at day 28. apr-BYHWD: Active principle region of BYHWD.
Influence of MAPK pathway blockers on NSE expression after BMSCs induction with apr-BYHWD
Reverse transcription-PCR (RT-PCR) confirmed NSE mRNA expression in the apr-BYHWD control group at days 7, 14 and 28. NSE mRNA expression in the apr-BYHWD + SB203580 group was significantly lower than those in the apr-BYHWD group at days 14 and 28 (P < 0.05; Figure 5, Table 2). At days 7, 14 and 28, NSE mRNA expression levels in the apr-BYHWD + PD98059 group were significantly lower than those in the apr-BYHWD group (P < 0.05; Figure 5, Table 2). Western blot analysis was used to detect expression of NSE protein in BMSCs after MAPK pathways were blocked. The results showed that NSE protein expression levels in the apr-BYHWD + SB203580 and apr-BYHWD + PD98059 groups, were lower than those in the apr-BYHWD group at days 7, 14 and 28 (P < 0.05; Figure 6, Table 2).
Figure 5.

Reverse transcription-PCR electropherogram of neuron specific enolase mRNA expression after the mitogen-activated protein kinase pathway was blocked.
1: apr-BYHWD group; 2: apr-BYHWD + SB203580 group; 3: apr-BYHWD + PD98059 group; M: Marker; BYHWD: Buyang Huanwu decoction; apr-BYHWD: active principle region of BYHWD; PD98059 and SB203580: cellular signaling pathway blocking agents.
Table 2.
Neuron specific enolase and nestin mRNA expression after apr-BYHWD induction, detected by reverse-transcription-PCR

Figure 6.
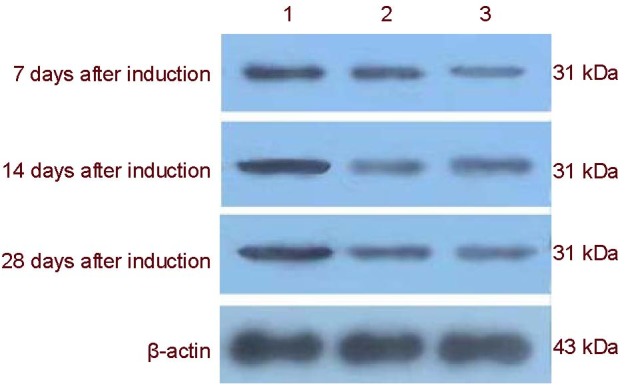
Western blot electropherogram of neuron specific enolase expression after the mitogen-activated protein kinase pathway was blocked.
1: apr-BYHWD group; 2: apr-BYHWD + SB203580 group; 3: apr-BYHWD + PD98059 group; BYHWD: Buyang Huanwu decoction; apr-BYHWD: active principle region of BYHWD; PD98059 and SB203580: cellular signaling pathway blocking agents.
Influence of MAPK pathway blockers on the nestin expression after BMSCs were induced with apr-BYHWD
Nestin mRNA expression was detected in the apr-BYHWD group at days 7, 14 and 28.
Low expression levels of nestin mRNA were detected in the apr-BYHWD + SB203580 group at day 28, and were significantly lower in comparison to the apr-BYHWD group (P < 0.05; Figure 7, Table 2).
Figure 7.
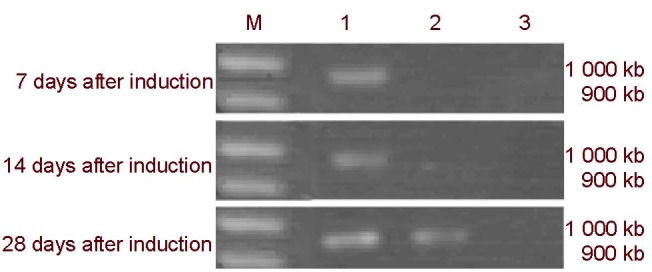
Reverse transcription-PCR electropherogram of nestin mRNA expression after the mitogen-activated protein kinase pathway was blocked.
1: apr-BYHWD group; 2: apr-BYHWD + SB203580 group; 3: apr-BYHWD + PD98059 group; M: Marker; BYHWD: Buyang Huanwu decoction; apr-BYHWD: active principle region of BYHWD; PD98059 and SB203580: cellular signaling pathway blocking agents.
Nestin mRNA expression was absent in the apr-BYHWD + PD98059 group at days 7, 14 and 28.
Western blot analysis was used to detect nestin protein expression in BMSCs after the MAPK pathway was blocked. Expression was absent in both the apr-BYHWD + SB203580 and apr-BYHWD + PD98059 groups at days 7 and 14. Low level nestin protein expression was detected at day 28 in both these groups, but was significantly lower compared to the apr-BYHWD group (P < 0.05; Figure 8, Table 3).
Figure 8.
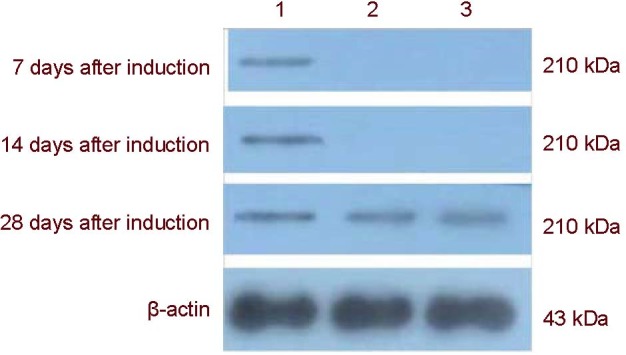
Western blot electropherogram of nestin protein expression after the mitogen-activated protein kinase pathway was blocked.
1: apr-BYHWD group; 2: apr-BYHWD + SB203580 group; 3: apr-BYHWD + PD98059 group. BYHWD: Buyang Huanwu decoction; apr-BYHWD: active principle region of BYHWD; PD98059 and SB203580: cellular signaling pathway blocking agents.
Table 3.
Neuron specific enolase and nestin expression after apr-BYHWD induction, detected by western blot analysis

DISCUSSION
Results from this study found that phosphorylated p38 expression in the MAPK pathway increased 7 days after apr-BYHWD induction in rat BMSCs, reaching a peak by 14 and 28 days. Phosphorylated ERK expression increased 28 days after induction. This provides evidence that apr-BYHWD can activate the MAPK pathway and that activation of p38MAPK pathway occurred earlier.
In this study, two cell signaling pathway inhibitors, PD98059 and SB203580, were used to block the MEK-ERK and p38MAPK signaling pathways respectively. Results demonstrated that at days 7, 14 and 28 after the MEK-ERK pathway was blocked, NSE gene and protein expression levels were decreased. After the p38MAPK pathway was blocked, NSE mRNA expression was decreased at days 14 and 28 and NSE protein expression was decreased at days 7, 14 and 28. Nestin mRNA and protein expression were absent at days 7 and 14, while low levels of expression were found at day 28. This confirms the MAPK pathway can be activated by apr-BYHWD and regulates NSE and nestin expression in BMSCs. Evidence suggests that the MEK-ERK and p38MAPK signaling pathways are involved in apr-BYHWD-induced BMSC differentiation. The drug concentration used for in vitro assessment was higher for the cells than would be found within serum or tissues, which brings bias for the experimental results and cannot explain the basis of BYHWD decoction. Further studies are required to conclusively determine the interactions of single components of BYHWD within this process.
In summary, MEK-ERK and p38MAPK signaling pathways are involved in apr-BYHWD-induced BMSC differentiation into neurons. Blocking MEK-ERK and p38MAPK signaling pathways can inhibit the effect of apr-BYHWD induction.
MATERIALS AND METHODS
Design
An in vitro experiment of cell induction and differentiation.
Time and setting
Experiments were performed from October 2011 to February 2012 in Ruikang Hospital Affiliated to Guangxi Traditional Chinese Medicine College, Level-3 Laboratory of Cell Molecular Biology of State Administration of Traditional Chinese Medicine of China.
Materials
Animals
Ten clean, male Sprague-Dawley rats, aged 6-8 weeks, weighing 130 ± 20 g, were provided by the Shanghai Experimental Animal Center (license No. SCXK (Hu) 2009-0004). All experimental disposals complied with the Guidance Suggestions for the Care and Use of Laboratory Animals, formulated by the Ministry of Science and Technology of China[15].
Drugs
BYHWD comprised 60 g Milkvetch Root, 6 g Red Peony Root, 6 g Szechwan Lovage Rhizome, 9 g Szechwan Lovage Rhizome, 9 g earthworm, 9 g peach seed and 9 g safflower. Herbs were purchased from the Department of Pharmacy, Affiliated Ruikang Hospital to Guangxi Traditional Chinese Medicine College and were identified by Chinese herbal medicine experts. The alcohol prescriptions were obtained via a water decoction and sedimentation method in the Drug Research and Development Basement, Affiliated Hospital of School of Pharmacy in Guangxi Traditional Chinese Medical College[16]. The effective components were independently extracted using the acidic precipitation and ion exchange resin chromatography method[17] and no confounding components were identified. Purity was greater than 70%, and the content of quality control substances was determined using high pressure liquid chromatography and a chemical analysis method according to the following standards[18]; content of ligustrazine was 9.76 mg/g alkaloids; content of astragaloside was 2.39 mg/g glycoside and content of amygdaloid was 17.9 mg/g glycoside. Volatile oil was emulsified with tween and the compatibility of crude drugs was prepared according to orthogonal design results[19], including 0.58 g alkaloid, 11.44 g polysaccharide, 8.74 g aglycone, 14.4 g glucoside and 2.5 g volatile oil. The concentration was adjusted with serum-free low-glucose-DMEM.
Methods
Isolation, culture and identification of BMSCs
Rats were euthanized following anesthesia and the tibia and femur were rapidly removed under sterile conditions. DMEM liquid was aspirated using an 8-gauge needle and the bone marrow cavity was rinsed repeatedly to extract and collect cells. BMSCs were cultured in DMEM (Gibco, New York, NY, USA) containing 10% fetal bovine serum (Hyclone, Logan, Utah, USA). Cells were counted and seeded onto 50-cm2 culture flaks at 1 × 109/mL, at 37°C under 5% CO2 and saturated humidity. The supernatant was removed 48 hours later and culture medium was replenished. Medium was changed every 2 days so hematopoietic cells in suspended growth were discarded, leaving the adherent BMSCs. On reaching 80% confluence, BMSCs were digested with 0.25% trypsin and collected. The aforementioned procedures were repeated to expand the BMSCs to passage 3. BMSCs at passage 3 were digested with 0.25% trypsin, prepared into a single cell suspension, blocked with 1% bovine serum albumin for 10 minutes and rinsed with PBS three times. Cells were then incubated with fluorescein isothiocyanate-labeled anti-CD11b, -CD45 and -CD90 antibodies (1:1 000; 0.5 mL; SERATEC, Gottingen, Germany). Cells in the control group were incubated with PBS at 4°C for 30 minutes and centrifuged at 1 500 r/min for 5 minutes. After PBS washing, cells were fixed with 1% paraformaldehyde, and surface antigen was detected using flow cytometry (FACScan BD, San Jose, CA, USA). In the high purity BMSC standard, the percentage of CD45 and CD11b positive cells was less than 5% and the CD90 positive rate was higher than 95%[20].
Phosphorylated protein ERK and p38 expression in apr-BYHWD-induced BMSCs
Total protein was extracted from BMSCs induced for 7, 14 and 28 days and protein concentrations were determined using the bicinchoninic acid method. Following sodium dodecyl sulfate-polyacrylamide gel and membrane transfer, the membranes were incubated with monoclonal rabbit anti-rat phosphorylated nestin, phosphorylated NSE and β-actin antibodies (1:300; Santa Cruz Biotechnology, Santa Cruz, CA, USA) overnight, followed by horseradish peroxidase-labeled goat anti-rabbit IgG (1:3 000; Santa Cruz Biotechnology) at room temperature for 2 hours. Following enhanced chemiluminescence for 1-10 minutes, results were detected. Absorbance was analyzed using Image-Pro Plus 6.0 (Media Cybernetics, Bethesda, MD, USA). Target protein expression was represented by absorbance ratio of target protein to β-actin protein (Santa Cruz Biotechnology).
Influence of MAPK signal pathway on apr-BYHWD-induced differentiation
BMSCs at passage 4 were randomly divided into three groups; apr-BYHWD group, apr-BYHWD + PD98059 group, and apr-BYHWD + SB203580 group.
(1) apr-BYHWD group: BMSCs were cultured with 3 mL medium containing apr-BYHWD (0.39%).
(2) apr-BYHWD + PD98059 group: BMSCs were cultured with 3 mL medium containing apr-BYHWD (0.39%) and 30 μL of 1 000 μM PD98059 (MEK-ERK signaling pathway inhibitor; Sigma, St. Louis, MO, USA). The final concentration of PD98059 was 10 μM.
(3) apr-BYHWD + SB203580 group: BMSCs were cultured with 3 mL medium containing apr-BYHWD (0.39%) and 30 μL of 1 500 μM SB203580 (p38MAPK signaling pathway inhibitor; Sigma). The final concentration of SB203580 was 15 μM.
Cells in all three groups were incubated for 24 hours, culture medium was removed and cells were rinsed twice with DMEM. Cells were then cultured with respective media (see above), which was replenished every 3 days. Cells were analyzed at days 7, 14 and 28 after induction of differentiation.
Nestin and NSE mRNA expression in BMSCs detected by RT-PCR
PCR primer sequences and amplified fragments are shown below:

At each specific time point after induction, BMSCs were harvested with 0.25% trypsin and prepared into 1 × 107/mL single cell suspension. Cells were exposed to Trizol (Invitrogen, Carlsbad, CA, USA) and total RNA was extracted. RNA integrity was determined through electrophoresis and RNA concentration from the absorbance at 260 and 280 nm using a DU640 protein nucleic acid analyzer (Beckman, Salt Lake City, UT, USA). Beta-actin served as the internal reference. According to primer sequences published in the NCBI Genbank, primers were designed using Primer Premier 5.0 (Premier Biosoft, Palo Alto, CA, USA) for Blast verification and synthesized by Invitrogen. RT-PCR was performed using a two-step method[21]. Samples were reversely transcribed using a gradient PCR instrument (Bio-Rad, Hercules, CA, USA) to 65°C for 5 minutes and to 42°C for 60 minutes. The cDNA was synthesized and denatured at 70°C for 5 minutes, then PCR was performed in 35 cycles (nestin: 94°C for 30 seconds, 54°C for 1 minute and 72°C for 1 minute; NSE: 94°C for 30 seconds, 56°C for 1 minute and 72°C for 1 minute), followed by 72°C extension for 10 minutes. Subsequent to PCR, cells were amplified with 2% agarose gel electrophoresis. The ratio of nestin NSE and beta-actin mRNA absorbance was measured using a gel imaging system (UVItec, London, United Kingdom). The experiment was repeated three times.
Western blot detection of phosphorylated ERK, phosphorylation p38, nestin and NSE protein expression
Total protein was extracted from BMSCs induced for different time points and protein concentration was determined with the BCA method[22]. Briefly, SDS-PAGE electrophoresis was carried out prior to and transfer onto a membrane. Rabbit anti-rat p-ERK, p-p38, nestin and NSE monoclonal antibodies (Santa Cruz Biotechnology) were diluted with TBST (1:300) and were exposed to the membrane with overnight incubation in a hybridization bag. After washes, membranes were exposed to HRP labeled goat anti-rabbit IgG (Santa Cruz Biotechnology; 1:3 000) at room temperature for additional 2 hours. ECL color and X-ray exposure were then carried out for 1-10 minutes in a darkroom[23]. The accumulated absorbance value was analyzed using a gel imaging system and Image-Pro Plus 6.0 (Media Cybernetics).
Statistical analysis
Quantitative data were expressed as mean ± SD and statistically analyzed with SPSS 17.0 software (SPSS, Chicago, IL, USA). Differences between groups were compared using one-way analysis of variance and mean differences between groups were compared with the paired least significant difference t-test. A P < 0.05 value was considered a significant difference.
Footnotes
Funding: The study was sponsored by the National Natural Science Foundation of China, No. 81102595; the Natural Science Foundation of Guangxi, No. 2012GXNSFAA053113.
Conflicts of interest: None declared.
Ethical approval: The pilot study was approved by the Animal Ethics Committee of the Guangxi Traditional Chinese Medical College in China.
(Edited by Yang XF, Chen X/Yang Y/Wang L)
REFERENCES
- [1].Liu Y, Lin R, Shi X, et al. The roles of buyang huanwu decoction in anti-inflammation, antioxidation and regulation of lipid metabolism in rats with myocardial ischemia. Evid Based Complement Alternat Med 2011. 2011 doi: 10.1093/ecam/neq028. 561396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [2].Zhao YN, Wu XG, Li JM, et al. Effect of BuYangHuanWu recipe on cerebral microcirculation in gerbils with ischemia-reperfusion. Sichuan Da Xue Xue Bao Yi Xue Ban. 2010;41(1):53–56. [PubMed] [Google Scholar]
- [3].Wang WR, Lin R, Zhang H, et al. The effects of Buyang Huanwu Decoction on hemorheological disorders and energy metabolism in rats with coronary heart disease. J Ethnopharmacol. 2011;137(1):214–220. doi: 10.1016/j.jep.2011.05.008. [DOI] [PubMed] [Google Scholar]
- [4].Wang L, Jiang DM. Neuroprotective effect of Buyang Huanwu Decoction on spinal ischemia/reperfusion injury in rats. J Ethnopharmacol. 2009;124(2):219–223. doi: 10.1016/j.jep.2009.04.045. [DOI] [PubMed] [Google Scholar]
- [5].Trzaska KA, Rameshwar P. Dopaminergic neuronal differentiation protocol for human mesenchymal stem cells. Methods Mol Biol. 2011;698:295–303. doi: 10.1007/978-1-60761-999-4_22. [DOI] [PubMed] [Google Scholar]
- [6].Yang Q, Mu J, Li Q, et al. A simple and efficient method for deriving neurospheres from bone marrow stromal cells. Biochem Biophys Res Commun. 2008;372(4):520–524. doi: 10.1016/j.bbrc.2008.05.039. [DOI] [PubMed] [Google Scholar]
- [7].Zhang YK, Han XY, Che ZY. Effects of buyang huanwu tang combined with bone marrow mesenchymal stem cell transplantation on the expression of VEGF and Ki-67 in the brain tissue of the cerebral ischemia-reperfusion model rat. J Tradit Chin Med. 2010;30(4):278–282. doi: 10.1016/s0254-6272(10)60056-8. [DOI] [PubMed] [Google Scholar]
- [8].Zhang YK, Yang GH. Effect of Buyang Huanwu decoction combined with mesenchymal stem cells transplantation on the expression of bFGF mrna in cerebral ischemia-reperfusion injury rats. Zhongyi Zazhi. 2011;52(7):589–591. [Google Scholar]
- [9].Deng CQ, Tang YH, He FY. Effects of each available composition and the combination of Buyanghuanwu decoction on cerebral ischemia in mice. Hunan Zhongyi Xueyuan Xuebao. 1999;19(4):1–4. [Google Scholar]
- [10].Alexson TO, Hitoshi S, Coles BL, et al. Notch signaling is required to maintain all neural stem cell populations-irrespective of spatial or temporal niche. Dev Neurosci. 2006;28(1-2):34–48. doi: 10.1159/000090751. [DOI] [PubMed] [Google Scholar]
- [11].Clark PA, Treisman DM, Ebben J, et al. Developmental signaling pathways in brain tumor-derived stem-like cells. Dev Dyn. 2007;236(12):3297–3308. doi: 10.1002/dvdy.21381. [DOI] [PubMed] [Google Scholar]
- [12].Jori FP, Napolitano MA, Melone MAB, et al. Molecular pathways involved in neural in vitro differentiation of marrow stromal stem cells. J Cell Biochem. 2005;94(4):645–655. doi: 10.1002/jcb.20315. [DOI] [PubMed] [Google Scholar]
- [13].Chu MS, Chang CF, Yang CC, et al. Signalling pathway in the induction of neurite outgrowth in human mesenchymal stem cells. Cell Signal. 2006;18(4):519–530. doi: 10.1016/j.cellsig.2005.05.018. [DOI] [PubMed] [Google Scholar]
- [14].Ma QY, Wen Y, Gu P, et al. The signal transduction pathway of NSCs’ differentiation induced by BMSCs conditioned medium. Xi Bao Sheng Wu Xue Za Zhi. 2009;31(3):424–428. [Google Scholar]
- [15].The Ministry of Science and Technology of the People's Republic of China. Guidance Suggestions for the Care and Use of Laboratory Animals. 2006-09-30 [Google Scholar]
- [16].Yuan J, Li YR, Chen Y, et al. Optimization of alcohol precipitation process for extract of Carthamus tinctorius by multiple guidelines grading method. Zhejiang Da Xue Xue Bao Yi Xue Ban. 2011;40(1):27–32. doi: 10.3785/j.issn.1008-9292.2011.01.006. [DOI] [PubMed] [Google Scholar]
- [17].Ma G, Chang X, Gong B, et al. Ligand exchange chromatographic separation of DL-amino acids based on monodisperse resin-bonded chiral stationary phase. Se Pu. 2007;25(5):723–727. [PubMed] [Google Scholar]
- [18].Eugster PJ, Guillarme D, Rudaz S, et al. Ultra high pressure liquid chromatography for crude plant extract profiling. J AOAC Int. 2011;94(1):51–70. [PubMed] [Google Scholar]
- [19].Deng CQ, Tang YH, He FY. Effects of each available composition and the combination of Buyanghuanwu decoction on cerebral ischemia in mice. Hunan Zhongyi Xueyuan Xuebao. 1999;19(4):1–4. [Google Scholar]
- [20].Chamberlain G, Fox J, Ashton B, et al. Concise review: mesenchymal stem cells: their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells. 2007;25(11):2739–2749. doi: 10.1634/stemcells.2007-0197. [DOI] [PubMed] [Google Scholar]
- [21].Zieglschmid V, Hollmann C, Gutierrez B, et al. Combination of immunomagnetic enrichment with multiplex RT-PCR analysis for the detection of disseminated tumor cells. Anticancer Res. 2005;25(3A):1803–1810. [PubMed] [Google Scholar]
- [22].Charbonneau DM, Meddeb-Mouelhi F, Beauregard M. A novel thermostable carboxylesterase from Geobacillus thermodenitrificans: evidence for a new carboxylesterase family. J Biochem. 2010;148(3):299–308. doi: 10.1093/jb/mvq064. [DOI] [PubMed] [Google Scholar]
- [23].You D, Cochain C, Loinard C, et al. Hypertension impairs postnatal vasculogenesis: role of antihypertensive agents. Hypertension. 2008;51(6):1537–1544. doi: 10.1161/HYPERTENSIONAHA.107.109066. [DOI] [PubMed] [Google Scholar]


