Abstract
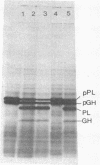
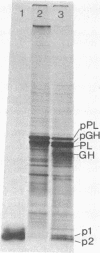
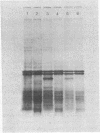
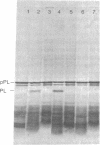
The protease(s) responsible for removing the amino-terminal extension of nascent presecretory proteins (signal peptidase) has been extracted from rough microsomes of dog pancreas with the detergent sodium deoxycholate. Preprolactin and pre-growth-hormone, prepared by in vitro translation of bovine pituitary RNA in the wheat germ system, were used to assay signal peptidase in the extract. When added to the wheat germ system during translation, the extract reduced the size of preprolactin and pre-growth-hormone to that of prolactin and growth hormone, respectively. Post-translational addition of the extract also reduced the size of preprolactin and pre-growth-hormone to that of the authentic hormones. The prolactin produced by post-translational cleavage of radiolabeled preprolactin has been shown, by partial amino-terminal sequence analysis, to have the correct amino terminus. This post-translational assay has permitted the investigation of the subcellular localization of the enzyme. Sodium deoxycholate extracts of rough microsomes were active, whereas extracts of smooth microsomes were inactive. However, without detergent treatment, neither rough nor smooth microsomes were capable of cleaving preprolactin in the post-translational assay. From this we conclude that the signal peptidase activity is confined to the rough endoplasmic reticulum and is latent. Finally, we have detected two small peptides which we believe could be the signal peptides generated by the endoproteolytic cleavage of preprolactin and pre-growth-hormone by signal peptidase.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aviv H., Leder P. Purification of biologically active globin messenger RNA by chromatography on oligothymidylic acid-cellulose. Proc Natl Acad Sci U S A. 1972 Jun;69(6):1408–1412. doi: 10.1073/pnas.69.6.1408. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birken S., Smith D. L., Canfield R. E., Boime I. Partial amino acid sequence of human placental lactogen precursor and its mature hormone form produced by membrane-associated enzyme activity. Biochem Biophys Res Commun. 1977 Jan 10;74(1):106–112. doi: 10.1016/0006-291x(77)91381-x. [DOI] [PubMed] [Google Scholar]
- Blobel G., Dobberstein B. Transfer of proteins across membranes. I. Presence of proteolytically processed and unprocessed nascent immunoglobulin light chains on membrane-bound ribosomes of murine myeloma. J Cell Biol. 1975 Dec;67(3):835–851. doi: 10.1083/jcb.67.3.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blobel G., Dobberstein B. Transfer of proteins across membranes. II. Reconstitution of functional rough microsomes from heterologous components. J Cell Biol. 1975 Dec;67(3):852–862. doi: 10.1083/jcb.67.3.852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burstein Y., Schechter I. Amino acid sequence of the NH2-terminal extra piece segments of the precursors of mouse immunoglobulin lambda1-type and kappa-type light chains. Proc Natl Acad Sci U S A. 1977 Feb;74(2):716–720. doi: 10.1073/pnas.74.2.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan S. J., Keim P., Steiner D. F. Cell-free synthesis of rat preproinsulins: characterization and partial amino acid sequence determination. Proc Natl Acad Sci U S A. 1976 Jun;73(6):1964–1968. doi: 10.1073/pnas.73.6.1964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devillers-Thiery A., Kindt T., Scheele G., Blobel G. Homology in amino-terminal sequence of precursors to pancreatic secretory proteins. Proc Natl Acad Sci U S A. 1975 Dec;72(12):5016–5020. doi: 10.1073/pnas.72.12.5016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobberstein B., Blobel G. Functional interaction of plant ribosomes with animal microsomal membranes. Biochem Biophys Res Commun. 1977 Feb 21;74(4):1675–1682. doi: 10.1016/0006-291x(77)90637-4. [DOI] [PubMed] [Google Scholar]
- Kemper B., Habener J. F., Ernst M. D., Potts J. T., Jr, Rich A. Pre-proparathyroid hormone: analysis of radioactive tryptic peptides and amino acid sequence. Biochemistry. 1976 Jan 13;15(1):15–19. doi: 10.1021/bi00646a003. [DOI] [PubMed] [Google Scholar]
- Lingappa V. R., Devillers-Thiery A., Blobel G. Nascent prehormones are intermediates in the biosynthesis of authentic bovine pituitary growth hormone and prolactin. Proc Natl Acad Sci U S A. 1977 Jun;74(6):2432–2436. doi: 10.1073/pnas.74.6.2432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurer R. A., Gorski J., McKean D. J. Partial amino acid sequence of rat pre-prolactin. Biochem J. 1977 Jan 1;161(1):189–192. doi: 10.1042/bj1610189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roman R., Brooker J. D., Seal S. N., Marcus A. Inhibition of the transition of a 40 S ribosome-Met-tRNA-i-Met complex to an 80 S ribosome-Met-tRNA-i-Met- complex by 7-Methylguanosine-5'-phosphate. Nature. 1976 Mar 25;260(5549):359–360. doi: 10.1038/260359a0. [DOI] [PubMed] [Google Scholar]
- Shields D., Blobel G. Cell-free synthesis of fish preproinsulin, and processing by heterologous mammalian microsomal membranes. Proc Natl Acad Sci U S A. 1977 May;74(5):2059–2063. doi: 10.1073/pnas.74.5.2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strauss A. W., Donohue A. M., Bennett C. D., Rodkey J. A., Alberts A. W. Rat liver preproalbumin: in vitro synthesis and partial amino acid sequence. Proc Natl Acad Sci U S A. 1977 Apr;74(4):1358–1362. doi: 10.1073/pnas.74.4.1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swank R. T., Munkres K. D. Molecular weight analysis of oligopeptides by electrophoresis in polyacrylamide gel with sodium dodecyl sulfate. Anal Biochem. 1971 Feb;39(2):462–477. doi: 10.1016/0003-2697(71)90436-2. [DOI] [PubMed] [Google Scholar]
- Yu S., Redman C. In vitro synthesis of rat pre-proalbumin. Biochem Biophys Res Commun. 1976 May 23;76(2):469–476. doi: 10.1016/0006-291x(77)90748-3. [DOI] [PubMed] [Google Scholar]