Abstract
Objectives
Concentrations of endothelin I (ET1) are elevated in CHF patients, and, like other biomarkers that reflect hemodynamic status and cardiac pathophysiology, are prognostic. The Singulex assay (Sgx-ET1) measures the active form of ET1, with a short in-vivo half-life and C-terminal endothelin-1 (CT-ET1) is measured by the Brahms assay and is a modified (degraded) product with longer half-life. We aimed to determine the prognostic importance of active and modified forms of endothelin 1 (Singulex and Brahms assays) in comparison with other commonly measured biomarkers of inflammation, hemodynamic status and cardiac physiology in CHF.
Design & Methods
Plasma biomarkers (Sgx-ET1, CT-ET1, NTproBNP, IL-6, TNFα, cTnI, VEGF, hs-CRP, Galectin-3, ST2) were measured in 134 NYHA class II and III CHF patients with systolic dysfunction. Prognostic importance of biomarkers for hospitalization or death were calculated by both logistic regression and Kaplan-Meier survival analyses.
Results
CT-ET1 (OR 5.2, 95% CI 1.7–15.7) and Sgx-ET1 (OR 2.9, CI 1.1–7.7) were independent predictors of hospitalization and death and additively predicted events after adjusting for age, sex and other significant biomarkers. Other biomarkers did not improve the model. Similarly, in Cox regression analysis, only CT-ET1 (HR 3.4, 95% CI 1.4–8.4), VEGF (2.7, 95% CI 1.3–5.4) and Sgx-ET1 (HR 2.6, 95% CI 1.2–5.6) were independently prognostic.
Conclusions
Elevated concentrations of endothelin 1 predict mortality and hospitalizations in HF patients. Endothelin 1 was more prognostic than commonly obtained hemodynamic, inflammatory and fibrotic biomarkers. Two different assays of endothelin 1 independently and synergistically were prognostic, suggesting either complementary information or extreme prognostic importance.
Keywords: Endothelin, Heart failure, congestive, prognosis
1.1 Introduction
Concentrations of endothelin 1 (ET1), a potent vasoconstrictor produced by endothelial cells, are elevated in heart failure (HF) patients, and, like other biomarkers that reflect hemodynamic status and cardiac pathophysiology, have been shown to be prognostic in HF patients.1,2 However, there are different forms of the peptide that are measured by two available ET1 assays. The Singulex assay (Sgx-ET1) measures the active form of ET1, with a short in vivo half-life. On the other hand, C-terminal endothelin 1 (CT-ET1) is measured by the Brahms assay and is a modified (degraded) product with a longer half-life. The prognostic importance of these forms of ET1 is unknown.
Furthermore, few studies have compared the prognostic importance of ET1 to the multiple biomarkers which are prognostic in HF patients. Natriuretic peptides, markers of inflammation, and indicators of fibrosis all can predict mortality and morbidity. Whether ET1 adds information to that provided by these biomarkers has not been fully characterized.
Therefore, we sought to determine the relative prognostic importance of the active and modified forms of ET1 in comparison with other commonly measured biomarkers of inflammation, hemodynamic status and cardiac physiology.
2.1 Material and methods
2.1.1
Plasma biomarkers were measured in 134 subjects with ischemic and non-ischemic HF that were recruited as part of the BETRHeart study evaluating the interaction of psychologic and physiologic characteristics of HF patients and their interactions with outcomes. Entry criteria included New York Heart Association (NYHA) classification II or III HF for at least 3 months and left ventricular ejection fraction ≤ 40%. This study was approved by the Human Volunteers Research committees at the University of Maryland Baltimore and the Uniformed Services University of the Health Sciences. All patients signed an informed consent.
2.1.2 Assays
Serum and plasma samples were assayed for a panel of biomarkers. Blood was drawn in K2 EDTA, heparinized, and plain tubes and immediately spun for 15 minutes in a refrigerated centrifuge. Serum and plasma were separated and stored at −20°C until assayed. Research-Use-Only (RUO) assays ET1 (Sgx-ET1), IL-6, TNFα, cardiac specific troponin I (cTnI), and Vascular endothelial growth factor (VEGF) were all measured using high sensitivity single-molecule counting technology (RUO, Erenna® Immunoassay System, Singulex, Inc.) at no charge. This system utilizes paramagnetic microparticles as the solid phase format in combination with single-molecule counting. In the Erenna system, the immunoassay complex formed on the MP surface results in the release of fluorescently labeled detection antibody. The resulting solution is sipped into a 100 μm flow capillary and photons are counted, via confocal microscopy, as they pass through a 2 μm interrogation space. CT-ET1 was measured on the Brahms Kryptor System (Thermo Fischer); this assay quantifies an inactive c-terminus degradation product of ET1, which has a longer half-life than the parent molecule. NT-proBNP and hs-CRP were both determined using the ElecSys 2010 analyzer (Roche Diagnostics, Indianapolis, IN).
Galectin 3 (BG Medicine, Boston, MA) and ST2 (Critical Diagnostics, San Diego, CA) were measured by ELISA in 115 patients. Because of the smaller number of measurements, these biomarkers were not included in the primary analyses. However, they were included in secondary analyses.
The ET-1, cTnI, IL-6, TNF-a, and VEGF assays were performed using single molecule counting technology. All of the assays, except VEGF, have been validated as laboratory developed tests in the Singulex CLIA licensed, CAP Accredited laboratory. The VEGF assay was at a validated development stage. The analytical characteristics are depicted in table 1.
Table 1.
Analytical characteristics of biomarkers
| Biomarker | Limit of Detection | Low Limit of Quantitation | Upper reporting limit | Upper Reference Range (99thpercentile for cTnI) | Precision Profile (CV) and Comments |
|---|---|---|---|---|---|
| IL-6 | 0.01 pg/mL | 0.38 pg/mL | 100 pg/mL | 7.2 pg/mL | 12% at 1.3 pg/mL 12% at 12 pg/mL |
| TNFα | 0.01 pg/mL | 0.11 pg/mL | 50 pg/mL | 4.3 pg/mL | 11% at 1.2 pg/mL 7% at 17.8 pg/mL |
| cTnI | 0.09 pg/mL | 0.40 pg/mL | 250 pg/mL | 7.1 pg/mL | 11% at 3.0 pg/mL 15% at 60.9 pg/mL |
| VEGF | 1.0 pg/mL | 6.0 pg/mL | 400 pg/mL | 65.8 pg/mL | 12% at 22.7 pg/mL 10% at 39.3 pg/mL |
| Sgx-ET1 | 0.07 pg/mL | 0.20 pg/mL | 250 pg/mL | 3.7 pg/mL | 14% at 1.8 pg/mL 7% at 7.8 pg/mL |
| CT-ET | 0.4 pmol/L | 3 pmol/L | 500 pmol/L | 73.9 pmol/L | <10% at 40–80 pmol/L <6% at >80 pmol/L: |
| NTproBNP | 5 pg/mL | 50 pg/mL | 35,000 pg/mL | < 75 years old < 125 pg/mL >75 years old <450 pg/mL |
ICON cutoffs for HF diagnosis: < 50 years old: >450 pg/mL 50–75 years old: >900 pg/mL >75 years old: >1800 pg/mL exclusion of acute heart failure (any adult age): <300 pg/mL |
| ST2 | 1.31 ng/mL | 2.35 ng/mL | 200 ng/mL | 35 ng/mL* | Storage Condition Stability Room temperature (22–28°C) 48 hours; Refrigerated (2–8°C) 7 days, Frozen (−20°C or −80°C) at least 18 months. |
| Galectin-3 | 1.13 ng/mL | 1.32 ng/mL | 94.8 ng/mL | 17.8 ng/mL** | 2.0% at 6.1 ng/mL 5.1% at 17.6 ng/mL 7.7% at 20.7 ng/mL 4.2% at 26.3 ng/mL 4.4% at 46.2 ng/mL 8.0% at 72.2 ng/mL |
Higher risk of adverse outcomes when ST2 levels are above this cutoff value.
Higher risk of adverse outcomes when Galectin-3 levels are above 17.8 ng/mL.
Since ET-1 is a biologically active small peptide derived from a larger precursor molecule and has a demonstrated short biological half-life in circulation (is rapidly cleared), it was important to understand the physical stability of the molecule in plasma. Sgx-ET1 was measured in 20 freshly drawn (day 0) EDTA plasma samples which were then stored at 2–8°C, for 1, 4, 8, and 10 days, and tested for ET-1 on the respective day. Sgx-ET1 did not show decay from day 0 values up to day 4. Day 8 & 10 samples showed diminished concentrations of SgX ET-1. Additionally 10 plasma samples were tested after being frozen at −80°C and thawed 0, 1, and 2 times. ET-1 concentrations were stable for up to 2 freeze-thaw cycles. Taken together, these findings demonstrate that ET-1 is reasonably stable in plasma for analytical testing.
2.1.3 Statistical analysis
The prognostic importance of the biomarkers for hospitalization or death were calculated by logistic regression, Cox’s regression, and Kaplan-Meier survival analyses.
Odd ratios (OR) were determined by dichotomizing biomarkers using the empirical distribution function. The cutpoints used are listed in table 2. Outcome was compared using both unadjusted and adjusted (for age, sex, and other biomarkers) logistic regression analysis. CRP was not included in adjusted analyses because it was statistically non-significant independently.
Table 2.
Biomarkers Dichotomized using the Empirical Distribution Function
| Biomarker | Cutpoint | Total CV at Cutpoint |
|---|---|---|
| IL-6 | 15 pg/mL | 13% |
| TNFα | 5.3 pg/mL | 9% |
| VEGF | 36.7 pg/mL | 10% |
| CT-ET1 | 80 pmol/L | 6% |
| Sgx-ET1 | 4.4 pg/mL | 11% |
| cTnI | 9.1 pg/mL | 8% |
| NTproBNP | 1000 pg/mL | 2% |
| CRP | 4.2mg/L | 3% |
Hazard ratios were used for Kaplan-Meier survival analyses to predict time to first event of death or hospitalization.
All statistical analyses was performed using SAS V9.3.
3.1 Results
Patient characteristics are described in table 3. The mean age was 59 ± 11 years, and 76% were male. They were evenly divided among NYHA classes II and III.
Table 3.
Subject Characteristics
| N | 134 |
| Female | 32 |
| Male | 102 |
| Age (years) (± SD) here down 129 pts | 57.2 ± 11.3 |
| Female | 54.6 ± 11.1 |
| Male | 58.0 ± 11.4 |
| Body mass index (± SD) | 30.9 ± 7.5 |
| Female | 30.0 ± 7.1 |
| Male | 33.8 ± 8.1 |
| Race (n) | |
| African American | 92 |
| Caucasian | 41 |
| North American Indian | 1 |
| Current Smoker (%) | 24.6% |
| History of Smoking (%) | 70.9% |
| NYHA Class (n) | |
| II | 74 |
| III | 60 |
| Ischemic Cardiomyopathy (%) | 41.8% |
| Hypertension (%) | 80.45% |
| Ejection fraction (± SD) | 23.1 ± 7.5 |
| Female | 24.6 ± 8.9 |
| Male | 22.6 ± 6.9 |
The correlation of Sgx-ET1 and CT-ET1 was poor, as indicated in figure 1. The overall Spearman correlation was 0.52.
Figure 1.
The relationship between endothelin 1 as measured by the Brahms and Singulex assays. The relationship was poor, with R = 0.52
Over 2.4 ± 1.0-year follow-up, 44 patients (33%) died or were hospitalized for HF. All of the measured biomarkers, with the exception of hs-CRP, were prognostic for these endpoints. The median concentrations are shown in table 4 and the unadjusted and adjusted hazard ratios for this clinical endpoint are shown in table 5.
Table 4.
Comparison of Subjects With and Without the Endpoint of HF Hospitalization or Death
| No HF or Death | Yes HF or Death | ||||
|---|---|---|---|---|---|
| Variable | N | Median | N | Median | p-value |
| Age (yrs) | 90 | 57 | 44 | 59.5 | 0.53 |
| IL-6 (pg/mL) | 90 | 3.1 | 44 | 4.5 | 0.06 |
| TNFα (pg/mL) | 90 | 4.3 | 44 | 6 | 0.02 |
| cTnI (pg/mL) | 90 | 7.4 | 44 | 14.2 | 0.005 |
| VEGF(pg/mL) | 90 | 35.3 | 44 | 46.3 | 0.04 |
| CRP (mg/L) | 88 | 3.5 | 42 | 4.6 | 0.54 |
| CT-ET1 (pmol/L) | 90 | 72.3 | 44 | 110 | <.0001 |
| Sgx-ET1 (pg/mL) | 90 | 4.1 | 44 | 5.2 | <.0001 |
| NTproBNP (pg/mL) | 90 | 980 | 44 | 3019 | <.0001 |
Table 5.
Hazard Ratio for the Endpoint of HF Hospitalization or Death by Cutpoint
| Unadjusted Cox Regression | Adjusted Cox Regression * | |||
|---|---|---|---|---|
| Biomarker | Hazard Ratio (95% CI) | P-value | Hazard Ratio (95% CI) | P-value |
| Sgx-ET1 | 3.8 (1.9, 7.5) | 0.0001 | 2.6 (1.2,5.6) | 0.02 |
| CT-ET1 | 6.4 (3.0,13.8) | <0.0001 | 3.4 (1.4,8.4) | 0.01 |
| IL6 | 3.4 (1.6,7.3) | 0.0020 | 2.1 (0.8,5.3) | 0.12 |
| TNFα | 2.4 (1.3,4.4) | 0.0046 | 1.3 (0.7,2.7) | 0.42 |
| VEGF | 2.5 (1.3,4.9) | 0.0060 | 2.7 (1.3,5.4) | 0.01 |
| cTnI | 3.0 (1.5,5.9) | 0.0016 | 1.4 (0.6,3.0) | 0.41 |
| NTproBNP | 3.7 (1.9,7.1) | <0.0001 | 1.1 (0.5,2.4) | 0.85 |
Adjusted for biomarkers, age, and sex
When comparing patients with logistic regression modeling for time to HF hospitalization or death as the outcome after adjusting for age, sex and other significant biomarkers, CT-ET1 (OR 5.2, 95% CI 1.7–15.7) and Sgx-ET1 (2.9, CI 1.1–7.7) were independent predictors and additively predicted events. The other biomarkers did not improve the model. When the model was adjusted for race and eGFR, the results were similar, with OR = 4.8 (95% CI 1.3–18.1) for CT-ET1 and OR = 3.2 (CI 1.1–9.3) for Sgx-ET1. Similar results were obtained when adding ejection fraction, NYHA class, etiology, race, eGFR and the presence or absence of diabetes mellitus to the model.
In a Cox regression analysis, only CT-ET1 (HR 3.4, 95% CI 1.4–8.4), VEGF (2.7, 95% CI 1.3–5.4) and Sgx-ET1 (HR 2.6, 95% CI 1.2–5.6) were independently prognostic.
By Kaplan-Meier analysis, the two ET1 assays added complementary information; the Kaplan-Meier curves are displayed in Figure 2. When Sgx-ET1 was divided into quartiles, the highest two quartiles had significantly worse prognosis than the lowest two quartiles (figure 3). This was true for CT-ET1 as well.
Figure 2.
Kaplan Meier survival curves of hospitalization or death in groups defined by having values above or below the median for the two endothelin 1 assays. All comparisons are significantly different except Sgx-ET1 high, CT-ET1 low compared with Sgx-ET1 low, CT-ET1 high.
Figure 3.
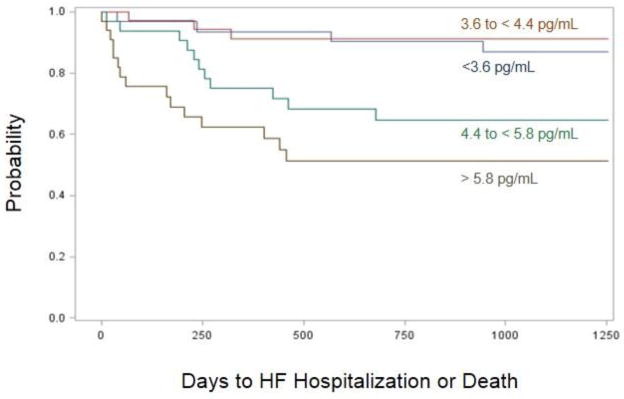
Kaplan Meier survival curves of hospitalization or death by ET1 quartiles. Patients with values in the lowest 2 quartiles have significantly worse outcomes.
Unadjusted analyses showed an OR for hospitalization or death of 4.0 (1.7–9.0) for galectin 3 and 3.0 (1.3–7.1) for ST2. However, when galectin 3 and ST2 were included in logistic regression analyses (in 115 patients), they did not add independent prognostic information (table 6).
Table 6.
Odds Ratio for the Endpoint of HF Hospitalization or Death, including Galectin 3 and ST2
| Unadjusted Logistical Regression | Adjusted Logistical Regression * | |||
|---|---|---|---|---|
| Biomarker | OR (95% CI) | P value | OR (95% CI) | P value |
| NTproBNP | 4.3 (1.6,11.4) | 0.0033 | 1.1 (0.2,5.1) | 0.90 |
| IL-6 | 2.9 (1.3,6.4) | 0.0277 | 1.1 (0.2,5.3) | 0.86 |
| TNFα | 2.6 (1.2,5.5) | 0.0108 | 0.9 (0.3,2.7) | 0.85 |
| cTnI | 3.9 (1.8,8.7) | 0.0008 | 2.0 (0.7,5.6) | 0.17 |
| VEGF | 2.9(1.3,6.4) | 0.0073 | 1.7 (0.6,4.6) | 0.28 |
| Sgx-ET1 | 4.7(2.1,10.5) | 0.0002 | 2.9 (1.1,7.8) | 0.03 |
| CT-ET1 | 9.0(3.7,21.7) | <.0001 | 4.2 (1.2,14.2) | 0.02 |
| galectin3 | 4.0(1.7,9.0) | 0.0010 | 1.9(0.7,5.4) | 0.24 |
| ST2 | 3.0(1.3,7.1) | 0.0106 | 0.7(0.2,2.3) | 0.58 |
Adjusted for biomarkers, age, and sex
15 of 134 patients died; 7 of these 15 did not have a hospitalization. CT-ET1 was the only significantly different biomarker for mortality after adjusted for age, sex and other biomarkers, (p = 0.0486, OR = 5.9 (1.0,35.0))
4.1 Discussion
This study demonstrated the strong prognostic ability of ET1 for predicting hospitalization or death. Furthermore, two assays for ET1, one for the active molecule (Sgx-ET1) and the other for an inactive metabolite (CT-ET1), were both independently prognostic and both were more strongly associated with outcomes than the other biomarkers investigated. These data suggest that elevations in ET1 either leads to a worse prognosis or is an extremely sensitive marker of heart failure severity (or both).
4.1.1 Comparison with Other Biomarkers
Prior studies have shown that ET1 is prognostic in heart failure, and have compared it to selected biomarkers. In comparison to natriuretic peptides, ET1’s prognostic importance has been mixed. Val-HeFT, a study of 2359 patients, showed that big ET1 was prognostic of morbidity and mortality, but BNP was more prognostic.2 In contrast, ET1 was more prognostic of death than BNP and N-proANP in univariate and multivariate regression analyses in another study of 109 patients3 and only big ET1 and ET1 independently predicted death in 47 patients.1 Similarly, chromogranin A and C-terminal endothelin precursor fragments were shown to add prognostic information to NT-proBNP in 127 patients.4
Prognostic comparisons with other biomarkers have previously been limited to selected biomarkers.5,6,7 While endothelin 1 has usually been highly prognostic, results have varied. Undoubtedly, the population studied and the characteristics of the assays can affect the relative prognostic importance of a biomarker. Low event rates might also have affected the results, but endothelin 1 generally predicts both mortality and hospitalization rate.
The present study included a wide range of neurohormonal, inflammatory and fibrotic markers. All assessed biomarkers, both novel and traditional, were found to be predictive of deteriorating clinical status among HF patients, with the exception of hs-CRP. However, both endothelin 1 assays were independently prognostic, while other biomarkers were not. We believe that since the present study is relatively small, the marked superiority of endothelin I to the other multiple biomarkers (both common and uncommon) should be further evaluated and needs to be replicated in larger studies.
4.1.2 Comparison of Endothelin 1 Assays
In the present study, CT-ET1 and Sgx-ET1 provided independent and additive prognostic value in patients with HF. Other prognostic biomarkers (with the exception of VEGF) did not provide additional power to these two biomarkers in adjusted Cox Regression or OR models. There are multiple possible explanations for the marked and independent prognostic importance of the endothelin 1 assays.
First, measurement of the two forms of the ET1 peptide may provide independent and complementary information. For example, the varying half-lives of these biomarkers may explain the independent prognostic contribution of the two endothelin assays. It is possible that the two forms of these ET1 peptides may provide an index of two distinct processes in HF pathophysiology. It is also possible that the variability of the assay (both physiologic and laboratory) is the cause of the poor correlation between assays. If that is the case, the independent prognostic importance of the two assays could be secondary to the strength of the prognostic importance of endothelin 1.
4.1.3 Limitations
The primary limitation of the present study is the relatively small number of patients evaluated. While the relationships of biomarker concentrations with both mortality and hospitalization were similar, it is possible that the strength of the relationship might be different in larger studies or studies in which a different patient population is studied. The specific biomarkers examined in this study might also impact the results.
4.1.4 Conclusions
The present study demonstrated that elevated concentrations of endothelin 1 predict both mortality and hospitalizations in patients with heart failure. Endothelin 1 was more prognostic than commonly obtained hemodynamic, inflammatory and fibrotic biomarkers. Furthermore, two different assays of endothelin 1 independently and synergistically were prognostic, suggesting either complementary information or extreme prognostic importance. Further studies of biomarkers should include endothelin 1 to further clarify the importance of this biomarker.
Highlights.
Elevated concentrations of endothelin 1 predicted both mortality and hospitalizations in patients with heart failure and the addition of other biomarkers to logistic regression analysis did not improve the model.
The Singulex assay, which measures the active form of ET1 and has a short in-vivo half-life, and the Brahms assay, which measures C-terminal endothelin-1 and is a modified (degraded) product with longer half-life, were independently and synergistically prognostic.
The findings of the present study suggest that either the 2 forms of endothelin 1 provide complementary information or that endothelin 1 has extreme prognostic importance.
Acknowledgments
Supported by NHLBI grant 1R01 HL085730.
We would like to thank BG Medicine for providing the reagents for Galectin 3 assays and Critical Diagnostics for providing the reagents for the ST2 assays.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Van Beneden R, Gurné O, Selvais PL, et al. Superiority of big endothelin-1 and endothelin-1 over natriuretic peptides in predicting survival in severe congestive heart failure: a 7 year follow-up study. J Card Failure. 2004;10:490–5. doi: 10.1016/j.cardfail.2004.04.001. [DOI] [PubMed] [Google Scholar]
- 2.Masson S, Latini R, Anand IS, et al. The prognostic value of big endothelin-1 in more than 2300 patients with heart failure enrolled in Valsartan Heart Failure Trial (Val-HeFT) J Card Failure. 2006;12:375–380. doi: 10.1016/j.cardfail.2006.02.013. [DOI] [PubMed] [Google Scholar]
- 3.Selvais PL, Robert A, Ahn S, et al. Direct comparison between endothelin-1, N-terminal proatrial natriuretic factor, and brain natriuretic peptide as prognostic markers of survival in congestive heart failure. J Cardiac Failure. 2000;6:201–7. doi: 10.1054/jcaf.2000.8833. [DOI] [PubMed] [Google Scholar]
- 4.Dieplinger B, Gegenhuber A, Struck J, et al. Chromogranin A and C-terminal endoythelin-1 precursor fragment add independent prognostic information to amino-terminal proBNP in patients with acute destabilized heart failure. Clinica Chimica Acta. 2009;400:91–96. doi: 10.1016/j.cca.2008.10.012. [DOI] [PubMed] [Google Scholar]
- 5.Masson S, Latini R, Carbonieri E, Moretti L, Rossi MG, Ciricugno S, Milani V, Marchioli R, Struck J, Bergmann A, Maggioni AP, Tognoni G, Tavazzi L GISSI-HF Investigators. The predictive value of stable precursor fragments of vasoactive peptides in patients with chronic heart failure: data from the GISSI-heart failure (GISSI-HF) trial. Eur J Heart Fail. 2010;12:338–47. doi: 10.1093/eurjhf/hfp206. [DOI] [PubMed] [Google Scholar]
- 6.Pousset F, Masson F, Chavirovskaia O, Isnard R, Carayon A, Golmard JL, Lechat P, Thomas D, Komajda M. Plasma adrenomedullin, a new independent predictor of prognosis in patients with chronic heart failure. Eur Heart J. 2000;21:1009–14. doi: 10.1053/euhj.1999.1904. [DOI] [PubMed] [Google Scholar]
- 7.Neuhold S, Huelsmann M, Strunk G, Struck J, Adlbrecht C, Gouya G, Elhenicky M, Pacher R. Prognostic value of emerging neurohormones in chronic heart failure during optimization of heart failure-specific therapy. Clin Chem. 2010;56:121–6. doi: 10.1373/clinchem.2009.125856. [DOI] [PubMed] [Google Scholar]




