Abstract
Ovarian cancer continues to be a leading cause of cancer related deaths for women. Anticancer agents effective against chemo-resistant cells are greatly needed for ovarian cancer treatment. Repurposing drugs currently in human use is an attractive strategy for developing novel cancer treatments with expedited translation into clinical trials. Therefore, we examined whether ormeloxifene (ORM), a non-steroidal Selective Estrogen Receptor Modulator (SERM) currently used for contraception, is therapeutically effective at inhibiting ovarian cancer growth. We report that ORM treatment inhibits cell growth and induces apoptosis in ovarian cancer cell lines, including cell lines resistant to cisplatin. Furthermore, ORM treatment decreases Akt phosphorylation, increases p53 phosphorylation, and modulates the expression and localization patterns of p27, cyclin E, cyclin D1, and CDK2. In a pre-clinical xenograft mouse ORM treatment significantly reduces tumorigenesis and metastasis. These results indicate that ORM effectively inhibits the growth of cisplatin resistant ovarian cancer cells. ORM is currently in human use and has an established record of patient safety. Our encouraging in vitro and pre-clinical in vivo findings indicate that ORM is a promising candidate for the treatment of ovarian cancer.
Keywords: Ormeloxifene, Novel therapies, Cisplatin resistance, Ovarian cancer, Apoptosis, Xenograft mice
1. Introduction
Ovarian cancer is the fifth leading cause of cancer mortality in women in the U.S. [1]. Due to the lack of clear symptoms and effective screening mechanisms, 62% of ovarian cancers are diagnosed at the distant stage, which has an average 5 year survival of only 27% [1]. While most ovarian cancer patients will initially respond to chemotherapy, over 66% of patients with advanced stage disease will relapse with disease that becomes resistant to current treatment options [2], highlighting a clear need for additional therapies to treat both initial and relapsed ovarian cancers.
While the discovery and characterization of de novo drugs may yield new therapies, there is an appealing option of identifying an effective ovarian cancer therapeutic from a compound that is already in human use, which would dramatically shorten the time and resources required to provide a new treatment option to patients. Ormeloxifene (ORM, also known as Centchroman) is a non-hormonal, non-steroidal oral method of contraception widely used in India [3, 4]. In an early report, Misra et al. (1989) conducted a trial on advanced breast cancer patients and suggested that ORM may be effective at inhibiting breast cancer [5]. About 38.5% of breast cancer female patients responded to the ormeloxifene therapy and the response to ormeloxifene treatment was more promising for bone, pulmonary, soft tissue, skin, and lymph-node metastases. More recently, ORM has shown anti-cancer effects with in vitro models of breast cancer, head and neck cancer, and chronic myeloid leukemia [6–11]. Moreover ORM is reported to have an excellent therapeutic index and is safe for chronic administration [12].
Herein, we have examined the effects of ORM on the growth of cisplatin sensitive (A2780) and cisplatin resistant (A2780-CP and SKOV3) ovarian cancer cell lines. We show evidence that ORM induces apoptosis and is capable of modulating several proteins involved in cell cycle regulation. ORM efficiently inhibited the growth and spread of ovarian cancer cells in a pre-clinical mouse model of ovarian cancer. Together, this data suggests that ORM may be an effective therapeutic for ovarian cancer and its history of safe human use provides additional evidence for the promising translation of ORM into clinical practice.
2. Materials and Methods
2.1. Cell culture, growth conditions, and treatment
The human ovarian carcinoma cell line SKOV3 was purchased from ATCC and upon receipt cells were expanded and frozen aliquots (passage < 6) were stored in liquid nitrogen. When needed, cells were thawed and grown for less than 6 months. The paired ovarian cancer cells lines, A2780 and A2780-CP cells were a gift from Dr. Howell (University of CA, San Diego). A2780-CP cells are a cisplatin resistant cell line derived from the parental A2780 cells [13]. SKOV3, also considered to be cisplatin resistant, was grown in DMEM supplemented with 10% fetal bovine serum (Atlanta Biologicals, Lawrenceville, GA), 10 nM non-essential amino acids, 100 nM sodium pyruvate, and 1× antibiotic/antimycotic (Gibco BRL, Grand Island, NY). A2780 and A2780cp were maintained as monolayer cultures in RPMI-1640 medium (HyClone Laboratories, Inc. Logan, UT) supplemented with 10% fetal bovine serum (Atlanta Biologicals) and 1× antibiotic/antimycotic (Gibco). All cells were cultured at 37°C in a humidified atmosphere (5% CO2). ORM was generously synthesized and provided by FH as described earlier [14]. ORM was solubilized in 100% ethanol and at the time of treatment, ORM was diluted into fresh cell culture media.
2.2. Cell Proliferation Assays
Cells were seeded at 5,000 cells per well in 96-well plates and allowed to attach overnight before ORM was added at various concentrations as indicated. Ethanol containing medium served as the vehicle control. The anti-proliferative effect of ORM was determined at 2 days using the CellTiter 96 AQeous One solution assay (Promega, Madison, WI) as described earlier [15]. The CellTiter reagent was added to each well (20 µL/well) and plates were incubated for 2 hrs at 37°C. The color intensity was measured at 492 nm using a microplate reader (BioMate 3 UV-Vis spectrophotometer, Thermo Electron Corporation, Waltham, MA). The anti-proliferative effect of each treatment was calculated as a percentage of cell growth with respect to the vehicle control.
2.3. Clonogenic assay
For the clonogenic assay, cells were seeded at 500 cells per 100 mm culture dish and allowed to attach overnight. The cells were treated with ORM (or ethanol for the vehicle control) and maintained under standard cell culture conditions at 37°C and 5% CO2 in a humid environment. After 10 days, the dishes were washed twice in PBS, fixed with methanol, stained with hematoxylin (Fisher Scientific, Pittsburgh, PA), washed with water and air dried. Visible colonies (~50 cells) were counted and the percent of colonies was calculated using the number of colonies formed in treatment divided by number of colonies formed in the vehicle control, as described earlier [16].
2.4. Cell Cycle and TUNEL analysis
Cells (5.0 × 105) were plated in a 100 mm dish and allowed to attach overnight. Cells were then exposed to ORM (10 and 20 µM) for 48 hours. After ORM treatment all cells (including those floating in the media) were collected, fixed with 70% ethanol, stained with Telford Reagent containing propidium iodide, incubated overnight at 4°C, and analyzed with an Accuri C6 flow cytometer (BD Biosciences). Cells with hypodiploid DNA (content less than G0/G1) were deemed apoptotic (sub- G0/G1). Alternatively, after ORM treatment, all cells were collected and prepared according to the APO-BrdU™ TUNEL Assay Kit (Invitrogen, Life Technologies) and analyzed with the Accuri C6 flow cytometer.
2.5. Mitochondrial Membrane Potential (ΔΨm)
Mitochondrial membrane potential (ΔΨm) was measured by uptake of tetramethylrhodamine (TMRE). TMRE is sequestered by active mitochondria and undergoes a dramatic increase in fluorescence intensity. Briefly, cells were treated with ORM (10–20 µM) at three different time points (6, 12, and 18 hours), incubated in 100 nM TMRE for 20 minutes, and fluorescence intensities were measured by flow cytometry. Mean fluorescence values are shown as a percent of the vehicle control (ethanol) values.
2.6. Western blotting
Whole cell lysates were prepared as described earlier [17]. Briefly, cells (1×106) were allowed to attach overnight and then treated with ORM for the indicated times. After ORM treatment, both adherent and floating cells were collected in 2× SDS lysis buffer (Santa Cruz Biotechnologies), sonicated, and the protein concentration was normalized using SYPRO Orange (Molecular Probes). SDS-PAGE electrophoresis was performed with a 4–20% gradient gel and resolved proteins were transferred onto a PVDF (BioRad) membrane. The following antibodies were used: PARP, Caspase 3, Caspase 9, Rb, phospho-p53 (ser-15), p21, p27, pAKT (ser-473) (each from Cell Signaling Technologies); p53, Cyclin E, and CdK2 (each from Santa Cruz Biotechnologies); and β-actin (Sigma). The primary antibody was detected by a species appropriate HRP-secondary antibody (Promega) followed by incubation with the Lumi-Light detection reagent (Roche).
2.7. Confocal microscopy
Cells were plated in 4 well chamber slides at 5×104 cells per chamber. Cells were allowed to attach and reach 60% confluency (approximately 24 hr) before being treated with 10uM ORM for 18 hr. Cells were then processed for confocal microscopy as described earlier [16]. Briefly, following treatment cells were rinsed with 1× HEPES/Hank buffer, fixed and permeabilized with ice cold methanol, washed with 1× PBS and blocked with 10% normal goat serum in PBS. Cells were then incubated with the primary antibody (source listed in prior section) followed by a species specific Alexa Fluor® 488 secondary antibody (Invitrogen). After washing, cells were stained with DAPI and coverslips were mounted in FluoroCare Anti-Fade mounting medium (BioCare Medical). Confocal microscopy was performed with an Olympus Fluoview FV1000 confocal microscope (Olympus Corporation).
2.8. In vivo tumor xenograft model
Six-week-old female athymic nude (nu/nu) mice (H arlan Laboratories) were injected intraperitoneally with A2780-CP cancer cells (5 × 106 cells in 400ul PBS). Immediately prior to injection, the cells were mixed with ORM (50 or 100ug) or vehicle (ethanol). The mice continued to receive the same treatment ORM (50 or 100µg/mouse) or vehicle (ethanol) via intraperitoneal (ip) injection once a week for four subsequent weeks. Once per week, the mice were also weighed to help monitor their health. On day 35 post cancer cell injection, mice were euthanized, and tumor burden and metastases were noted. Mouse studies were carried out following procedures approved by the Sanford Research Institutional Animal Care and Use Committee.
2.9. Statistical Analysis
All experiments were performed in triplicate and statistical significance (p<0.05) was determined using a two-tailed student t-test with equal variance.
3. Results
3.1. ORM inhibits growth of ovarian cancer cell lines
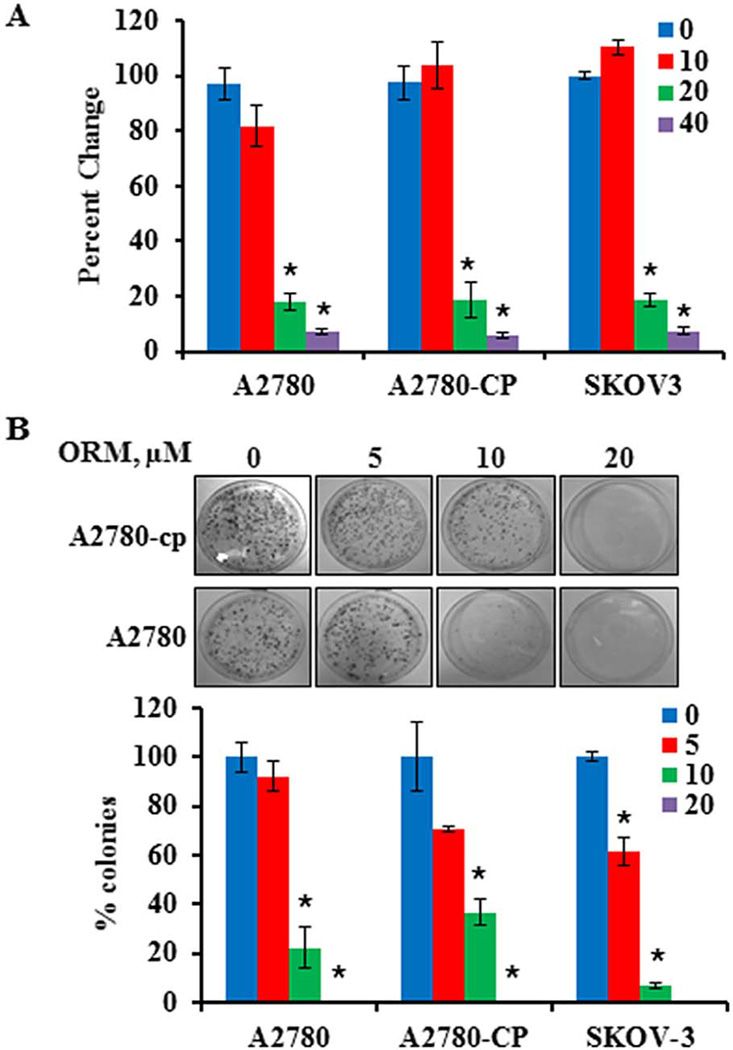
To test whether ORM is effective at inhibiting ovarian cancer cell growth, we evaluated ORM’s effect on ovarian cancer cell lines, including OVCAR-3 (Fig. S1), cisplatin sensitive (A2780), resistant (A2780-CP), and semi-resistant (SKOV-3) cells (Fig. 1). As determined with MTS assays, there was a dramatic decrease in cell growth at doses greater than 10µM (Fig. 1A), and this finding was supported by standard cell counting assays (data not shown). Colony formation assays were performed to determine the longer term effects of ORM on the growth of ovarian cancer cells. ORM treatment significantly reduced clonogenicity in each of the ovarian cancer cell lines tested (Fig. 1B).
Fig. 1. Ormeloxifene treatment inhibits growth of chemo-sensitive (A2780) and chemo-resistant (A2780-CP and SKOV3) ovarian cancer cell lines.
A) Proliferation was determined with an MTS assay 48 hours after ORM addition and normalized to control wells treated with appropriate amounts of vehicle (ethanol, set at 100%, labeled as 0 ORM). Columns: mean, Bars: SE, n=3. *indicates p value: <0.05. B) A colony forming assay was conducted to determine the long term effect of ORM treatment on the clonogenic potential of ovarian cancer cells. Colonies were counted and expressed as a percentage of the number of colonies in the vehicle control (ethanol, set at 100%, labeled as 0 ORM). Columns: mean, Bars: SE, n=3. *indicates p<0.05. Representative plates are shown for A2780-CP cells.
3.2. ORM induced apoptosis is the primary cause of cell death and is linked to the intrinsic apoptotic pathway
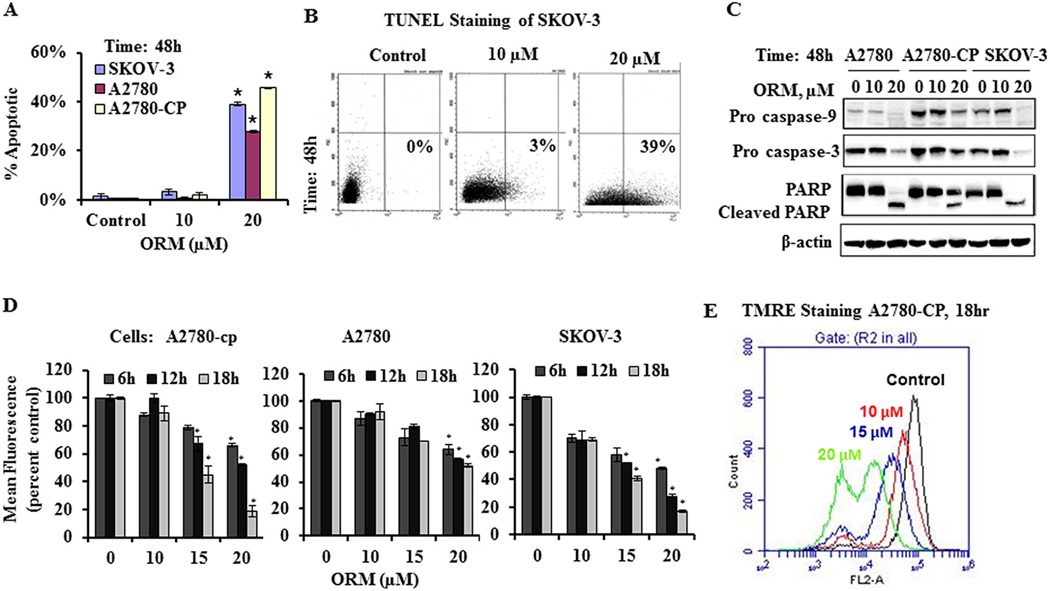
Following ORM treatment, cells displayed typical signs of apoptosis, including cell shrinkage, rounding-up, and blebbing of the cell membrane. To further examine the cell death induced by ORM treatment, we evaluated ORM treated cells for markers of apoptosis. Ovarian cancer cell lines were treated with ORM for 48 hours and then processed for the TUNEL assay to identify DNA fragmentation, a hallmark of late stage apoptosis. Flow cytometry analysis revealed a significant increase in TUNEL positive cells following ORM treatment (Fig. 2A and B). We further investigated the molecular events underlying ORM induced cell death/apoptosis in ovarian cancer cells by immunoblotting for caspases 3 and 9, which are both cleaved in response to ORM treatment as shown by a decrease in the full length proteins (Fig. 2C). In response to ORM treatment, we also observed the cleavage of full length PARP, another classical marker of apoptosis (Fig. 2C).
Fig. 2. ORM treatment causes apoptosis through decreased mitochondrial membrane potential (ΔΨm) and activated apoptosis associated proteins.
A–B) ORM treatment leads to apoptosis. Ovarian cancer cells were treated with ORM for 48 hours and processed for staining with the APO-BrdU™ TUNEL Assay Kit. DNA fragmentation was analyzed by flow cytometry. A) Percent TUNEL positive cells are shown for each cell line tested. *indicates p value: <0.05 compared to vehicle control. B) Representative FSC/FL-1 plots are shown for SKOV-3 cells. Data are representative of one of three similar experiments. C) ORM treatment leads to cleavage of caspases 3 and 9 and PARP. Cells were treated with indicated concentrations of ORM for 48 h, protein lysates were collected, 20µg of protein was resolved by SDS-PAGE and immunoblotted with primary antibodies as indicated. β-actin was used as an internal loading control. Western blots shown are representative from one of two similar experiments. D and E) ORM decreases mitochondrial potential in a dose and time dependent response. Cells were treated with ORM as indicated and live cells were incubated in TMRE and analyzed by flow cytometry. D) Graphs show the change in mean fluorescence values normalized to vehicle control (ethanol, labeled as 0 ORM)). *indicates p value: <0.05. E) A representative histogram of A2780-CP after 18h of ORM treatment is shown. Data are representative of one of three similar experiments.
To assess factors important early in apoptosis, we examined the mitochondrial potential of ovarian cancer cells treated with ORM for 6, 12, and 18 hours. A significant decrease in mitochondrial membrane potential was observed in a both time and dose dependent manner (Fig. 2D and E). While significant in each cell line, the change was especially notable in A2780-CP cells where the mitochondrial potential was decreased by 50% after a 6hr treatment with 20µM ORM (Fig. 2E). Taken together, these findings suggest that ORM treatment induces apoptosis in ovarian cancer cells through a mitochondrial dependent pathway.
3.3. ORM treatment alters cell cycle distribution in ovarian cancer cells
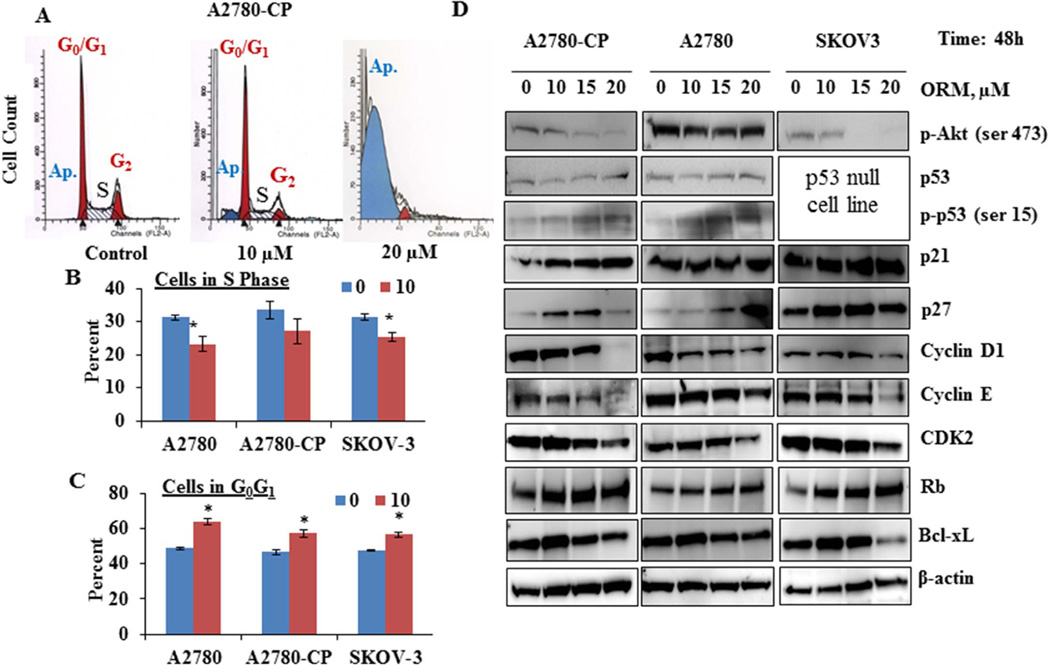
We investigated the effect of ORM on cell cycle phase distribution. Results showed that cells were arrested at the Sub G1 phase, the indicator of apoptosis after treatment with ORM. The cells in Sub G1 phase increased on ORM treatment in all tested ovarian cancer cells. This exceeded upto 80% in A2780-cp cells after 20 µM of ORM treatment, while cells in the S phase were decreased from 13 to 1% in each cell line. While ORM at 20 µM and greater dramatically inhibited cell growth and induced apoptosis, lower doses of ORM decreased the percent of cells in the S-phase of the cell cycle while increasing the percent of cells in G0G1 (Fig. 3 A–C). (Figure 3A).
Fig. 3. ORM treatment induces changes in cell cycle related proteins in ovarian cancer cells.
A–C) ORM treatment alters cell cycle distribution. After 48h of ORM treatment, adherent and floating cells were collected, fixed with 70% ethanol, stained with Telford Reagent containing propidium iodide, incubated overnight at 4°C, and analyzed by flow cytometry for cell cycle analysis. A) Representative histograms are shown for A2780-CP cells. Graphs show the percent of cells in B) G0G1 and C) S phase. *indicates p value: <0.05. D) ORM treatment alters proteins associated with cell cycle progression. Cells were treated with indicated concentrations of ORM for 48 h, protein lysates were collected, separated on SDS-PAGE, and immunoblotted for proteins as indicated. β-actin was used as an internal control. Western blots shown are representative from one of two similar experiments.
3.4. ORM treatment modulates multiple cell cycle proteins
We next investigated the effect of ORM treatment on the modulation of several proteins involved in cell cycle progression (Fig. 3D). ORM treatment dramatically inhibited the phosphorylation of Akt in A2780-CP and SKOV3 cell lines. ORM treatment increased overall levels of total and phosphorylated p53 in A2780-CP and A2780 cells lines. SKOV3 is a p53 null cell line, indicating that ORM may function through both p53 dependent and independent mechanisms. Corresponding with both Akt inhibition and p53 phosphorylation, ORM treatment increased cell cycle inhibitors, p21 and p27. ORM treatment also decreased targets of p21 and p27: cyclin D1, cyclin E and CDK2 at 10µM which further decreased with the increase in concentration. Additionally, ORM treatment increased retinoblastoma protein (Rb), an important tumor suppressor protein involved in cell cycle regulation. Besides the cell cycle related proteins, ORM also inhibited Bcl-xL, which confers resistance to several chemotherapeutic agents in ovarian cancer [18–20].
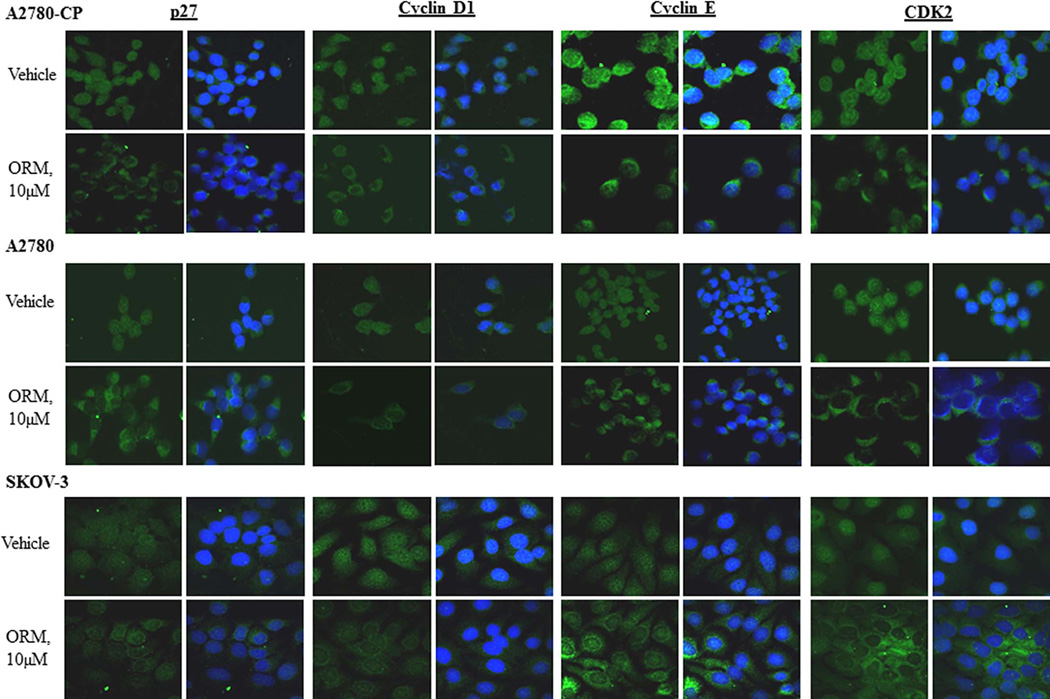
To confirm changes in cell cycle regulatory proteins, we performed immunofluorescence and assessed both protein levels and changes in localization patterns. At an early time point (18hr), treatment with 10 µM ORM clearly reduced the nuclear localization patterns of p27, cyclin D1, cyclin E and CDK2, as shown by confocal microscopy (Fig. 4). Therefore, in addition to inducing apoptosis, ORM treatment also inhibits the AKT pathway and downstream regulators of cell cycle progression.
Fig. 4. ORM treatment decreases nuclear localization of cell cycle related proteins.
Ovarian cancer cells, A2780, A2780-CP and SKOV-3 cells were treated with 10 µM ORM as indicated for 18h, fixed, and stained for the detection of p27, cyclin D1, cyclin E, and CDK2 (green staining) with DAPI counter stain (nuclear marker, blue). Confocal fluorescence microscopy shows that ORM decreases nuclear localization of cell cycle related proteins. (Image on the left is green only, right hand image is merged with the DAPI nuclear counter stain).
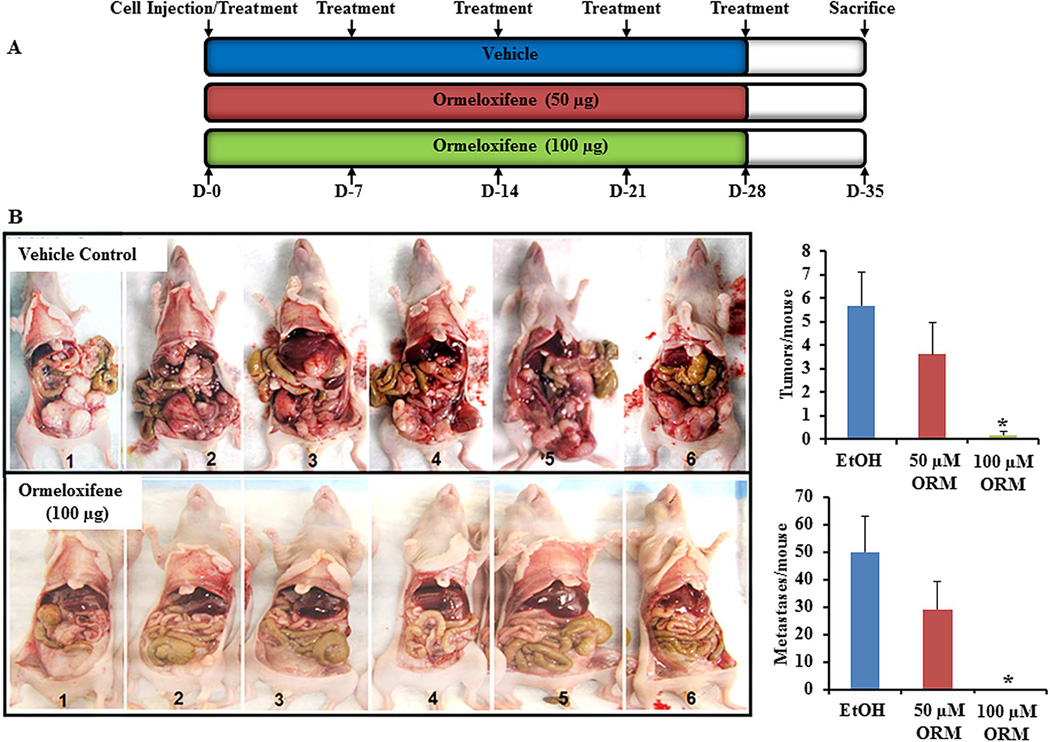
3.5. ORM reduced ovarian cancer growth in a pre-clinical mouse model
To further support the anti-cancer effects of ORM, we tested ORM treatment in a pre-clinical murine ovarian cancer xenograft model with cisplatin resistant A2780-CP cells. Mice were treated with a scheduled dose of ORM (or vehicle) as indicated for five consecutive weeks, beginning at the time of intraperitoneal injection of the tumor cells (Fig. 5A). The mice showed no apparent side effects from the used doses of ORM. In vehicle treated mice, a swollen abdomen, indicative of ascites formation along with heavily peritoneal carcinomatosis, was observed within 2 weeks of A2780-CP cell injection. While 100% mice receiving vehicle developed ascites and exhibited number of solid tumors, mice treated with the dose (100µg) of ORM had no detectable tumors. Upon further examination, we also found that there were almost no/fewer metastases in the treatment (ORM) groups compared to control groups (Fig. 5B). All mice were sacrificed at day 35 post cancer cell injection because vehicle-treated animals appeared with severe ascites formation and visible tumor growth in accordance with the animal care protocol. However, ORM treated mice did not exhibit a sign of ascites formation and tumor growth. These data further confirm that ORM could be an effective chemotherapeutic agent for ovarian cancer.
Fig. 5. ORM inhibits ovarian cancer tumorigenesis in mice.
A) Schedule of ormeloxifene treatment in ovarian cancer xenograft mouse model. Mice were injected with 50 or 100 µg ORM once a week for 5 weeks as indicated (D=days). Ethanol (EtOH) was the vehicle control. B) Images of mice from each group injected with ORM or ethanol control. Graphs show the average number of tumors (top) and metastases (bottom) observed in mice treated with ORM or ethanol (vehicle control). Data expressed as mean ± SE of 6 mice in each group. *indicates p value: <0.001.
4.0. Discussion
Despite advances in therapy, overall survival of women with ovarian cancer remains poor [1]. Cisplatin, or carboplatin combined with paclitaxel, remain the most commonly used chemotherapeutic agents, but most patients with recurrent disease develop multi-drug resistance [21], indicating the need for more effective agents against ovarian cancer. While new compounds may be discovered, it is also useful to evaluate currently used drugs for their anti-cancer potential. In keeping with this strategy, our current findings show ORM efficiently inhibits ovarian cancer cell growth. ORM has been in use as a post-coital or weekly oral contraceptive in India since 1991 and has proven to be safe and effective at preventing pregnancy at a dose of 30 mg once a week [22]. ORM may also be a useful agent for hormone-related clinical disorders, such as dermatitis, osteoporosis, restenosis, and uterine fibroids [22]. Additionally, ORM has exhibited anti-cancer activity in cell lines derived from breast, head and neck, and chronic myeloid leukemia cancers [6–11].
Resistance to platinum-based therapies is an important limitation of current ovarian cancer therapy [21]. Apoptosis is a common response to many classes of chemotherapeutic agents, including platinum-based therapies. In the current study, ORM treatment (20 µM and higher) induced apoptosis in the ovarian cancer cell lines tested, including A2780-CP and SKOV-3 cell lines, which are resistant to cell death induced by cisplatin. To assess involvement of mitochondrial factors, we examined the effect of ORM on mitochondrial potential, as measured by TMRE fluorescence. Mitochondrial outer membrane permeabilization (MOMP) is considered the ‘point of no return’ for apoptosis and the onset of MOMP is associated with a loss of mitochondrial potential (ΔΨm) [23]. ORM treatment decreased the mitochondrial membrane potential (measured by TMRE fluorescence) in both time- and concentration-dependent manner in all ovarian cancer cell lines tested. ORM inhibited the pro-survival protein, Bcl-xL, likely increasing caspase activation and apoptosis [18–20]. The mitochondria and associated proteins have been recognized as important targets for developing new cancer therapies [24], further suggesting that ORM may be a highly effective ovarian cancer treatment, even in platinum resistant situations.
Uncontrolled cell cycle progression is a hallmark of cancer cells [25]. Cell cycle progression is regulated by the activity of cyclins, a family of proteins that activate the cell-cycle dependent kinases (Cdks) [26]. Cyclin E activates CDK2 protein near the start of the S phase and cyclin D1 is a critical regulator of the G0/G1 phase [27]. At low doses (i.e. 10 µM) ORM did not induce apoptosis within 48 hours, but did decrease the population of cells in the S-phase of the cell cycle while increasing the population of cells in G0G1. Upon examination, ORM treatment altered the expression of several cell cycle related proteins in a manner that would be expected to decrease progression through the cell cycle. This finding is consistent with the results found in breast cancer cells where ORM induced apoptosis, altered the cell cycle related proteins and arrested cells at the G0/G1 cell cycle phase [6, 8].
Akt is also known to promote chemoresistance in human ovarian cancer cells [28, 29] and pharmacologic agents that inhibit Akt signaling might preferentially kill tumor cells that have a dependence on Akt activity for survival not shared by normal cells [30, 31]. Srivastava et al. (2011) have demonstrated that ORM inhibits PI3K/AKT pathway in Head and neck cancer [9]. Similar effects were found in ovarian cancer where ORM treatment decreased Akt phosphorylation, which is a critical modulator of cell replication and inhibits downstream cell cycle inhibitor pathways, such as p53, p21 and p27. Interestingly, in our study ORM increased cell cycle inhibitors, p21 and p27 as well as phosphorylation of p53. In ovarian carcinoma, p53 mutations may confer resistance to DNA-damaging agents as a consequence of reduced cell ability to undergo apoptosis [32]. ORM treatment induced apoptosis in p53 null cell line, SKOV3, in a manner similar to A2780 and A2780-CP, suggesting a p53-independent pathway of cell cycle inhibition and apoptosis.
The downstream targets of p21 and p27 (cyclin E, cyclin D1 and CDK2) were decreased in a dose dependent manner. Immunofluorescence analysis revealed a dramatic change in nuclear localization of these same proteins following 18 hours of treatment with 10 µM of ORM. This data provides a possible explanation for the observed increase in G0G1 population and corresponding decrease in cells in the S phase after ORM treatment and suggests that ORM may act through multiple molecular pathways to reduce ovarian cancer cell growth.
We employed a pre-clinical xenograft mouse model of ovarian cancer to investigate ORM’s in vivo efficacy on cisplatin resistant A2780-CP ovarian cancer cells. When administered via IP injection, ORM was very effective at inhibiting both tumor formation and metastasis within the peritoneal cavity. In healthy female volunteers, plasma levels of ORM have been reported to reach approximately 122ng/ml following a 60mg dose of ORM [33]. Additionally, ORM has been shown to accumulate in tissues at levels higher than detected in plasma [34]. In our study, we administered ORM through intraperitoneal injection at relatively high, but reasonable doses. Subsequent studies are needed to determine the efficacious dose required for oral delivery of ORM. Importantly, ORM treatment did not result in any observed adverse effects in mice and was well tolerated.
In conclusion, given the safe history of ORM use in humans, ORM is a promising candidate for ovarian cancer treatment. Our current work identifies both apoptosis and cell cycle disruption as methods of effective inhibition of ovarian cancer cell growth in both cisplatin sensitive and resistance cell lines. This study shows the potential of ormeloxifene as a therapeutic agent for ovarian cancer.
Supplementary Material
Highlights for the Review.
Results demonstrate that Ormeloxifene is effective at inhibiting ovarian cancer growth.
Effective inhibition of cisplatin sensitive and resistance ovarian cancer cell growth involves apoptosis and cell cycle disruption.
Therapeutic effects were assessed in pre-clinical mouse model of cisplatin resistant human ovarian cancer.
Ormeloxifene treatment efficiently reduces tumor load and metastasis in a mouse model.
Ormeloxifene is prospected to be an interesting molecule that can be useful for ovarian cancer treatment.
Acknowledgements
We thank Dr. Stephen B. Howell, University of California, San Diego, for providing cell lines (A2780 and A2780-CP) that were used in the study. This project is supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under grant number 5P20GM103548 (Cancer CoBRE, to K. Miskimins); Department of Defense (PC073887 to SCC and PC073643 MJ); and the National Institutes of Health (RO1 CA142736 to SCC and UO1 CA162106A to SCC and MJ). The authors thank Cathy Christopherson (Sanford Research/ USD) for editorial assistance.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Potential conflicts of interest
The authors declare that there are no financial and non-financial competing interests.
Contributor Information
Diane M. Maher, Email: Diane.Maher@sanfordhealth.org.
Sheema Khan, Email: skhan14@uthsc.edu.
Jordan Nordquist, Email: jlnordquist@ole.augie.edu.
Mara C. Ebeling, Email: ebeli017@umn.edu.
Nichole A. Bauer, Email: nikki4370@gmail.com.
Lucas Kopel, Email: Lucas.kopel@sdstate.edu.
Man Mohan Singh, Email: singhmm@rediffmail.com.
Fathi Halaweish, Email: Fathi.Halaweish@sdstate.edu.
Maria C. Bell, Email: Maria.Bell@sanfordhealth.org.
Meena Jaggi, Email: mjaggi@uthsc.edu.
Subhash C. Chauhan, Email: schauha1@uthsc.edu.
References
- 1.Siegel R, Naishadham D, Jemal A. Cancer statistics, 2013. CA. Cancer J. Clin. 2013;63:11–30. doi: 10.3322/caac.21166. [DOI] [PubMed] [Google Scholar]
- 2.Coleman RL, Monk BJ, Sood AK, Herzog TJ. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nat. Rev. Clin. Oncol. 2013;10:211–224. doi: 10.1038/nrclinonc.2013.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bansode FW, Chauhan SC, Makker A, Singh MM. Uterine luminal epithelial alkaline phosphatase activity and pinopod development in relation to endometrial sensitivity in the rat. Contraception. 1998;58:61–68. doi: 10.1016/s0010-7824(98)00055-9. [DOI] [PubMed] [Google Scholar]
- 4.Kamboj VP, Setty BS, Chandra H, Roy SK, Kar AB. Biological profile of Centchroman--a new post-coital contraceptive. Indian J. Exp. Biol. 1977;15:1144–1150. [PubMed] [Google Scholar]
- 5.Misra NC, Nigam PK, Gupta R, Agarwal AK, Kamboj VP. Centchroman--a non-steroidal anti-cancer agent for advanced breast cancer: phase-II study. Int. J. Cancer. 1989;43:781–783. doi: 10.1002/ijc.2910430506. [DOI] [PubMed] [Google Scholar]
- 6.Nigam M, Ranjan V, Srivastava S, Sharma R, Balapure AK. Centchroman induces G0/G1 arrest and caspase-dependent apoptosis involving mitochondrial membrane depolarization in MCF-7 and MDA MB-231 human breast cancer cells. Life Sci. 2008;82:577–590. doi: 10.1016/j.lfs.2007.11.028. [DOI] [PubMed] [Google Scholar]
- 7.Mishra R, Tiwari A, Bhadauria S, Mishra J, Murthy PK, Murthy PS. Therapeutic effect of centchroman alone and in combination with glycine soya on 7,12-dimethylbenz[alpha]anthracene-induced breast tumor in rat. Food Chem. Toxicol. 2010;48:1587–1591. doi: 10.1016/j.fct.2010.03.029. [DOI] [PubMed] [Google Scholar]
- 8.Nigam M, Singh N, Ranjan V, Zaidi D, Sharma R, Nigam D, Gupta DK, Sundaram S, Balapure AK. Centchroman mediated apoptosis involves cross-talk between extrinsic/intrinsic pathways and oxidative regulation. Life Sci. 2010;87:750–758. doi: 10.1016/j.lfs.2010.10.015. [DOI] [PubMed] [Google Scholar]
- 9.Srivastava VK, Gara RK, Bhatt ML, Sahu DP, Mishra DP. Centchroman inhibits proliferation of head and neck cancer cells through the modulation of PI3K/mTOR pathway. Biochem. Biophys. Res. commun. 2011;404:40–45. doi: 10.1016/j.bbrc.2010.11.049. [DOI] [PubMed] [Google Scholar]
- 10.Pal P, Kanaujiya JK, Lochab S, Tripathi SB, Bhatt ML, Singh PK, Sanyal S, Trivedi AK. 2-D gel electrophoresis-based proteomic analysis reveals that ormeloxifen induces G0–G1 growth arrest and ERK-mediated apoptosis in chronic myeloid leukemia cells K562. Proteomics. 2011;11:1517–1529. doi: 10.1002/pmic.201000720. [DOI] [PubMed] [Google Scholar]
- 11.Singh N, Zaidi D, Shyam H, Sharma R, Balapure AK. Polyphenols sensitization potentiates susceptibility of MCF-7 and MDA MB-231 cells to Centchroman. PLoS ONE. 2012;7:e37736. doi: 10.1371/journal.pone.0037736. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Singh MM. Centchroman, a selective estrogen receptor modulator, as a contraceptive and for the management of hormone-related clinical disorders. Med. Res. Rev. 2001;21:302–347. doi: 10.1002/med.1011. [DOI] [PubMed] [Google Scholar]
- 13.Katz EJ, Andrews PA, Howell SB. The effect of DNA polymerase inhibitors on the cytotoxicity of cisplatin in human ovarian carcinoma cells. Cancer commun. 1990;2:159–164. doi: 10.3727/095535490820874515. [DOI] [PubMed] [Google Scholar]
- 14.MY, Ji X, Liu Y, Jin T, Song P. J. Chinese Pharm. Sci. 1998;7:69–71. [Google Scholar]
- 15.Maher DM, Bell MC, O'Donnell EA, Gupta BK, Jaggi M, Chauhan SC. Curcumin suppresses human papillomavirus oncoproteins, restores p53, Rb, and PTPN13 proteins and inhibits benzo[a]pyrene-induced upregulation of HPV E7. Mol. Carcinog. 2011;50:47–57. doi: 10.1002/mc.20695. [DOI] [PubMed] [Google Scholar]
- 16.Chauhan SC, Ebeling MC, Maher DM, Koch MD, Watanabe A, Aburatani H, Lio Y, Jaggi M. MUC13 mucin augments pancreatic tumorigenesis. Mol. Cancer Ther. 2012;11:24–33. doi: 10.1158/1535-7163.MCT-11-0598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chauhan SC, Vannatta K, Ebeling MC, Vinayek N, Watanabe A, Pandey KK, Bell MC, Koch MD, Aburatani H, Lio Y, Jaggi M. Expression and functions of transmembrane mucin MUC13 in ovarian cancer. Cancer Res. 2009;69:765–774. doi: 10.1158/0008-5472.CAN-08-0587. [DOI] [PubMed] [Google Scholar]
- 18.Liu JR, Fletcher B, Page C, Hu C, Nunez G, Baker V. Bcl-xL is expressed in ovarian carcinoma and modulates chemotherapy-induced apoptosis. Gynecol. Oncol. 1998;70:398–403. doi: 10.1006/gyno.1998.5125. [DOI] [PubMed] [Google Scholar]
- 19.Schniewind B, Christgen M, Kurdow R, Haye S, Kremer B, Kalthoff H, Ungefroren H. Resistance of pancreatic cancer to gemcitabine treatment is dependent on mitochondria-mediated apoptosis. Int. J. Cancer. 2004;109:182–188. doi: 10.1002/ijc.11679. [DOI] [PubMed] [Google Scholar]
- 20.Minn AJ, Rudin CM, Boise LH, Thompson CB. Expression of bcl-xL can confer a multidrug resistance phenotype. Blood. 1995;86:1903–1910. [PubMed] [Google Scholar]
- 21.Coleman RL, Monk BJ, Sood AK, Herzog TJ. Latest research and treatment of advanced-stage epithelial ovarian cancer. Nat. Rev. Clin. oncol. 2013 doi: 10.1038/nrclinonc.2013.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lal J. Clinical pharmacokinetics and interaction of centchroman--a mini review. Contraception. 2010;81:275–280. doi: 10.1016/j.contraception.2009.11.007. [DOI] [PubMed] [Google Scholar]
- 23.Galluzzi L, Morselli E, Kepp O, Vitale I, Rigoni A, Vacchelli E, Michaud M, Zischka H, Castedo M, Kroemer G. Mitochondrial gateways to cancer. Mol. Asp. Med. 2010;31:1–20. doi: 10.1016/j.mam.2009.08.002. [DOI] [PubMed] [Google Scholar]
- 24.Hockenbery DM. Targeting mitochondria for cancer therapy. Environ. Mol. Mutagen. 2010;51:476–489. doi: 10.1002/em.20552. [DOI] [PubMed] [Google Scholar]
- 25.Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell. 2011;144:646–674. doi: 10.1016/j.cell.2011.02.013. [DOI] [PubMed] [Google Scholar]
- 26.Sherr CJ. The Pezcoller lecture: cancer cell cycles revisited. Cancer Res. 2000;60:3689–3695. [PubMed] [Google Scholar]
- 27.Matsushime H, Roussel MF, Ashmun RA, Sherr CJ. Colony-stimulating factor 1 regulates novel cyclins during the G1 phase of the cell cycle. Cell. 1991;65:701–713. doi: 10.1016/0092-8674(91)90101-4. [DOI] [PubMed] [Google Scholar]
- 28.Fraser M, Bai T, Tsang BK. Akt promotes cisplatin resistance in human ovarian cancer cells through inhibition of p53 phosphorylation and nuclear function. Int. J. Cancer. 2008;122:534–546. doi: 10.1002/ijc.23086. [DOI] [PubMed] [Google Scholar]
- 29.Abedini MR, Muller EJ, Bergeron R, Gray DA, Tsang BK. Akt promotes chemoresistance in human ovarian cancer cells by modulating cisplatin-induced, p53-dependent ubiquitination of FLICE-like inhibitory protein. Oncogene. 2010;29:11–25. doi: 10.1038/onc.2009.300. [DOI] [PubMed] [Google Scholar]
- 30.Brognard J, Clark AS, Ni Y, Dennis PA. Akt/protein kinase B is constitutively active in non-small cell lung cancer cells and promotes cellular survival and resistance to chemotherapy and radiation. Cancer Res. 2001;61:3986–3997. [PubMed] [Google Scholar]
- 31.West KA, Sianna Castillo S, Dennis PA. Activation of the PI3K/Akt pathway and chemotherapeutic resistance. Drug Resist. Updat. 2002;5:234–248. doi: 10.1016/s1368-7646(02)00120-6. [DOI] [PubMed] [Google Scholar]
- 32.Righetti SC, Della Torre G, Pilotti S, Menard S, Ottone F, Colnaghi MI, Pierotti MA, Lavarino C, Cornarotti M, Oriana S, Bohm S, Bresciani GL, Spatti G, Zunino F. A comparative study of p53 gene mutations, protein accumulation, and response to cisplatin-based chemotherapy in advanced ovarian carcinoma. Cancer Res. 1996;56:689–693. [PubMed] [Google Scholar]
- 33.Lal J, Asthana OP, Nityanand S, Gupta RC. Pharmacokinetics of centchroman in healthy female subjects after oral administration. Contraception. 1995;52:297–300. doi: 10.1016/0010-7824(95)00213-t. [DOI] [PubMed] [Google Scholar]
- 34.Paliwal JK, Gupta RC. Tissue distribution and pharmacokinetics of centchroman. A new nonsteroidal postcoital contraceptive agent and its 7-desmethyl metabolite in female rats after a single oral dose. Drug Metabol. Dispos. 1996;24:148–155. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.