ABSTRACT
Replication-deficient rabies virus (RABV)-based vaccines induce rapid and potent antibody responses via T cell-independent and T cell-dependent mechanisms. To further investigate early events in vaccine-induced antibody responses against RABV infections, we studied the role of macrophages as mediators of RABV-based vaccine immunogenicity. In this report, we show that a recombinant matrix gene-deleted RABV-based vaccine (rRABV-ΔM) infects and activates primary murine macrophages in vitro. Immunization of mice with live RABV-based vaccines results in accumulation of macrophages at the site of immunization, which suggests that macrophages in tissues support the development of effective anti-RABV B cell responses. However, we show that draining lymph node macrophages, but not macrophages at the site of immunization, are essential for the generation of germinal center B cells, follicular T helper cells, and RABV-specific antibodies. Our findings have implications for the design of new RABV-based vaccines for which early immunological events are important for the protection against RABV in postexposure settings.
IMPORTANCE More than two-thirds of the world's population live in regions where rabies is endemic. Postexposure prophylaxis is the primary means of treating humans. Identifying immunological principles that guide the development of rapid and potent antibody responses against rabies infections will greatly increase our ability to produce more-effective rabies vaccines. Here we report that macrophages in the draining lymph node, but not in the tissue at the site of immunization are important for vaccine-induced antibody responses to rabies. Information gleaned from this study may help guide the development of a single-dose vaccine against rabies infections.
INTRODUCTION
Rabies is a zoonotic viral encephalitis responsible for more than 55,000 human deaths annually (1). Rabies exposure is treated with postexposure prophylaxis (PEP), consisting of antirabies immunoglobulin and inactivated rabies virus (RABV) vaccine over the course of several weeks (2). While safe and effective, this protocol is costly and cumbersome for use in developing countries where rabies remains endemic (3).
Our lab has developed a live replication-deficient RABV-based vaccine, rRABV-ΔM, which lacks the matrix (M) gene (4). A single immunization with rRABV-ΔM quickly induces high titers of protective anti-RABV antibodies in mice, dogs, and nonhuman primates (4, 5). We have also used rRABV-ΔM to delineate host anti-RABV immunity, demonstrating that rRABV-ΔM induces both a potent germinal center (GC) response and a T cell-independent antibody response (6) that includes early titers of anti-RABV IgM that contribute to host protection (7). rRABV-ΔM also infects and activates primary B cells in vitro, inducing an antigen-presenting cell (APC) phenotype (8, 9).
Macrophages are phagocytic cells of the innate immune system that mediate a range of physiologic and immune functions and are ubiquitous within peripheral tissues and secondary lymphoid organs (10, 11). Resident tissue macrophages are abundant in muscle as well as the dermis, where they perform vital immune surveillance and muscle/connective tissue remodeling functions (10). These macrophages are derived from embryonic precursor cells, and under normal homeostasis, they undergo a low level of replication to sustain the population without the need for replenishment from bloodstream monocytes. Upon mechanical injury or microbial infection, fibroblasts and resident immune cells release chemokines such as CCL2 (chemokine C-C motif ligand 2) that recruits monocytes from the circulating blood to the site of damage (11, 12). Depending on the balance of chemical signals such as conserved pathogen markers (lipopolysaccharide and viral RNA/DNA), inflammatory cytokines (alpha/beta interferon [IFN-α/β] and interleukin 1 [IL-1]), and tissue growth factors (fibroblast growth factor [FGF] and vascular endothelial growth factor [VEGF]), activated resident and recruited macrophages can be polarized into the broadly characterized proinflammatory M1 phenotype or the wound-healing M2 phenotype (12–15). M1 polarization is characterized by inducible expression of proinflammatory cytokines such as tumor necrosis factor alpha (TNF-α) and activation toward potent phagocytosis and an antigen-presenting phenotype, which may induce migration of activated macrophages to the draining lymph node (LN) to participate in the development of the adaptive antibody and T-helper responses (11, 16). M2 polarization is characterized by induction of arginase and inducible nitric oxide synthase (iNOS) intracellular expression and activation toward anti-inflammatory functions that promote tissue regeneration and remodeling by fibroblasts (16). While M1 type activation may directly contribute to the antibody response in the draining LN, M2 activation can be physiologically important in the repair of tissues providing physical barriers to further infection (11, 13, 14, 16).
Resident macrophages of the LN include medullary macrophages in the medullary cords of the LN and subcapsular sinus (SCS) macrophages, which line the surface of the follicle, facing the antigen-rich lymph-filled outer sinus of the efferent lymphatic drainage system (11). While medullary macrophages serve to phagocytose and degrade excess particulate antigens that have flowed through the LN cortex, the subcapsular sinus macrophages are of particular interest in the early LN immune response as they receive first contact with lymph-borne antigen reaching the LN from the site of immunization (11). Possessing a lower phagocytic rate than medullary and other tissue macrophages, macrophages lining the subcapsular sinus are specialized for the trapping and translocation of lymph-borne antigens from the sinus lumen to the B cell-rich follicles underlying the basal surface (11, 17). As such, they have been found to be critical in mouse models for the trapping of vesicular stomatitis virus (VSV) in the draining LN and presentation of viral antigens to B cells (18). Their depletion results in the death of the mouse from what is in wild-type mice a nonlethal dose of VSV due to the systemic spread of virus from the skin inoculation site (19).
Previous studies have demonstrated that bone marrow-derived macrophage cell lines are infected by attenuated RABV in vitro. RABV establishes a productive, long-term in vitro infection in murine bone marrow-derived monocytes, which stimulates the development of an antiviral state in the cells and expression of markers of antigen presentation such as CD86 (20, 21). However, no studies have examined the response of primary macrophages to RABV or the role macrophages play in B cell responses to RABV vaccination in vivo.
The susceptibility of bone marrow-derived and cell line macrophages to RABV infection, coupled with their varied and important functions at both the sites of intramuscular (i.m.) or intradermal (i.d.) immunization and the draining LN, makes macrophages a cell type likely to be implicated in promoting the generation of a protective response to RABV-based vaccines. We hypothesized that LN macrophage subsets are significant contributors to the early and potent antibody response observed with rRABV-ΔM due to their proximity to B cell zones and their ability to influence B cell antigen exposure. We aimed to test this hypothesis using the well-described clodronate-loaded liposome (CLL) macrophage depletion technique. Liposomal encapsulation of clodronate targets the drug to macrophage populations, inducing rapid and highly specific apoptosis while leaving other immune cell types, including dendritic cells, intact (22). This protocol enables site-specific transient depletion of macrophage subsets, which we used to target immunization site and/or LN-resident macrophages during the early stages of the immune response to rRABV-ΔM immunization. Using this method, we show that macrophages in the LN, but not in the immunization site tissue, are essential to the development of germinal center B cells, follicular T helper cells, and anti-RABV antibody responses.
MATERIALS AND METHODS
In vitro macrophage infections and flow cytometry.
Spleens were collected from 6- to 10-week-old C57BL/6 mice (NCI), homogenized, and cultured in RPMI-based splenocyte medium (9) for 2 days with rRABV-ΔM or UV-inactivated rRABV (UV-rRABV) at a multiplicity of infection (MOI) of 5 or mock infected (8). Cells were harvested, blocked with CD16/32 Fc block (BD Biosciences) in phosphate-buffered saline (PBS) buffer containing 2% fetal bovine serum (FBS) (fluorescence-activated cell sorting [FACS] buffer), and then stained for markers, including RABV nucleoprotein (RABV-N)–fluorescein isothiocyanate (FITC) (Fujirebio Diagnostics), F4/80-peridinin chlorophyll protein (PerCP)-Cy5.5, CD11b-allophycocyanin, major histocompatibility complex class II (MHCII)-Alexa Fluor 700, and CD80-phycoerythrin (PE). Samples were fixed in 3% paraformaldehyde and immediately analyzed on a BD LSRII flow cytometer. Data were analyzed using FlowJo (Treesoft) and Prism 5 (Graphpad) software.
Mouse immunizations and tissue macrophage staining.
Six- to 10-week-old female C57BL/6 mice were immunized in the hind leg medial quadriceps or hind leg footpads with 107 focus-forming units (FFU) of rRABV-ΔM or PBS. Two days later, muscle and footpad samples were collected on ice, minced into small pieces, and digested with Pronase (Calbiochem) (1,000 U/ml) for 1 h at 37°C, with trituration every 15 min to aid digestion. Digested tissue was filtered through a 100-μm cell strainer, and the filtrate was dissolved in 5 ml of 30% Percoll in PBS (Sigma), which was layered over 5 ml of 70% Percoll in PBS. Percoll gradients were centrifuged at 1,000 × g for 20 min, and cells at the 30%/70% Percoll interface were collected (protocol adapted from references 19 and 21). The cells were then stained for CD11b and F4/80 as described above, and samples were analyzed on a BD LSRII flow cytometer before being analyzed using FlowJo (Treesoft) and Prism 5 (Graphpad) software.
Macrophage depletion with clodronate liposomes.
Clodronate-loaded liposomes (CLLs) (Clodrosomes; Encapsula Nano Sciences) (5 mg/ml clodronate), or sham PBS injections, were administered to female C57BL/6 mice aged 6 to 10 weeks by intravenous (i.v.) retro-orbital (150 μl) and i.d. footpad (50 μl) routes on days −3, −1, +1, and +3 relative to immunization on day 0 with 103 FFU rRABV-ΔM or 50 μl PBS. Depletion was verified on days 0 to +4 by staining spleen, popliteal LN, or footpad skin samples for CD11b and F4/80, and samples were analyzed by flow cytometry as described above. Blood was collected by intraorbital puncture on days 5, 7, and 10 postimmunization, and serum antibodies to RABV glycoprotein (RABV-G) were measured by an enzyme-linked immunosorbent assay (ELISA) as described in reference 23. On day 10 postimmunization, draining popliteal LNs were collected, homogenized, and stained with B220-PerCP, Fas-PE-Cy7, GL7-FITC, CD3ε-Alexa Fluor 450, CD4-allophycocyanin, chemokine (C-X-C motif) receptor 5 (CXCR5)–PerCP–Cy5.5, and PD-1–PE as described in references 6 and 24. Samples were run on a BD LSRII flow cytometer and analyzed using FlowJo (Treesoft) and Prism 5 (Graphpad) software.
This work was approved and completed in compliance with Thomas Jefferson University Institutional Animal Care and Use Committee (IACUC).
RESULTS
Live RABV-based vaccines infect and activate primary murine macrophages.
As previously published by our research group (8, 9), a small population of non-B cells from primary cultures of mouse splenocytes are infected by RABV-based vaccines. To delineate whether primary murine macrophages are infected and activated by live RABV-based vaccines, naive mouse splenocytes were cultured in vitro for 2 days with rRABV-ΔM or UV-inactivated recombinant RABV (UV-rRABV) at a multiplicity of infection (MOI) of 5 or mock infected (8). Significant infection of primary macrophages (CD11bhi F4/80hi as described in reference 25) was observed with rRABV-ΔM compared to mock infection or UV-rRABV treatment (Fig. 1B and E), as determined by intracellular staining for RABV nucleoprotein (RABV-N). RABV-infected macrophages upregulated expression of MHCII (Fig. 1C and F) and, to a lesser but still significant extent, the costimulatory receptor CD80 (Fig. 1D and G), suggesting activation to an APC phenotype (12). Macrophages from splenocyte cultures containing UV-rRABV showed no significant elevation of RABV-N staining nor any significant upregulation of MHCII or CD80 compared to macrophages from mock-infected splenocyte cultures. In our in vitro infection system, splenic macrophages thus respond to live but not inactivated RABV.
FIG 1.
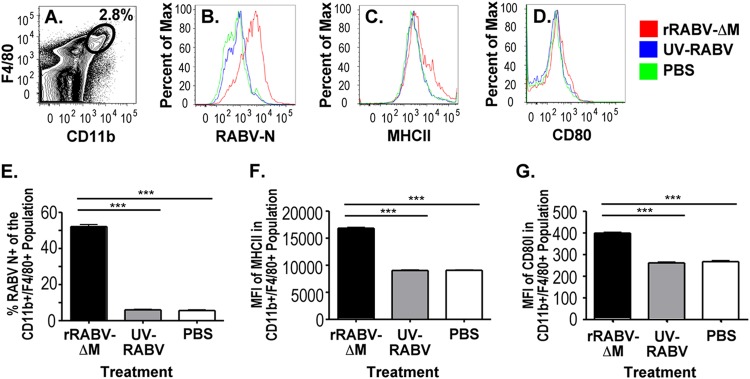
In vitro infection of primary mouse splenic macrophages by rRABV-ΔM. Primary murine splenocytes were infected for 2 days with rRABV-ΔM or UV-inactivated RABV (UV-RABV) at an MOI of 5 and then analyzed for macrophage infection and activation. (A) Example of CD11b+ F4/80+ macrophage population flow cytometry gating strategy in a sample of primary splenocytes. (B to D) Overlaid histograms of RABV nucleoprotein (RABV-N) (B), MHCII (C), and CD80 (D) staining. The percentage of the maximum is shown on the y axes in panels B to D. (E to G) Percent RABV-N-positive (RABV-N+) (E) and mean fluorescence intensity (MFI) of MHCII (F) and CD80 (G) expression of CD11b+ F4/80+ cells by in vitro treatment. There were six mice per group for all groups. Values that are significantly different (P < 0.001) by unpaired, two-tailed Student's t test are indicated by a bar and asterisks (***).
Live RABV-based vaccines induce the accumulation of macrophages at the site of immunization.
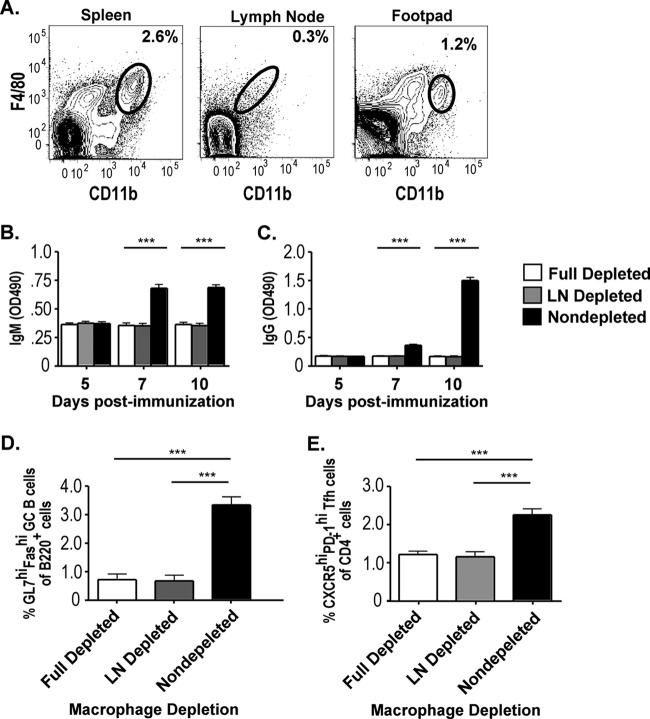
Macrophages are the most abundant type of immune cell present in muscle (14, 15), the most common site of RABV immunization, and are also present in the dermis of skin, which serves as an alternate site of immunization for RABV (2). Therefore, having demonstrated that rRABV-ΔM infects and activates primary macrophages in vitro, we examined the in vivo response of tissue macrophages to inoculation by rRABV-based vaccines. Two days following an intramuscular hind leg injection of 107 FFU of rRABV-ΔM or a mock immunization with 50 μl of PBS, muscle was collected and stained for CD11b and F4/80. Macrophages were more numerous in hind leg muscle immunized with rRABV-ΔM than in muscle given a sham immunization (Fig. 2A to C). Foreleg muscles of mice immunized in the hind leg showed no increase in macrophage populations (data not shown), suggesting a local effect of RABV at the immunization site. Repeating this protocol with hind footpad skin demonstrated a similar elevation of local dermal macrophage populations 2 days following intradermal rRABV-ΔM immunization (Fig. 2D).
FIG 2.

Immunization of mouse hind leg muscle and footpad skin with live RABV-based vaccines induces the accumulation of macrophages at the inoculation site. C57BL/6 mice were immunized intramuscularly or intradermally (footpad) with 107 FFU of rRABV-ΔM or mock immunized with PBS alone. Two days postimmunization, samples of muscle or footpad skin were collected and analyzed for the presence of macrophage population by flow cytometry analyses. (A and B) Representative contour plots of CD11b+ F4/80+ cells collected from processed muscle samples of mice infected with rRABV-ΔM (A) or mock infected with PBS (B). (C) Percent macrophage composition of processed muscle tissue from mice immunized with the indicated vector (n = 8). (D) Percent macrophage composition of processed footpad skin from mice immunized with rRABV-ΔM versus mice immunized with PBS (n = 3). Values that are significantly different (P < 0.001) by unpaired, two-tailed Student's t test are indicated by a bar and asterisks (***).
LN-resident, but not immunization site macrophages, are critical for anti-RABV B cell responses.
We next aimed to determine a functional role for these elevated macrophage populations at the immunization site compared to lymph node-resident macrophages, in driving the immune response to rRABV-based vaccines. Clodronate-loaded liposomes (CLLs) (22) were administered both retro-orbitally and intradermally, routes that allowed us to deplete systemic reservoir, draining LN, and immunization site macrophage populations from mice prior to immunization (13, 22, 26). Macrophage depletions were verified in spleen, popliteal LNs, and footpad on days 0 to +4 by verifying the specific absence of CD11bhi F4/80hi macrophages and the continued presence of CD11bhi CD11chi dendritic cells (Fig. 3A and data not shown). Sham-depleted control mice demonstrated robust germinal center (GC) B cell (Fig. 3B and C) and T follicular helper (Tfh) cell (Fig. 3D and E) responses and antibody responses (Fig. 4) to rRABV-ΔM immunization, responses that were ablated in mice depleted of macrophages, with the exception of a very limited, highly suppressed GC B cell response. Thus, macrophages play a role in mediating the response to rRABV immunization as demonstrated by the absence of an antibody response in mice systemically depleted of macrophages.
FIG 3.
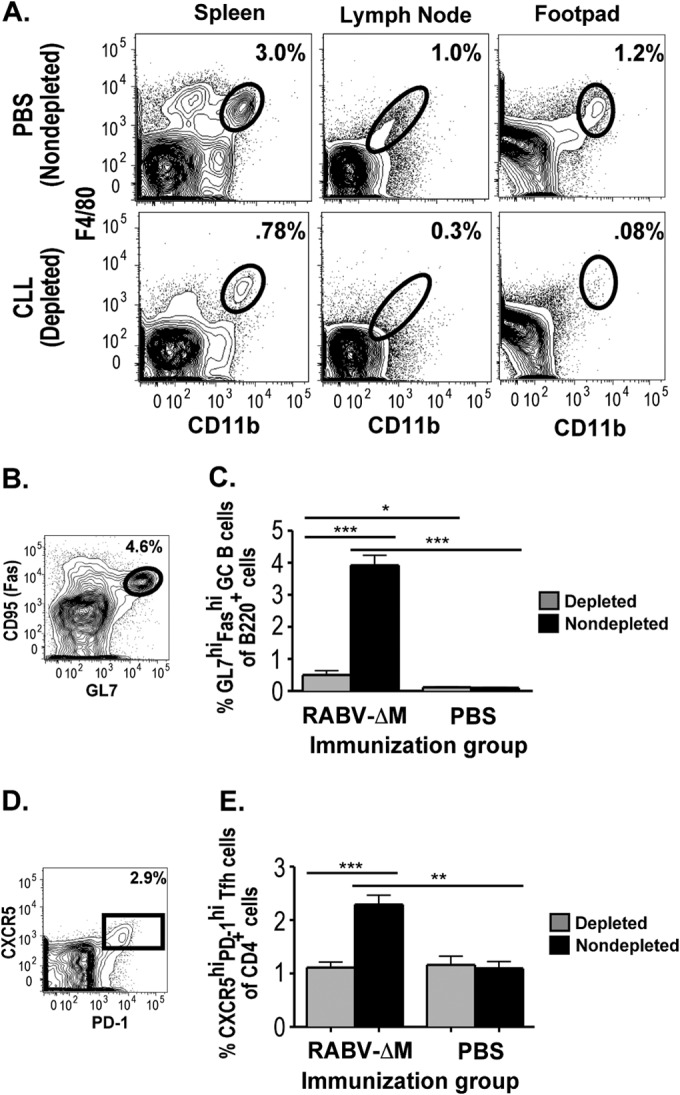
CLL-mediated depletion profoundly suppresses the development of GC B cells and Tfh cells in response to 103 FFU rRABV-ΔM. Mice were treated with clodronate-loaded liposomes (CLL) retro-orbitally and intradermally to deplete macrophages from systemic reservoirs, popliteal LNs, and footpad prior to immunization with 103 FFU rRABV-ΔM or mock immunized with PBS. (A) Representative contour plots demonstrating depletion of CD11b+ F4/80+ macrophages by CLL treatment in spleen, popliteal LNs, and footpad skin on day 1 postimmunization. (B) Representative gating strategies for popliteal LN GC B cells (B220+ Fashi GL7hi). (C) Percent popliteal LN GC B cells out of total B220+ B cells. (D) Representative gating strategy for popliteal LN Tfh cells (CD3ε+ CD4+ CXCR5hi PD-1hi). (E) Percent popliteal LN Tfh cells out of total CD4+ T cells in macrophage-depleted versus non-macrophage-depleted (nondepleted) mice (n = 8), immunized with either rRABV-ΔM or PBS. Values that are significantly different are indicated by a bar and asterisks as follows: *, P < 0.05; **, P < 0.01; ***, P < 0.001.
FIG 4.
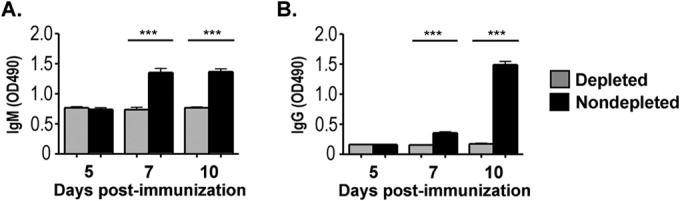
CLL-mediated depletion of macrophages eliminates the antibody responses to 103 FFU of rRABV-ΔM. Blood was collected as a source of serum from the mice described in the legend to Fig. 3. (A and B) Anti-RABV-G IgM (A) or IgG (B) antibody titers were measured by ELISA from macrophage-depleted versus non-macrophage-depleted mice immunized with rRABV-ΔM or PBS (n = 8). Values that are significantly different (P < 0.001) are indicated by a bar and asterisks (***).
The contributions of immunization site- and draining LN-based macrophages to vaccine-induced immunity can be separated by targeting CLL-mediated depletion to specific tissues (13). Female C57BL/6 mice aged 6 to 10 weeks were depleted of macrophages by CLL injections via an i.m. route (gastrocnemius, 50 μl) to target popliteal LN macrophages alone (26) or via i.v. plus i.d. routes (as in Fig. 3) to target footpad, popliteal LN, and systemic reservoir macrophage populations. The CLL treatment, immunization schedule, macrophage depletion verification, and sample collection and analysis were performed as described in the legend to Fig. 3, and the specific absence of macrophages from the popliteal lymph node was confirmed (Fig. 5A). “LN Depleted” mice with intact footpad and systemic macrophage populations but depleted of LN macrophages lacked GC B cell and Tfh responses, as well as RABV-specific antibodies, to i.d. immunization with 103 FFU of rRABV-ΔM, similar to “Full Depleted” mice lacking macrophages at all sites (Fig. 5). Therefore, draining LN macrophages are essential mediators of the immune response to intradermal footpad rRABV immunization, while macrophages at the immunization site are not sufficient to drive the early antibody and germinal center responses.
FIG 5.
LN macrophages are essential for the antibody and germinal center responses to 103 FFU of rRABV-ΔM. Mice were treated with CLLs. The CLLs were injected intramuscularly to deplete popliteal LN macrophages alone or intravenously and intradermally to target footpad, popliteal LN, and systemic reservoir macrophage populations. Immunization, macrophage depletion verification, and sample collection were performed as described in the legends to Fig. 3 and 4. (A) Representative gating strategy verifying depletion of macrophages in the LN but not in the spleen or footpad. (B) Levels of RABV-G-specific IgM from CLLs injected intramuscularly (LN depletion alone) versus CLLs injected intravenously and intradermally (footpad, LN, and systemic macrophage depletion) versus PBS-injected (non-macrophage-depleted) mice, immunized with rRABV-ΔM. (C) Levels of RABV-G-specific IgG. (D) Percent popliteal LN GC B cells out of total B220+ B cells on day 10 postimmunization. (E) Popliteal LN T follicular helper cells as a percentage of total CD4+ T cells on day 10 postimmunization. There were eight mice per group for all groups. Values that are significantly different (P < 0.001) are indicated by a bar and asterisks (***).
DISCUSSION
In this report, we demonstrated the in vitro infection and activation of macrophages by an rRABV-ΔM vaccine, as well as the role macrophages play in the in vivo response to rRABV-ΔM immunization. Although macrophages accumulate at the site of rRABV-ΔM immunization, we found that LN macrophages alone are essential for the early antibody and germinal center responses to rRABV immunization.
Splenic macrophages infected in vitro by rRABV-ΔM are activated toward an antigen presentation phenotype, as demonstrated by the elevation of the markers MHCII and CD80. These data are in line with our previous studies on in vitro infection of B cells, which displayed a similar activation marker profile upon infection with rRABV-ΔM. However, while B cells cultured with rRABV-UV in vitro (described in reference 8) displayed modest increases in RABV-N and activation marker staining, splenic macrophages cultured under the same conditions responded only to live rRABV infection. Although macrophages are more potent antigen-presenting cells than B cells, it is clear from our data that not all infected macrophages display elevated levels of MHCII and CD80 on day 2 of culture. Explanations for this include the possibility that by choosing a single time point to analyze, we miss activation marker upregulation on infected macrophages both before and after day 2 of in vitro culture. Our studies of infected B cells show that infection begins as early as day 1 of culture and continues to increase until at least day 4, while activation marker upregulation does not follow such a pattern of accumulation. Thus, it is possible that some of the infected macrophages we detect on day 2 include cells that have already downregulated or that have yet to upregulate activation marker expression in response to infection. On the basis of this data, we conclude that infected macrophages have the ability to become activated in the presence of live RABV-based vaccination.
Our in vivo studies provide clear demonstration of the essential role that LN macrophages play in the early response to rabies-based vaccines. The importance of mouse splenic macrophages to the GC response to keyhole limpet hemocyanin immunization was previously demonstrated (27), but limited investigation has been made into the role of immunization site tissue and LN macrophages in promoting effective viral immunization. With a high dose (106 FFU) of rRABV-ΔM, LN macrophage depletion impacted the strength of the GC response but not titers of RABV-specific antibodies (data not shown), perhaps due to alternative B cell activation and antibody production mechanisms that function only at higher antigen concentrations, implying a dose-dependent requirement for macrophages in RABV vaccine-induced immunity.
The interface between LN macrophages and B cells presents a particularly intriguing setting for a model of immune activation by RABV-based vaccine immunization. A layer of subcapsular sinus (SCS) macrophages coats the LN follicles, separating the B cell zone from the efferent lymph. SCS macrophages have been implicated as an important target for lymph-borne viruses and antigen (11, 17). The phagocytic capacity of SCS macrophages is less than connective tissue or medullary macrophage subsets, enabling antigen to be transported intact from the outer surface to the inner surface of the subcapsular lining, where it is presented to the B cell-rich follicle (11). This occurs via both internal and surface mechanisms that work in concert to polarize the SCS macrophage toward efficient presentation of lymph-borne antigens to B cells (19). Furthermore, cross talk between the SCS macrophages and B cells works to preserve the structural integrity of the LN follicle via cytokines such as lymphotoxin β (19). Just as work using the VSV model has demonstrated the importance of the SCS macrophage layer to the B cell response to VSV infection (18, 19), it seems likely that this macrophage population is also responsible for our observation that LN macrophage populations are necessary for the antibody response to rRABV-based vaccines. A mouse model linking diphtheria toxin receptor expression to the CD169 gene, a characteristic surface marker of subcapsular sinus macrophages, leads to targeted ablation of this macrophage subset following administration of diphtheria toxin (28). This spares medullary LN and peripheral tissue macrophage populations and would therefore be an ideal model to test our hypothesis of the importance of subcapsular sinus macrophage-B cell interactions to the development of an antibody response.
The inability of i.d. tissue macrophages at the immunization site to promote any significant early primary antibody response in the absence of LN macrophages was surprising, given the potency of activated, differentiated macrophages as inflammatory pathogen sensors and APCs in draining LNs (12, 14). However, this finding is consistent with the diverse functions of tissue macrophages in tissue repair and regeneration (14, 15), which may be the purpose of the accumulated macrophages our lab detected at sites of intramuscular and intradermal high-dose rRABV-ΔM immunization. Further characterization of these macrophages as tissue-resident or recent inflammatory recruits from the circulation could illuminate their role in RABV immunity (29).
Closer studies of the connective tissue macrophage responses to RABV-based vaccine administration could also enhance our understanding of rabies pathogenesis. Macrophages have been implicated as potential reservoirs of RABV virus due to their susceptibility to RABV infection, their ubiquitous presence in peripheral tissues, and their ability to enter the lymph and blood circulatory systems upon activation (21). This has been a cause for clinical concern in the transplant of solid organs, where organs harvested from a deceased donor who was not known to have rabies encephalitis at the time of harvest have subsequently triggered lethal rabies infections in immunosuppressed recipients (30). Infected macrophages and monocytes are hypothesized to be responsible for the transmission of infection in these cases (30). We observed accumulation of macrophages at the site of rabies immunization, which could be due either to proliferation of resident skin and muscle tissue macrophages or to recruitment of circulating monocytes via CCR2 (chemokine C-C motif receptor 2) and CX3CR1 (fractalkine receptor 1) sensing of chemokines released by infected and damaged tissues (13, 14). Characterization and mapping the fate of these populations could provide insight into the dissemination of RABV via infected macrophages (14). Although these tissue macrophage populations appeared dispensable for an antibody response to rRABV-ΔM immunization, infected macrophages could nevertheless be important for the local immune and repair responses to RABV infection or for the persistence of RABV in peripheral tissues.
Our work suggests that one of the keys to early immunogenicity of live rRABV vaccines is LN macrophages, and targeting these macrophages for activation whether by adjuvants or activating molecules such as ICAM-1 (intercellular adhesion molecule 1) (9) may further enhance the potent immunogenicity we observe with rRABV-ΔM. However, our short-term depletion studies did not examine the role of macrophage populations in mediating the long-term memory and plasma cell response to rRABV-based vaccines. While immunization site macrophages appear unnecessary for the early antibody response, they may yet play roles in limiting the spread of pathogenic rabies or in repairing muscle tissue following the mechanical and inflammatory stress of an injection.
Overall, our work here has demonstrated the contribution of LN macrophages to the antibody response to rRABV-ΔM immunization, opening new avenues of research for the understanding and enhancement of RABV vaccine immunogenicity.
ACKNOWLEDGMENTS
This work was supported by NIH/NIAID grants R01AI079211 and R21AI096907 to J.P.M.
REFERENCES
- 1.World Health Organization. 2013. WHO rabies fact sheet, no. 99. World Health Organization, Geneva, Switzerland. [Google Scholar]
- 2.World Health Organization. 1997. WHO recommendations on rabies post-exposure treatment and the correct technique of intradermal immunization against rabies. World Health Organization, Geneva, Switzerland. [Google Scholar]
- 3.McGettigan JP. 2010. Experimental rabies vaccines for humans. Expert Rev Vaccines 9:1177–1186. doi: 10.1586/erv.10.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Cenna J, Hunter M, Tan GS, Papaneri AB, Ribka EP, Schnell MJ, Marx PA, McGettigan JP. 2009. Replication-deficient rabies virus-based vaccines are safe and immunogenic in mice and nonhuman primates. J Infect Dis 200:1251–1260. doi: 10.1086/605949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.McGettigan JP, David F, Figueiredo MD, Minke J, Mebatsion T, Schnell MJ. 2014. Safety and serological response to a matrix gene-deleted rabies virus-based vaccine vector in dogs. Vaccine 32:1716–1719. doi: 10.1016/j.vaccine.2014.01.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dorfmeier CL, Lytle AG, Dunkel AL, Gatt A, McGettigan JP. 2012. Protective vaccine-induced CD4+ T cell-independent B cell responses against rabies infection. J Virol 86:11533–11540. doi: 10.1128/JVI.00615-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dorfmeier CL, Shen S, Tzvetkov EP, McGettigan JP. 2013. Reinvestigating the role of IgM in rabies virus postexposure vaccination. J Virol 87:9217–9222. doi: 10.1128/JVI.00995-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lytle AG, Norton EJ, Dorfmeier CL, Shen S, McGettigan JP. 2013. B cell infection and activation by rabies virus-based vaccines. J Virol 87:9097–9110. doi: 10.1128/JVI.00800-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Norton JE Jr, Lytle AG, Shen S, Tzvetkov EP, Dorfmeier CL, McGettigan JP. 2014. ICAM-1-based rabies virus vaccine shows increased infection and activation of primary murine B cells in vitro and enhanced antibody titers in-vivo. PLoS One 9:e87098. doi: 10.1371/journal.pone.0087098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Geissmann F, Manz MG, Jung S, Sieweke MH, Merad M, Ley K. 2010. Development of monocytes, macrophages, and dendritic cells. Science 327:656–661. doi: 10.1126/science.1178331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gray D, MacLennan IC, Bazin H, Khan M. 1982. Migrant mu+ delta+ and static mu+ delta- B lymphocyte subsets. Eur J Immunol 12:564–569. doi: 10.1002/eji.1830120707. [DOI] [PubMed] [Google Scholar]
- 12.Martinez-Pomares L, Gordon S. 2007. Antigen presentation the macrophage way. Cell 131:641–643. doi: 10.1016/j.cell.2007.10.046. [DOI] [PubMed] [Google Scholar]
- 13.Yona S, Kim K, Wolf Y, Mildner A, Varol D, Breker M, Strauss-Ayali D, Viukov S, Guilliams M, Misharin A, Hume D, Perlman H, Malissen B, Zelzer E, Jung S. 2013. Fate mapping reveals origins and dynamics of monocytes and tissue macrophages under homeostasis. Immunity 38:79–91. doi: 10.1016/j.immuni.2012.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tidball JG. 2005. Inflammatory processes in muscle injury and repair. Am J Physiol Regul Integr Comp Physiol 288:R345–R353. doi: 10.1152/ajpregu.00454.2004. [DOI] [PubMed] [Google Scholar]
- 15.Arnold L, Henry A, Poron F, Baba-Amer Y, van Rooijen N, Plonquet A, Gherardi RK, Chazaud B. 2007. Inflammatory monocytes recruited after skeletal muscle injury switch into antiinflammatory macrophages to support myogenesis. J Exp Med 204:1057–1069. doi: 10.1084/jem.20070075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Murray PJ, Wynn TA. 2011. Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol 11:723–737. doi: 10.1038/nri3073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Cyster JG. 2010. B cell follicles and antigen encounters of the third kind. Nat Immunol 11:989–996. doi: 10.1038/ni.1946. [DOI] [PubMed] [Google Scholar]
- 18.Junt T, Moseman EA, Iannacone M, Massberg S, Lang PA, Boes M, Fink K, Henrickson SE, Shayakhmetov DM, Di Paolo NC, van Rooijen N, Mempel TR, Whelan SP, von Andrian UH. 2007. Subcapsular sinus macrophages in lymph nodes clear lymph-borne viruses and present them to antiviral B cells. Nature 450:110–114. doi: 10.1038/nature06287. [DOI] [PubMed] [Google Scholar]
- 19.Moseman EA, Iannacone M, Bosurgi L, Tonti E, Chevrier N, Tumanov A, Fu Y, Hacohen N, von Andrian UH. 2012. B cell maintenance of subcapsular sinus macrophages protects against a fatal viral infection independent of adaptive immunity. Immunity 36:415–426. doi: 10.1016/j.immuni.2012.01.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Nakamichi K, Inoue S, Takasaki T, Morimoto K, Kurane I. 2004. Rabies virus stimulates nitric oxide production and CXC chemokine ligand 10 expression in macrophages through activation of extracellular signal-regulated kinases 1 and 2. J Virol 78:9376–9388. doi: 10.1128/JVI.78.17.9376-9388.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ray NB, Ewalt LC, Lodmell DL. 1995. Rabies virus replication in primary murine bone marrow macrophages and in human and murine macrophage-like cell lines: implications for viral persistence. J Virol 69:764–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.van Rooijen N, Hendrikx E. 2010. Liposomes for specific depletion of macrophages from organs and tissues. Methods Mol Biol 605:189–203. doi: 10.1007/978-1-60327-360-2_13. [DOI] [PubMed] [Google Scholar]
- 23.Cenna J, Tan GS, Papaneri AB, Dietzschold B, Schnell MJ, McGettigan JP. 2008. Immune modulating effect by a phosphoprotein-deleted rabies virus vaccine vector expressing two copies of the rabies virus glycoprotein gene. Vaccine 26:6405–6414. doi: 10.1016/j.vaccine.2008.08.069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Dorfmeier CL, Tzvetkov EP, Gatt A, McGettigan JP. 2013. Investigating the role for IL-21 in rabies virus vaccine-induced immunity. PLoS Negl Trop Dis 7:e2129. doi: 10.1371/journal.pntd.0002129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schleicher U, Bogdan C. 2009. Generation, culture and flow-cytometric characterization of primary mouse macrophages. Methods Mol Biol 531:203–224. doi: 10.1007/978-1-59745-396-7_14. [DOI] [PubMed] [Google Scholar]
- 26.Iannacone M, Moseman EA, Tonti E, Bosurgi L, Junt T, Henrickson SE, Whelan SP, Guidotti LG, von Andrian UH. 2010. Subcapsular sinus macrophages prevent CNS invasion on peripheral infection with a neurotropic virus. Nature 465:1079–1083. doi: 10.1038/nature09118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nikbakht N, Shen S, Manser T. 2013. Macrophages are required for localization of antigen-activated B cells to the follicular perimeter and the subsequent germinal center response. J Immunol 190:4923–4927. doi: 10.4049/jimmunol.1300350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gray EE, Friend S, Suzuki K, Phan TG, Cyster JG. 2012. Subcapsular sinus macrophage fragmentation and CD169+ bleb acquisition by closely associated IL-17-committed innate-like lymphocytes. PLoS One 7:e38258. doi: 10.1371/journal.pone.0038258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hashimoto D, Chow A, Noizat C, Teo P, Beasley MB, Leboeuf M, Becker CD, See P, Price J, Lucas D, Greter M, Mortha A, Boyer SW, Forsberg EC, Tanaka M, van Rooijen N, Garcia-Sastre A, Stanley ER, Ginhoux F, Frenette PS, Merad M. 2013. Tissue-resident macrophages self-maintain locally throughout adult life with minimal contribution from circulating monocytes. Immunity 38:792–804. doi: 10.1016/j.immuni.2013.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Srinivasan A, Burton EC, Kuehnert MJ, Rupprecht C, Sutker WL, Ksiazek TG, Paddock CD, Guarner J, Shieh WJ, Goldsmith C, Hanlon CA, Zoretic J, Fischbach B, Niezgoda M, El-Feky WH, Orciari L, Sanchez EQ, Likos A, Klintmalm GB, Cardo D, LeDuc J, Chamberland ME, Jernigan DB, Zaki SR, Rabies in Transplant Recipients Investigation Team . 2005. Transmission of rabies virus from an organ donor to four transplant recipients. N Engl J Med 352:1103–1111. doi: 10.1056/NEJMoa043018. [DOI] [PubMed] [Google Scholar]