Abstract
Recent research suggests that the nervous system controls muscles by activating flexible combinations of muscle synergies to produce a wide repertoire of movements. Muscle synergies are like building blocks, defining characteristic patterns of activation across multiple muscles that may be unique to each individual, but perform similar functions. The identification of muscle synergies has strong implications for the organization and structure of the nervous system, providing a mechanism by which task-level motor intentions are translated into detailed, low-level muscle activation patterns. Understanding the complex interplay between neural circuits and biomechanics that give rise to muscle synergies will be critical to advancing our understanding of neural control mechanisms for movement.
Introduction
How do humans and animals successfully interact with the complex and unpredictable dynamics of the natural environment? In motor control, task-level goals such as moving the hand to a target, walking through a door, or orienting the body with respect to gravity must be translated into complex muscle activation patterns that produce the movement. Studies of motor systems ranging from those of invertebrates to those of humans suggest that the nervous system uses flexible combinations of just a few muscle synergies—the elements from which complex muscle activation patterns are constructed—to produce a wide range of motor behaviors [1,2,3•,4•,5•,6]. We define a muscle synergy to be a vector specifying relative levels of muscle activation (cf. [7,8]). The absolute level of activation of each muscle synergy is presumed to be modulated by a single neural command signal. For a given motor task, several muscle synergies are activated in varying combinations to produce the motor behavior [9].
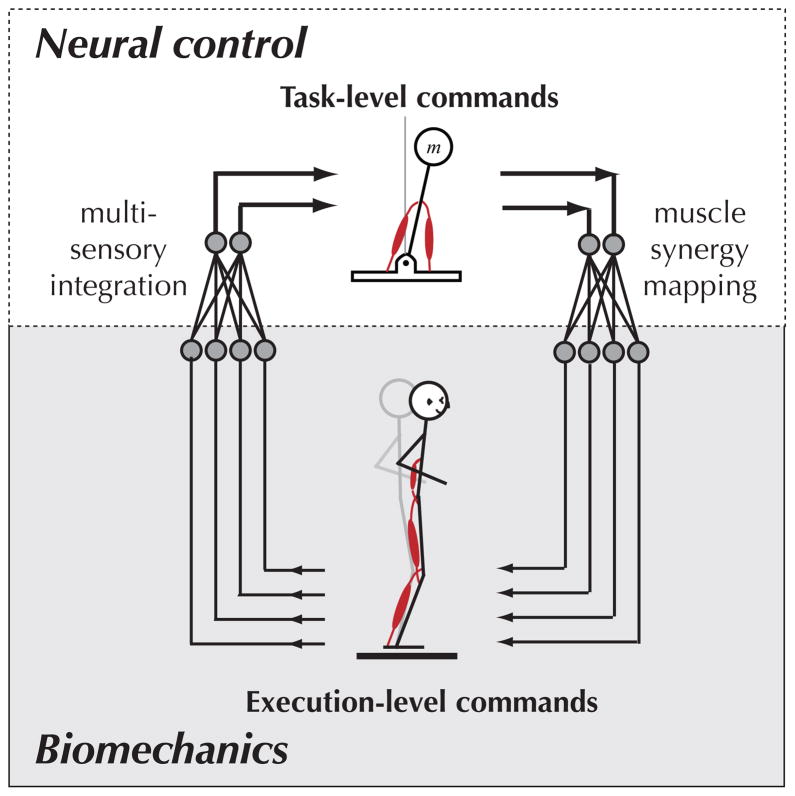
We propose that the nervous system uses muscle synergies as a set of heuristic solutions to transform task-level goals into detailed spatiotemporal patterns of muscle activation (Figure 1). Muscle synergies may therefore represent the bottom of a hierarchal neural control structure in which higher neural centers operate on increasingly conceptual variables related to task-level motor performance [10–14]. This structure mirrors the series of hierarchal transformations that occur in many sensory processing systems [15,16]. The existence of muscle synergies also implies that from among many possible motor solutions, a limited set are chosen by each individual—addressing Bernstein’s degrees-of-freedom problem [17]. Thus, at least on behaviorally short time-scales, motor patterns are constrained by the available library of muscle synergies, influencing motor performance.
Figure 1.
Muscle synergies allow task-level neural commands to be translated into execution-level muscle activation patterns. This hierarchal structure mirrors that of multisensory integration systems.
Here we review recent findings regarding the characteristics and functions of muscle synergies in a variety of motor tasks. We will focus on several open questions in the field: Do muscle synergies produce task-level functions? Are muscle synergies innate or learned? How many muscle synergies are required for task performance? We propose that appropriate neuromechanical models can help to answer these questions.
Muscle synergies
Do muscle synergies produce task-level functions, or are they an artifact of a sophisticated analysis? Recently, results from many areas have demonstrated that the activity of muscle synergies can be correlated to functional outputs related to task performance [1,7,18,19]. During standing balance control, a small set of muscle synergies can be identified that co-activate muscles throughout the limbs and trunk. For any given perturbation, one or more muscle synergies may be activated, so that their combined influences define the resulting muscle activation pattern [9]. The activity of each muscle synergy is directionally tuned, responding to specific directions of center-of-mass (CoM) motion in both voluntary [8] and reactive postural adjustments [3•,4•,20], suggesting an appealing link between muscle synergy activity and higher motor centers (e.g., [21]). In cats, muscle synergy activation has been more specifically correlated with the direction of the force vector produced by the hindlimb for postural stabilization [4•,20]. Further supporting the idea that a few descending signals determine muscle activation patterns, trial-by-trial variations in human postural control can be explained by variations in muscle synergy activation levels [3•]. Because robust muscle synergies must be identified in data sets in which the number of muscles and experimental conditions exceed the number of underlying muscle synergies, they reflect structure in the data rather than structure in the experimental design [22].
Experimental evidence also suggests that – rather than reflecting the state of local sensory or reflex networks during any particular postural task – muscle synergy function is generalized across tasks [4•,20]. When postural configuration is changed, proprioceptive information [23], H-reflex excitability [24], and even intrinsic electrical properties of spinal motoneurons [25] are altered. Despite these alterations, changes in postural responses to perturbations in a range of different postural configurations can be accounted for by modulating the activation levels of a common set of muscle synergies [4•]. Further, when there is explicit sensory loss in the visual, vestibular, or somatosensory systems, the spatial tuning characteristics of individual muscles are retained, suggesting that muscle synergy patterns are unaffected by sensory deficits [26,27]. Similarly, muscle synergies producing locomotor behaviors are largely retained after deafferentation [28]. Although sensory information appears to alter the amplitude and timing of neural commands to muscle synergies, muscle synergy patterns themselves do not appear to be affected.
Are muscle synergies innate or learned? In humans, rudimentary postural responses emerge as early as 4–5 months of age [29], suggesting that the underlying muscle synergies may be innate to some degree. It is possible that muscle synergies for postural control are encoded in cells at the level of the spinal cord [30,31] or brainstem [32•], and are similar to cortico-motoneuronal cells that coordinate hand muscles for grasp [33••,34]. However, inter-subject variations in both muscle synergy patterns and the number of muscle synergies suggest that muscle synergies are shaped by adaptive processes. If this is the case, the morphology and experience of each individual may interact in unexpected ways over time [35], resulting in a unique set of muscle synergy patterns. More subtly, these adaptive processes themselves may vary depending on context [36•,37]. It is therefore reasonable to expect that adaptation may occur differently – and at different rates – for muscle synergy patterns and for descending commands [38•]. Fundamentally, mechanics dictates that responses to postural perturbations across subjects must be similar in terms of kinetic and kinematic variables (cf. [39]). Accordingly, in cats, the directional tuning and force outputs of each muscle synergy are consistent across animals, but the specific muscular patterns of each muscle synergy vary considerably across animals [4•]. Despite these variations, the particular muscle synergy pattern chosen by a subject – whether cat or human – is stable across days and does not appear to be rapidly modified. In contrast, levels of muscle synergy activation, which we presume to reflect descending neural commands, change both across and within experimental conditions [3•,4•].
Finally, how many muscle synergies are required for task performance? Clinically, muscle synergies have been associated with constraints on movement in motor deficit, for example the “pathological synergies” associated with stroke [40,41]. How can we reconcile this conception of muscle synergies with the above studies demonstrating healthy subjects using muscle synergies as a flexible, dextrous strategy? We hypothesize that even in healthy subjects, motor patterns are in fact constrained by the available library of muscle synergies, limiting motor performance to well within the boundaries imposed by musculoskeletal mechanics (cf. [33••,42]). Perhaps then, the difference between some conditions of motor deficit and motor skill is simply a matter of the number of available muscle synergies and the appropriateness of those muscle synergies [43,44].
Neuromechanical modeling
Neuromechanical modeling studies [45] may help resolve these and other pertinent questions regarding muscle synergies. In this section we will outline the advantages of this integrative approach.
Anatomically-detailed biomechanical models are critical for estimating muscle synergy function. Because of interactions between musculoskeletal elements, the function of any single muscle or muscle synergy cannot be examined in isolation. As all muscles accelerate joints they do not cross, proximal and distal muscles must be co-activated to produce stable task function [46–48,49•]. This idea is strikingly apparent when one considers animals without obvious rigid structure (e.g., [50•]).
However, biomechanical models in themselves are insufficient to reveal neural control mechanisms, but rather provide a landscape of possible solutions available to the nervous system. In most natural behaviors, task-level goals can be equivalently achieved with different kinetic or kinematic strategies [51–53], which can themselves be equivalently achieved with different spatial and temporal patterns of muscle activation [54•,55,56]. Therefore, biomechanical models do not uniquely determine muscle activation patterns, nor do they predict muscle synergies, but rather they delineate the large “solution space” afforded by the musculoskeletal system for task performance.
What computations might determine the way the nervous system coordinates muscles? One possibility is that the nervous system explicitly encodes an appropriate transformation function (e.g., an “inverse internal model” [57]), perhaps selected to optimize various performance criteria [56,58•,59,60]. These explanations produce good estimates of experimental measures, generally describing mean neural behaviors without estimating variations from that mean. In general, such models do not directly address how such computations might be inplemented, but instead assume that the nervous system is unconstrained in its plasticity [61].
Neural models that incorporate relevant properties and constraints of neural processing are also necessary to understand how muscle synergies might be encoded in the nervous system. Information representation in the nervous system may be limited by metabolic constraints, making some computational structures more favorable than others [16,62•]. Such “sparse” representations appear to encode explicit features in the environment in an efficient manner [16]. Additionally, the nervous system adapts through statistical learning processes [36•,37,63], so that computational structures may reflect the prior experience of the individual. In turn, conservative mechanisms may limit the context and extent of adaptation [36•,64].
Although the constraints of the nervous system are important, the solution space afforded by the nervous system is still very large. In neural systems, the same network can be modified to produce a variety of outputs [50•,65], which in turn can be equivalently produced by a many different parameter states [66,67]. Therefore, neural models are also insufficient to specify muscle synergies, providing only a landscape of possible motor output patterns.
We propose that muscle synergies emerge from the interacting constraints and features of the nervous and musculoskeletal systems. Our rationale is supported by computational studies of motor cortex topography demonstrating that functionally-organized regions of the cortex may arise from interactions between the biomechanical characteristics of the behavioral repertoire and the biases in the nervous system towards co-localizing neurons that process similar information [68,69•,70]. As an example relevant to muscle synergies, consider the energetic constraints on the musculoskeletal and nervous systems during locomotion. Movement patterns are energetically efficient in a mechanical sense when joint motions are functionally immobilized or linearly correlated (e.g. “inverted pendulum,” or “spring-mass” dynamics in locomotion [71–74]). Simultaneously, energetic efficiency in neural systems — limiting the number of neurons dedicated to encode task performance — may favor piecewise-linear representations of complex elements [16,52]. Thus the combined neural and mechanical energetic pressures may give rise to a motor control strategy of activating linear combinations of muscle synergies that coordinate the musculoskeletal system to act in low-dimensional movement patterns [33••,67].
Muscle synergies may allow higher centers in the nervous system to encode task-level variables, perhaps enabling faster adaptation to environmental demands. In postural control a few variables encoding overall body motion are sufficient to specify muscle synergy activation levels over the time-course of a postural response [54••], eliminating the need to actively control lower-level variables, for example individual joints. This type of dimensional reduction in the neuromechanical system may also explain why simple biomechanical models can predict complex motor behaviors [71–74], as well as compensatory strategies in motor deficit [54••,75•]. But, it is important to note that these low-dimensional and linear behaviors arise from specific relationships between many nonlinear components within the neural and musculoskeletal systems [16,55,67,76••], and do not imply that the systems themselves are linear. Muscle synergies may reflect a sparse code for motor tasks, whereby higher centers can rapidly reconfigure the coordination of task-level commands to muscle synergies, which in turn coordinate specific elements in the periphery that produce functional behaviors [67,77]. Muscle synergies therefore represent a solution to an inverse “binding problem” typical of sensory systems by encoding functional, task-relevant muscle coordination patterns [78]. Thus, muscle synergies may be stable over short-term motor adaptation, but over longer time scales, muscle synergies themselves may also change [38•]. Because of the large solution space of muscle synergies sufficiently near the energetically optimal operating regions defined by simple biomechanical models, a cascade of ancillary factors may influence the specific muscle synergy patterns within each individual [79].
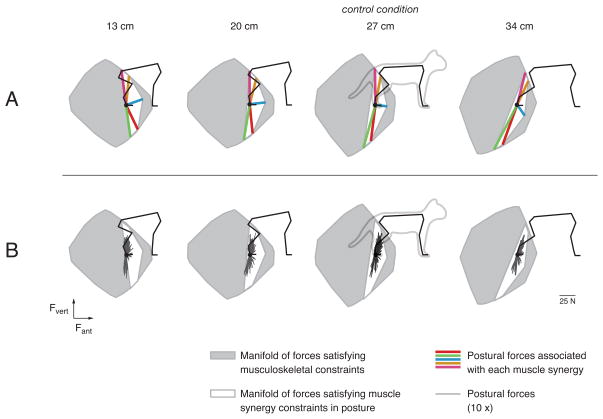
Neuromechanical models can be a practical way to estimate the degree to which motor patterns are constrained by the available library of muscle synergies and whether these constraints influence motor performance. Due to adaptive processes, in statistically-likely tasks motor performance using muscle synergies can approximate that of an optimal controller [10,46,80•]. However, in statistically-unlikely conditions, this approximation may be degraded (cf. [52]). As an example, using a neuromechanical model of the cat hindlimb [81•] we demonstrated that muscle synergies used for postural control in the cat restrict the force-production capability of the limb (Figure 2, [81•]), but may also reduce moments about the CoM when the cat stands in a postural configuration approximating its natural stance. However, when the cat is required to assume postural configurations away from the “preferred” condition, postural forces rotate with the sagittal limb axis, creating large moments about the CoM. This rotation is not imposed by biomechanical limitations, but appears to be a consequence of using identical muscle synergies in disparate postural configurations. Similarly, neuromechanical models of both finger force generation and pedaling in humans demonstrate that muscle activation patterns corresponding to maximal task performance appear to be retained at sub-maximal levels [46,82], possibly in order to achieve a range of related behaviors using the smallest number of muscle synergies. These studies suggest that from the perspective of the nervous system, there may be some “cost” associated with increasing the number of muscle synergies; however, this has yet to be explicitly compared to that of the sub-optimal performance that may arise as a consequence of using fewer muscle synergies.
Figure 2.
The force-production capability of the cat hindlimb is restricted when an identical set of muscle synergies is used for balance control in different postural configurations (adapted from [81•]). A: The gray polygons represent the manifold of possible endpoint forces in a neuromechanical model of the cat hindlimb, given musculoskeletal constraints. From left to right, postural configuration is altered by increasing the “stance distance”, or the anterior-posterior distance between the feet. The most natural, “preferred” postural configuration in the third column is denoted by the cartoon cat. Colored lines denote the force vectors associated with each experimentally-observed muscle synergy. These synergy force vectors rotate with the limb axis as postural configuration changes. The white polygons represent the restricted manifold of possible endpoint forces when the experimentally-identified muscle synergies are used at all postures. B: Manifolds from A are overlaid with recorded postural forces. The “synergy-limited” manifolds predict the systematic rotation of postural forces as stance distance increases.
Finally, neuromechanical models may help explain the redundancy that exists between neural and biomechanical motor control strategies. Biomechanical mechanisms may perform computation typically attributed to active neural control [74,83•]. This idea has been linked to Bernstein’s [17] concept of “preparing the periphery” [50]. In posture, stabilization of the body occurs with equal frequency across individuals through feedforward activation of muscles or through sensory feedback control [84•]. These strategies predict qualitatively different muscle synergy patterns. In the first, a muscle pattern is selected to increase the stiffness of the system, rejecting perturbations using biomechanical properties of the musculature. In the second, a muscle pattern is selected to increase the compliance of the system, to facilitate the effectiveness of the active response (NE Bunderson, TJ Burkholder, LH Ting, American Society of Biomechanics, 2007, also J Biomech in review). The decision to use any particular balance of these two strategies within each individual may be a heuristic process based on experience. Similarly, any particular muscle synergy pattern may represent a unique coordination solution that emerges from complex, multifaceted interactions between the components of the neuromechanical system. Future work investigating the robustness, flexibility, and emergence of muscle synergies depends upon the development of neuromechanical models as well as evaluation techniques to quantify the interactions of components within the models [76••].
Acknowledgments
This work was supported by NIH grants HD-46922 and NS-053822.
Abbreviations
- CoM
center of mass
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
• of special interest
•• of outstanding interest
- 1.Klein Breteler MD, Simura KJ, Flanders M. Timing of muscle activation in a hand movement sequence. Cereb Cortex. 2007;17:803–815. doi: 10.1093/cercor/bhk033. [DOI] [PubMed] [Google Scholar]
- 2.Tresch MC, Saltiel P, Bizzi E. The construction of movement by the spinal cord. Nat Neurosci. 1999;2:162–167. doi: 10.1038/5721. [DOI] [PubMed] [Google Scholar]
- •3.Torres-Oviedo G, Ting LH. Muscle synergies characterizing human postural responses. J Neurophysiol. 2007 doi: 10.1152/jn.01360.2006. In human postural responses, trial-by-trial variations in muscle activation patterns previously associated with adaptation and habituation were accounted for by variations in muscle synergy activation levels across trials. This suggests that descending cognitive and adaptive influences alter postural strategies through the selection and coordination of muscle synergies. [DOI] [PubMed] [Google Scholar]
- •4.Torres-Oviedo G, Macpherson JM, Ting LH. Muscle synergy organization is robust across a variety of postural perturbations. J Neurophysiol. 2006;96:1530–1546. doi: 10.1152/jn.00810.2005. Muscle synergies for postural control were found to produce consistent endpoint force vectors in a limb-referenced coordinate system. These synergies were robustly used in postural configurations that altered the biomechanical contributions of each muscle synergy to postural stabilization, as well as across different perturbations that dramatically altered sensory information eliciting muscle synergy activation. [DOI] [PubMed] [Google Scholar]
- •5.d’Avella A, Portone A, Fernandez L, Lacquaniti F. Control of Fast-Reaching Movements by Muscle Synergy Combinations. J Neurosci. 2006;26:7791–7810. doi: 10.1523/JNEUROSCI.0830-06.2006. The authors identify time-varying muscle synergy patterns during reaching movements in humans. They contrast their time-varying muscle synergy formulation with the “static” formulation used by other researchers [2–4] to demonstrate that their formulation may provide a more compact representation in the nervous system. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Flash T, Hochner B. Motor primitives in vertebrates and invertebrates. Curr Opin Neurobiol. 2005;15:660–666. doi: 10.1016/j.conb.2005.10.011. [DOI] [PubMed] [Google Scholar]
- 7.Krishnamoorthy V, Scholz JP, Latash ML. The use of flexible arm muscle synergies to perform an isometric stabilization task. Clin Neurophysiol. 2007;118:525–537. doi: 10.1016/j.clinph.2006.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Krishnamoorthy V, Goodman S, Zatsiorsky V, Latash ML. Muscle synergies during shifts of the center of pressure by standing persons: identification of muscle modes. Biol Cybern. 2003;89:152–161. doi: 10.1007/s00422-003-0419-5. [DOI] [PubMed] [Google Scholar]
- 9.Ting LH. Dimensional reduction in sensorimotor systems: a framework for understanding muscle coordination of posture. Computational Neuroscience: Theoretical Insights into Brain Function. In: Cisek P, Drew T, Kalaska JF, editors. Prog Brain Res. Vol. 165. Amsterdam: Elsevier; 2007. pp. 301–325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Todorov E, Li W, Pan X. From task parameters to motor synergies: A hierarchical framework for approximately-optimal control of redundant manipulators. J Robot Syst. 2005;22:691–710. doi: 10.1002/rob.20093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Loeb GE, Brown IE, Cheng EJ. A hierarchical foundation for models of sensorimotor control. Exp Brain Res. 1999;126:1–18. doi: 10.1007/s002210050712. [DOI] [PubMed] [Google Scholar]
- 12.Scott SH. Optimal feedback control and the neural basis of volitional motor control. Nat Rev Neuro. 2004;5:534–546. doi: 10.1038/nrn1427. [DOI] [PubMed] [Google Scholar]
- 13.Deliagina TG, Orlovsky GN, Zelenin PV, Beloozerova IN. Neural Bases of Postural Control. Physiology. 2006;21:216–225. doi: 10.1152/physiol.00001.2006. [DOI] [PubMed] [Google Scholar]
- 14.Gurfinkel VS, Ivanenko Yu P, Levik Yu S, Babakova IA. Kinesthetic reference for human orthograde posture. Neuroscience. 1995;68:229–243. doi: 10.1016/0306-4522(95)00136-7. [DOI] [PubMed] [Google Scholar]
- 15.Poggio T, Bizzi E. Generalization in vision and motor control. Nature. 2004;431:768–774. doi: 10.1038/nature03014. [DOI] [PubMed] [Google Scholar]
- 16.Olshausen BA, Field DJ. Sparse coding of sensory inputs. Curr Opin Neurobiol. 2004;14:481–487. doi: 10.1016/j.conb.2004.07.007. [DOI] [PubMed] [Google Scholar]
- 17.Bernstein N. The Coordination and Regulation of Movements. New York: Pergamon Press; 1967. [Google Scholar]
- 18.Ivanenko YP, Cappellini G, Dominici N, Poppele RE, Lacquaniti F. Coordination of locomotion with voluntary movements in humans. J Neurosci. 2005;25:7238–7253. doi: 10.1523/JNEUROSCI.1327-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.d’Avella A, Bizzi E. Shared and specific muscle synergies in natural motor behaviors. Proc Natl Acad Sci USA. 2005;102:3076–3081. doi: 10.1073/pnas.0500199102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ting LH, Macpherson JM. A limited set of muscle synergies for force control during a postural task. J Neurophysiol. 2005;93:609–613. doi: 10.1152/jn.00681.2004. [DOI] [PubMed] [Google Scholar]
- 21.Georgopoulos AP, Kalaska JF, Caminiti R, Massey JT. On the Relations between the Direction of Two-Dimensional Arm Movements and Cell Discharge in Primate Motor Cortex. J Neurosci. 1982;2:1527–1537. doi: 10.1523/JNEUROSCI.02-11-01527.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Macpherson JM. How flexible are muscle synergies? In: Humphrey DR, Freund H-J, editors. Motor Control: Concepts and Issues. Wiley Press; 1991. pp. 33–47. [Google Scholar]
- 23.Bosco G, Eian J, Poppele RE. Phase-specific sensory representations in spinocerebellar activity during stepping: evidence for a hybrid kinematic/kinetic framework. Exp Brain Res. 2006;175:83–96. doi: 10.1007/s00221-006-0530-7. [DOI] [PubMed] [Google Scholar]
- 24.Knikou M, Rymer Z. Effects of changes in hip joint angle on H-reflex excitability in humans. Exp Brain Res. 2002;143:149–159. doi: 10.1007/s00221-001-0978-4. [DOI] [PubMed] [Google Scholar]
- 25.Hyngstrom AS, Johnson MD, Miller JF, Heckman CJ. Intrinsic electrical properties of spinal motoneurons vary with joint angle. Nat Neurosci. 2007;10:363–369. doi: 10.1038/nn1852. [DOI] [PubMed] [Google Scholar]
- 26.Stapley PJ, Ting LH, Hulliger M, Macpherson JM. Automatic postural responses are delayed by pyridoxine-induced somatosensory loss. J Neurosci. 2002;22:5803–5807. doi: 10.1523/JNEUROSCI.22-14-05803.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Inglis JT, Horak FB, Shupert CL, Jones-Rycewicz C. The importance of somatosensory information in triggering and scaling automatic postural responses in humans. Exp Brain Res. 1994;101:159–164. doi: 10.1007/BF00243226. [DOI] [PubMed] [Google Scholar]
- 28.Cheung VCK, d’Avella A, Tresch MC, Bizzi E. Central and sensory contributions to the activation and organization of muscle synergies during natural motor behaviors. J Neurosci. 2005;25:6419–6434. doi: 10.1523/JNEUROSCI.4904-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Massion J. Postural Control Systems in Developmental Perspective. Neurosci Biobehav Rev. 1998;22:465–472. doi: 10.1016/s0149-7634(97)00031-6. [DOI] [PubMed] [Google Scholar]
- 30.Giszter SF, Mussa-Ivaldi FA, Bizzi E. Convergent force fields organized in the frog’s spinal cord. J Neurosci. 1993;13:467–491. doi: 10.1523/JNEUROSCI.13-02-00467.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ivanenko YP, Poppele RE, Lacquaniti F. Spinal cord maps of spatiotemporal alpha-motoneuron activation in humans walking at different speeds. J Neurophysiol. 2005 doi: 10.1152/jn.00767.2005. [DOI] [PubMed] [Google Scholar]
- •32.Schepens B, Drew T. Independent and Convergent Signals From the Pontomedullary Reticular Formation Contribute to the Control of Posture and Movement During Reaching in the Cat. J Neurophysiol. 2004;92:2217–2238. doi: 10.1152/jn.01189.2003. The authors demonstrate the role of reticulospinal neurons, which have divergent distal projection patterns, in postural adjustments during a voluntary task. These brainstem neurons are a possible neuroanatomical substrate for encoding muscle synergies for postural control. [DOI] [PubMed] [Google Scholar]
- ••33.Gentner R, Classen J. Modular organization of finger movements by the human central nervous system. Neuron. 2006;52:731–742. doi: 10.1016/j.neuron.2006.09.038. The authors compare kinematic synergies in hand movements elicited by transcranial magnetic stimulation (TMS) to kinematic synergies in hand movements elicited through passive joint manipulation. They note that while eight synergies are necessary to approximate passive motions, indicative of the high-dimensional biomechanical apparatus of the hand, only four are required to represent TMS-elicited movements. This result suggests that the dimensional collapse arises from neural control mechanisms, and is not inherent in the musculoskeletal biomechanics. [DOI] [PubMed] [Google Scholar]
- 34.Lemon RN. The G. L. Brown Prize Lecture. Cortical control of the primate hand. Exp Physiol. 1993;78:263–301. doi: 10.1113/expphysiol.1993.sp003686. [DOI] [PubMed] [Google Scholar]
- 35.Santos VJ, Valero-Cuevas FJ. Reported anatomical variability naturally leads to multimodal distributions of Denavit-Hartenberg parameters for the human thumb. IEEE Trans Biomed Eng. 2006;53:155–163. doi: 10.1109/TBME.2005.862537. [DOI] [PubMed] [Google Scholar]
- •36.Thoroughman KA, Fine MS, Taylor JA. Trial-by-trial motor adaptation: a window into elemental neural computation. Computational Neuroscience: Theoretical Insights into Brain Function. In: Cisek P, Drew T, Kalaska JF, editors. Prog Brain Res. Vol. 165. Amsterdam: Elsevier; 2007. pp. 373–382. The authors argue that the way we learn is a function of experience. They demonstrate that learning rules are context-dependent, such that the same motor errors may result in the qualitatively different “proportional” versus “categorical” motor adaptation strategies. [DOI] [PubMed] [Google Scholar]
- 37.Krakauer JW, Mazzoni P, Ghazizadeh A, Ravindran R, Shadmehr R. Generalization of Motor Learning Depends on the History of Prior Action. PLoS Biol. 2006;4:e316. doi: 10.1371/journal.pbio.0040316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- •38.Smith MA, Ghazizadeh A, Shadmehr R. Interacting Adaptive Processes with Different Timescales Underlie Short-Term Motor Learning. PLoS Biol. 2006;4:e179. doi: 10.1371/journal.pbio.0040179. The authors use a force-field adaptation paradigm in humans to identify two interacting mechanisms – acting at different timescales – underlying short-term motor adaptation in reaching. They suggest that at least the faster of these two mechanisms is dependent on cerebellar function. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Alexandrov AV, Frolov AA, Horak FB, Carlson-Kuhta P, Park S. Feedback equilibrium control during human standing. Biol Cybern. 2005:1–14. doi: 10.1007/s00422-005-0004-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Cirstea MC, Levin MF. Compensatory strategies for reaching in stroke. Brain. 2000;123:940–953. doi: 10.1093/brain/123.5.940. [DOI] [PubMed] [Google Scholar]
- 41.Gowland C, deBruin H, Basmajian JV, Plews N, Burcea I. Agonist and antagonist activity during voluntary upper-limb movement in patients with stroke. Phys Ther. 1992;72:624–633. doi: 10.1093/ptj/72.9.624. [DOI] [PubMed] [Google Scholar]
- 42.Valero-Cuevas FJ. A mathematical approach to the mechanical capabilities of limbs and fingers. Progress in Motor Control V; August 17–20, 2005; State College, Pennsylvania. 2006. [Google Scholar]
- 43.Turton A, Fraser C, Flament D, Werner W, Bennett KMB, Lemon RN. Organization of cortico-motoneuronal projections from the primary motor cortex: evidence for task-related function in monkey and man. In: Thilmann A, Burke DJ, Rymer WZ, editors. Spacticity - Mechanisms and Management. Berlin & Heidelberg: Springer-Verlag; 1993. pp. 8–24. [Google Scholar]
- 44.Turton A, Wroe S, Trepte N, Fraser C, Lemon RN. Contralateral and ipsilateral EMG responses to transcranial magnetic stimulation during recovery of arm and hand function after stroke. Electroencephalography and clinical neurophysiology. 1996;101:316–328. doi: 10.1016/0924-980x(96)95560-5. [DOI] [PubMed] [Google Scholar]
- 45.Nishikawa K, Biewener AA, Aerts P, Ahn AN, Chiel HJ, Daley MA, Daniel TL, Full RJ, Hale ME, Hedrick TL, et al. Neuromechanics: an integrative approach for understanding motor control. Integr Comp Biol. 2007;47:16–54. doi: 10.1093/icb/icm024. [DOI] [PubMed] [Google Scholar]
- 46.Raasch CC, Zajac FE. Locomotor strategy for pedaling: Muscle groups and biomechanical functions. J Neurophysiol. 1999;82:515–525. doi: 10.1152/jn.1999.82.2.515. [DOI] [PubMed] [Google Scholar]
- 47.Nozaki D, Nakazawa K, Akai M. Uncertainty of knee joint muscle activity during knee joint torque exertion: the significance of controlling adjacent joint torque. J Appl Physiol. 2005;99:1093–1103. doi: 10.1152/japplphysiol.00365.2005. [DOI] [PubMed] [Google Scholar]
- 48.Zajac FE. Muscle coordination of movement: A perspective. J Biomech. 1993;26:109–124. doi: 10.1016/0021-9290(93)90083-q. [DOI] [PubMed] [Google Scholar]
- •49.van Antwerp KW, Burkholder TJ, Ting LH. Inter-joint coupling effects on muscle contributions to endpoint force and acceleration in a musculoskeletal model of the cat hindlimb. J Biomech. 2007 doi: 10.1016/j.jbiomech.2007.06.001. The authors use a detailed neuromechanical model to demonstrate that the inter-joint coupling between multiple muscles must be considered when evaluating functional motor outputs. Even when identical muscles are activated, the specific ratio of activation may change the endpoint force direction by up to 180°. This demonstrates one of the mechanical principles that may drive muscle synergy organization. [DOI] [PMC free article] [PubMed] [Google Scholar]
- •50.Ye H, Morton DW, Chiel HJ. Neuromechanics of coordination during swallowing in Aplysia californica. J Neurosci. 2006;26:1470–1485. doi: 10.1523/JNEUROSCI.3691-05.2006. The authors examine the neuromechanical coordination of swallowing in Aplysia. They demonstrate that interactions among the feeding musculature can dynamically reconfigure the biomechanical function of identified motor neurons. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Yang JF, Scholz JP, Latash ML. The role of kinematic redundancy in adaptation of reaching. Exp Brain Res. 2007;176:54–69. doi: 10.1007/s00221-006-0602-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Todorov E. Optimality principles in sensorimotor control. Nat Neurosci. 2004;7:907–915. doi: 10.1038/nn1309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Kuo AD. An optimal state estimation model of sensory integration in human postural balance. J Neural Eng. 2005;2:S235–249. doi: 10.1088/1741-2560/2/3/S07. [DOI] [PubMed] [Google Scholar]
- ••54.Lockhart DB, Ting LH. Optimal sensorimotor transformations for balance. Nat Neurosci. 2007;10:1329–1336. doi: 10.1038/nn1986. The authors demonstrate that commands from a hierarchal, task-level feedback controller are directly used to modulate the temporal activity of many muscles during a postural task in cats. Their results suggest that center of mass kinematics are used to specify time-dependent activation of muscle synergies, which in turn coordinate execution-level elements (muscles) in the periphery. Moreover, the optimal combination of task-level information used to define temporal muscle activation patterns is altered after sensory loss. [DOI] [PubMed] [Google Scholar]
- 55.Gottlieb GL. Muscle activation patterns during two types of voluntary single-joint movement. J Neurophysiol. 1998;80:1860–1867. doi: 10.1152/jn.1998.80.4.1860. [DOI] [PubMed] [Google Scholar]
- 56.van Bolhuis BM, Gielen CC. A comparison of models explaining muscle activation patterns for isometric contractions. Biol Cybern. 1999;81:249–261. doi: 10.1007/s004220050560. [DOI] [PubMed] [Google Scholar]
- 57.Kawato M. Internal models for motor control and trajectory planning. Curr Opin Neurobiol. 1999;9:718–727. doi: 10.1016/s0959-4388(99)00028-8. [DOI] [PubMed] [Google Scholar]
- •58.Kurtzer I, Pruszynski JA, Herter TM, Scott SH. Primate Upper Limb Muscles Exhibit Activity Patterns That Differ From Their Anatomical Action During a Postural Task. J Neurophysiol. 2006;95:493–504. doi: 10.1152/jn.00706.2005. The authors note that two muscle synergies are sufficient to explain the preferred directions of muscles in the primate forelimb during a planar postural task. However, they demonstrate that minimizing total muscle activity using different criteria can lead to an identical pattern. They argue that patterned covariation does not require muscle synergies. [DOI] [PubMed] [Google Scholar]
- 59.Valero-Cuevas FJ, Zajac FE, Burgar CG. Large index-fingertip forces are produced by subject-independent patterns of muscle excitation. J Biomech. 1998;31:693–703. doi: 10.1016/s0021-9290(98)00082-7. [DOI] [PubMed] [Google Scholar]
- 60.Fagg AH, Shah A, Barto AG. A Computational Model of Muscle Recruitment for Wrist Movements. J Neurophysiol. 2002;88:3348–3358. doi: 10.1152/jn.00621.2002. [DOI] [PubMed] [Google Scholar]
- 61.Harris CM, Wolpert DM. Signal-dependent noise determines motor planning. Nature. 1998;394:780–784. doi: 10.1038/29528. [DOI] [PubMed] [Google Scholar]
- •62.Denève S, Duhamel J-R, Pouget A. Optimal Sensorimotor Integration in Recurrent Cortical Networks: A Neural Implementation of Kalman Filters. J Neurosci. 2007;27:5744–5756. doi: 10.1523/JNEUROSCI.3985-06.2007. The authors demonstrate an elegant neural-network implementation of a Kalman filter for optimal state estimation. They discuss the implications of modularity in neural networks in terms of a trade-off between avoiding combinatorial expansion in number of neurons, and sacrificing accuracy. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Körding KP, Wolpert DM. Bayesian integration in sensorimotor learning. Nature. 2004;427:244–247. doi: 10.1038/nature02169. [DOI] [PubMed] [Google Scholar]
- 64.Rich MM, Wenner P. Sensing and expressing homeostatic synaptic plasticity. Trends Neurosci. 2007;30:119–125. doi: 10.1016/j.tins.2007.01.004. [DOI] [PubMed] [Google Scholar]
- 65.Weimann JM, Marder E. Switching neurons are integral members of multiple oscillatory networks. Curr Biol. 1994;4:896–902. doi: 10.1016/s0960-9822(00)00199-8. [DOI] [PubMed] [Google Scholar]
- 66.Prinz AA, Bucher D, Marder E. Similar network activity from disparate circuit parameters. Nat Neurosci. 2004;7:1345–1352. doi: 10.1038/nn1352. [DOI] [PubMed] [Google Scholar]
- 67.Leonardo A. Degenerate coding in neural systems. J Comp Physiol, A. 2005;191:995–1010. doi: 10.1007/s00359-005-0026-0. [DOI] [PubMed] [Google Scholar]
- 68.Aflalo TN, Graziano MS. Possible origins of the complex topographic organization of motor cortex: reduction of a multidimensional space onto a two-dimensional array. J Neurosci. 2006;26:6288–6297. doi: 10.1523/JNEUROSCI.0768-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ••69.Graziano MS, Aflalo TN. Rethinking cortical organization: moving away from discrete areas arranged in hierarchies. Neuroscientist. 2007;13:138–147. doi: 10.1177/1073858406295918. The authors argue that the historically daunting organization of the monkey motor cortex results from interactions between simple rules governing the activity of the biomechanical and neural systems. They demonstrate a model that reproduces several features of the motor cortex organization by projecting the statistics of the biomechanical repertoire onto a 2-D cortical sheet governed by simple rules such as “like attracts like.”. [DOI] [PubMed] [Google Scholar]
- 70.Graziano M. The organization of behavioral repertoire in motor cortex. Annu Rev Neurosci. 2006;29:105–134. doi: 10.1146/annurev.neuro.29.051605.112924. [DOI] [PubMed] [Google Scholar]
- 71.Srinivasan M, Ruina A. Computer optimization of a minimal biped model discovers walking and running. Nature. 2006;439:72–75. doi: 10.1038/nature04113. [DOI] [PubMed] [Google Scholar]
- 72.Holmes P, Full R, Koditschek DE, Guckenheimer J. The dynamics of legged locomotion: models, analyses, and challenges. SIAM Review. 2006;48:207–304. [Google Scholar]
- 73.Kuo AD. The six determinants of gait and the inverted pendulum analogy: A dynamic walking perspective. Hum Mov Sci. 2007;26:617–656. doi: 10.1016/j.humov.2007.04.003. [DOI] [PubMed] [Google Scholar]
- 74.Blickhan R, Seyfarth A, Geyer H, Grimmer S, Wagner H, Gunther M. Intelligence by mechanics. Phil Trans R Soc A. 2007;365:199–220. doi: 10.1098/rsta.2006.1911. [DOI] [PubMed] [Google Scholar]
- •75.Fonseca ST, Holt KG, Fetters L, Saltzman E. Dynamic resources used in ambulation by children with spastic hemiplegic cerebral palsy: relationship to kinematics, energetics, and asymmetries. Phys Ther. 2004;84:344–354. discussion 355–348. The authors demonstrate that hemiplegic gait in cerebral palsy can be characterized well with two simple biomechanical models – and “inverted-pendulum” model in the unaffected limb, and a “spring-mass” model in the affected limb. [PubMed] [Google Scholar]
- ••76.Mitchell CS, Lee RH. Output-based comparison of alternative kinetic schemes for the NMDA receptor within a glutamate spillover model. J Neural Eng. 2007;4:380–389. doi: 10.1088/1741-2560/4/4/004. The authors develop an output-based multi-variate analysis technique called model-relational analysis to examine the correlated outputs within a model of N-methyl-D-aspartate receptor kinetics. They use model relational analysis to analyze and discern the contributions of complex interactions amongst model components that are hidden in overall model behavior. This technique may be applicable to the analysis of experimental data and model outputs in a wide range of systems. [DOI] [PubMed] [Google Scholar]
- 77.Fiete IR, Hahnloser RHR, Fee MS, Seung HS. Temporal Sparseness of the Premotor Drive Is Important for Rapid Learning in a Neural Network Model of Birdsong. J Neurophysiol. 2004;92:2274–2282. doi: 10.1152/jn.01133.2003. [DOI] [PubMed] [Google Scholar]
- 78.Jackson A, Gee VJ, Baker SN, Lemon RN. Synchrony between Neurons with Similar Muscle Fields in Monkey Motor Cortex. Neuron. 2003;38:115–125. doi: 10.1016/s0896-6273(03)00162-4. [DOI] [PubMed] [Google Scholar]
- 79.Sentis L, Khatib O. Synthesis of whole-body behaviors through hierarchical control of behavioral primitives. Int J Hum Rob. 2005;2:505–518. [Google Scholar]
- •80.Chhabra M, Jacobs RA. Properties of synergies arising from a theory of optimal motor behavior. Neural Comput. 2006;18:2320–2342. doi: 10.1162/neco.2006.18.10.2320. The authors demonstrate that performance of a simulated reaching task is equivalent using muscle activation patterns predicted by an optimal control formulation or using muscle synergy approximations of those patterns. [DOI] [PubMed] [Google Scholar]
- •81.McKay JL, Ting LH. Functional muscle synergies constrain force production during postural tasks. J Biomech. 2007 doi: 10.1016/j.jbiomech.2007.09.012. The authors use a neuromechanical model to validate the robustness of muscle synergy force outputs revealed in [4] and to demonstrate the constraints on functional capabilities that result from using generalized muscle synergies across multiple tasks. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Valero-Cuevas FJ. Predictive modulation of muscle coordination pattern magnitude scales fingertip force magnitude over the voluntary range. J Neurophysiol. 2000;83:1469–1479. doi: 10.1152/jn.2000.83.3.1469. [DOI] [PubMed] [Google Scholar]
- •83.Valero-Cuevas FJ, Yi JW, Brown D, McNamara RV, 3rd, Paul C, Lipson H. The tendon network of the fingers performs anatomical computation at a macroscopic scale. IEEE Trans Biomed Eng. 2007;54:1161–1166. doi: 10.1109/TBME.2006.889200. The authors quantify the nonlinear tension-routing capability of cadaveric and simulated fingers. They demonstrate that muscle activation patterns producing the same net tendon forces proximally can generate different torques at distal joints. The authors suggest that for any neuromechanical system “part of the controller [may be] embedded in the anatomy” through this type of biomechanical computation. These results suggest that muscle synergy structures could reflect biomechanical computations performed by the tendon structure during manipulation. [DOI] [PubMed] [Google Scholar]
- •84.Gurfinkel V, Cacciatore TW, Cordo P, Horak F, Nutt J, Skoss R. Postural Muscle Tone in the Body Axis of Healthy Humans. J Neurophysiol. 2006;96:2678–2687. doi: 10.1152/jn.00406.2006. The authors demonstrate that axial postural muscle tone can be tonically or dynamically modulated in response to slow trunk rotations. About half of the individuals tested maintained constant muscle activity during trunk rotation, corresponding to higher trunk stiffness. The other half dynamically modulated muscle activity during trunk rotation, increasing the compliance of the trunk. They suggest that dynamic regulation of postural tone is related to the excitability of lower level neural structures, which may be modified to regulate the degree of mobility or stability of the system. [DOI] [PubMed] [Google Scholar]