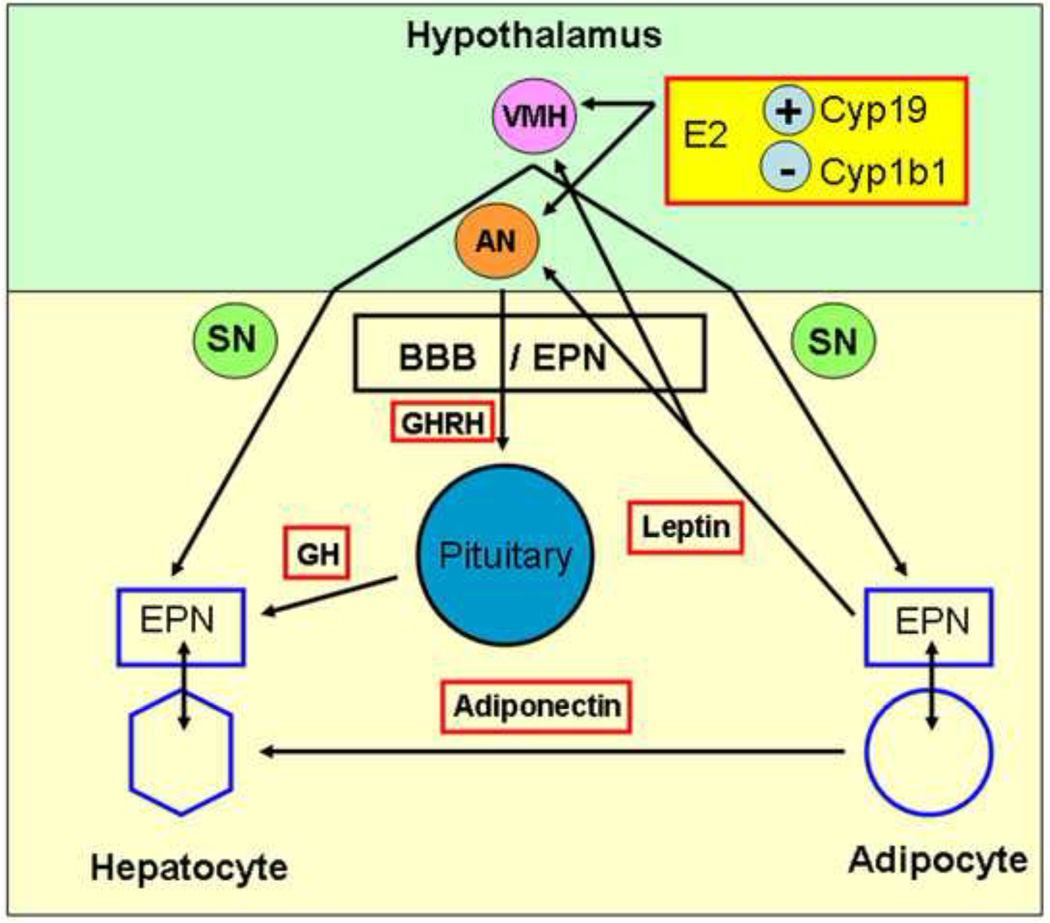
Figure 9. Representation of proposed systemic effects of Cyp1b1 deletion that increase effects of leptin and growth hormone on the liver.
Cyp1b1 deletion effects on liver gene expression indicate increased leptin and growth hormone action, each of which are regulated in the hypothalamus. Hypothesis: Cyp1b1 deletion targets two important activities A) The endothelial-pericyte niche (EPN), which provides local regulation of adjacent cells, is disrupted in Cyp1b1-ko mice, B) Estradiol (E2) levels are increased, particularly in the hypothalamus, when local removal of E2 (produced by aromatase) via Cyp1b1 metabolism is decreased. Increased transfer of leptin across the blood brain barrier (BBB) increases activation of sympathetic neurons via the ventromedial hypothalamus (VMH) and release of GH releasing hormone (GHRH) from the arcuate nucleus (AN), which stimulates pulsatile release of GH from the pituitary [21, 101]. Similar EP niche activity functions in liver sinusoids (endothelia and stellate pericytes) [71] and adipose [32] to provide local controls over adjacent hepatocytes and adipocytes, respectively.