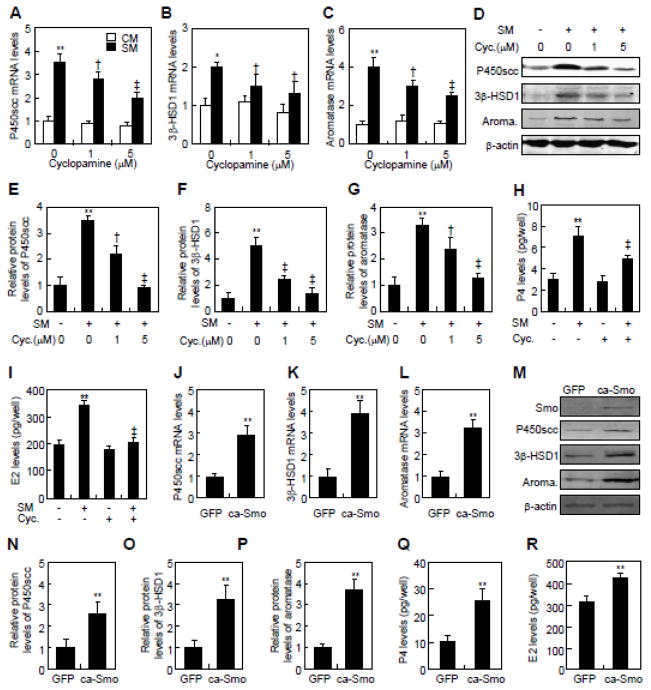
Fig. 2.
Smo is involved in Hh-induced conversion of cholesterol in JEG-3 cells. (A–C) P450scc, 3β-HSD1, and aromatase mRNA levels in JEG-3 cells in response to control medium (CM) and Shh conditional medium (SM) in the presence of indicated concentrations of cyclopamine. (D–F) Quantification via densitometry (n=3) and statistical analysis of bands of G. The data shown represent average fold of each group compared with the control group. (G) P450scc, 3β-HSD1 and aromatase protein levels in JEG-3 cells in response to control medium (−) or Shh conditional medium (SM) in the presence of indicated concentrations of cyclopamine. (H, I) P4 and E2 levels in JEG-3 cells, after 48 hrs of treatments with control medium (−) and Shh conditional medium (S.M., +) in either the presence (+) or the absence (−) of 5 μM cyclopamine (Cyc). (J–L) P450scc, 3β-HSD1 and aromatase mRNA levels in JEG-3 cells, after infection with either control or constitutively active form of Smo (ca-Smo)-expressing lentiviruses for 48 hrs. (M) P450scc, 3β-HSD1 and aromatase protein levels in JEG-3 cells, after infection with either control or ca-Smo-expressing lentiviruses for 48 h. (N–P) Quantification via densitometry (n=3) and statistical analysis of bands of M. The data shown represent average fold of ca-Smo-transfected groups compared with the GFP-transfected groups. (Q, R) P4 and E2 levels in JEG-3 cells, after infection with either control or ca-Smo-expressing lentiviruses for 48 hrs. RNA and protein abundance normalized to β-actin, respectively. **, ‡p<0.01, *, † p<0.05; n=3, error bar, SD.