Abstract
Objective:
We investigated the association between regional white and gray matter volume loss and performance on executive functions (EFs) in patients with penetrating traumatic brain injury (pTBI).
Methods:
We studied 164 pTBI patients and 43 healthy controls from the Vietnam Head Injury Study. We acquired CT scans for pTBI patients and divided them according to lesion localization (left and right prefrontal cortex [PFC]). We administered EF tests (Verbal Fluency, Trail Making, Twenty Questions) and used voxel-based lesion symptom mapping (VLSM) and group-based correlational and multiple regression analyses to examine the relative influence of gray and white matter lesions on EF recovery.
Results:
The VLSM analysis revealed that white and gray white matter lesions were associated with impaired EFs. In the left PFC lesion group, damage to the PFC gray matter, anterior corona radiata, and superior longitudinal fasciculus (SLF) were most correlated with functional recovery. Verbal Fluency, which involves a broad fronto-temporo-parietal network, was best predicted by SLF lesion volume. Trail Making and Twenty Questions, which is associated with more focal left frontal damage, was better predicted by PFC lesions.
Conclusions:
Our results indicated that white matter volume loss can be a superior predictor of recovery and a crucial factor driving clinical outcome in functions involving a broad network such as Verbal Fluency. White matter damage may place additional burden on recovery by deteriorating signal transmission between cortical areas within a functional network.
Traumatic brain injury (TBI) is a major public health problem worldwide.1 While physical deficits following TBI generally improve over time, cognitive impairments can persist2,3 and cause permanent disability.1,4
Some of the most persistent deficits after TBI involve executive functions (EFs), resulting in impaired mental flexibility and planning.5,6 Extensive functional neuroimaging and lesion-mapping evidence indicates that the prefrontal cortex (PFC) plays a central role in EFs.6–8 EFs also involve interactions between frontal and posterior brain regions.9,10
To date, the majority of lesion-mapping studies have focused on the cortical gray matter correlates of functional impairment, yet white matter involvement has rarely been quantified. Thus, the relative role of white matter compared to gray matter damage in mediating plasticity and recovery of function remains unclear. The development of stereotaxic white matter atlases based on high-resolution DTI data11 enables quantification of white matter tract damage in TBI patients.12
White matter integrity after brain damage may have an important influence on recovery. Deep lesions that reached white matter were associated with worse language outcome years after penetrating TBI (pTBI).13 White matter tract integrity and volume contralateral to injury can predict aphasia recovery in stroke patients.14 A more precise understanding of the relative role of gray and white matter loss for impairing functional recovery would be useful to clinicians planning and charting therapeutic interventions.
This study aimed to quantitatively examine the relative contributions of gray and white matter damage in predicting functional recovery of EFs in a well-characterized pTBI sample.
METHODS
Participants and behavioral evaluation.
Participants were drawn from Phase 3 of the Vietnam Head Injury Study, which is a longitudinal study of American male combat veterans with focal pTBI and matched combat veterans without brain injury.15 For this study, we selected all right-handed male veterans who participated in Phase 3 (164 pTBI and 43 healthy controls [HC]). For more detailed analyses, 2 subgroups were selected from the pTBI sample: one group with pure left PFC lesions (LPFC, n = 34) and one with pure right PFC lesions (RPFC, n = 46).
Participants underwent 5–7 days of neuropsychological testing between 2003 and 2006 at the National Naval Medical Center in Bethesda, Maryland. We examined performance on 3 tests from the Delis-Kaplan Executive Function System (D-KEFS)16 as EF measures: Verbal Fluency, Trail Making, and Twenty Questions. Details of the D-KEFS tests and other control neuropsychological tests can be found in e-Methods on the Neurology® Web site at Neurology.org (tables 1 and 2).
Table 1.
Demographics, neuropsychological control measures, and EF tests (mean ± SD) for pTBI patients and HC
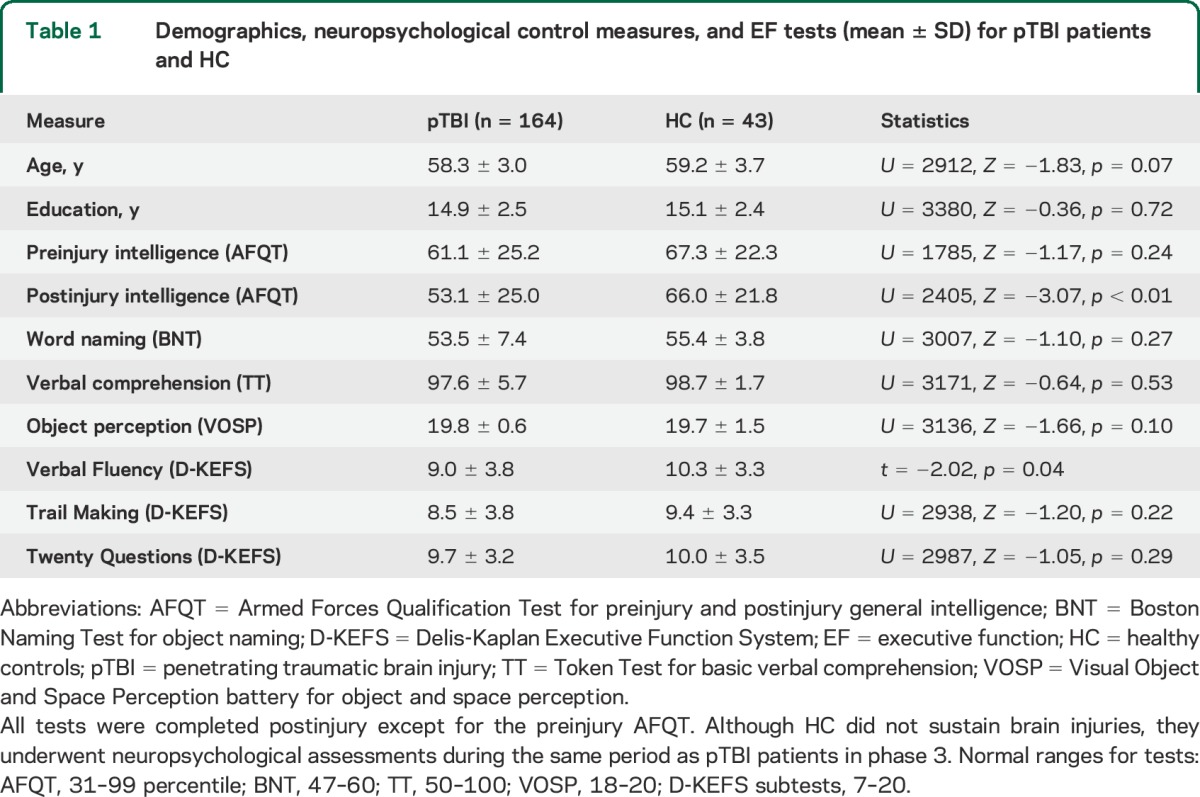
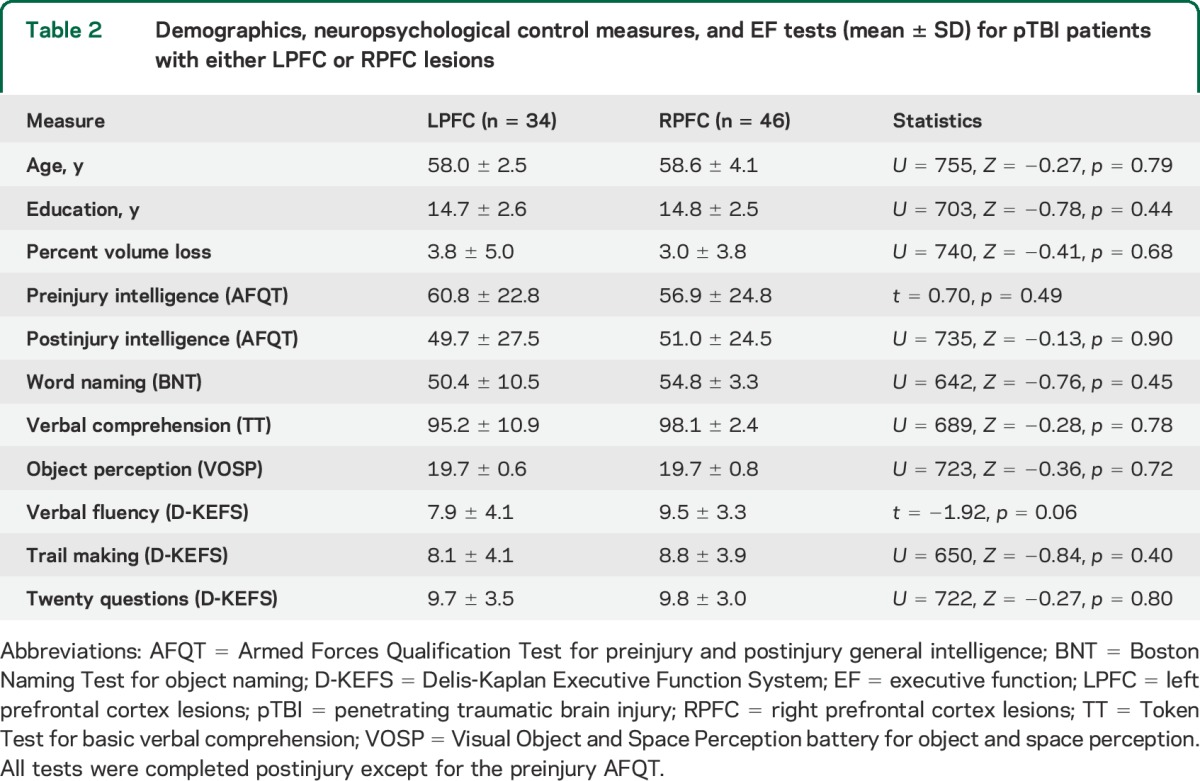
Table 2.
Demographics, neuropsychological control measures, and EF tests (mean ± SD) for pTBI patients with either LPFC or RPFC lesions
Standard protocol approvals, registrations, and patient consents.
Written informed consent was obtained from all participants. The National Naval Medical Center and the National Institute of Health institutional review board approved all the study procedures.
CT scan acquisition and lesion mapping.
Axial CT scans were acquired as described in e-Methods. Since metal was retained in the brain due to penetrating wounds or surgical materials, MRI scans could not be acquired. Lesion density overlap maps were created for all pTBI patients and selected LPFC and RPFC groups. Voxel-based lesion system mapping (VLSM) was applied to the whole brain for each EF measure as described in e-Methods. The anatomic location of significant clusters was identified using the automated anatomical labeling atlas for gray matter17 and the ICBM-DTI-81 atlas for white matter.11 We used percentage of volume loss in selected regions of interest (e-Methods) to establish the extent of damage used in subsequent analyses.
Statistical analysis.
Behavioral analyses were conducted using IBM (Armonk, NY) SPSS 21.0 with significance levels set to 0.05 (2-tailed) for all analyses. Parametric tests (independent samples t test and one-way analysis of variance [ANOVA]) were performed on normally distributed data, and nonparametric tests (Mann-Whitney U tests and Kruskal-Wallis test) on non-normally distributed data. Bivariate Spearman correlations were calculated between test scores and lesion size in gray matter (PFC) and white matter (anterior corona radiata [ACR] or superior longitudinal fasciculus [SLF]) for all pTBI patients, and LPFC and RPFC subgroups. We only conducted analysis on white matter tracts that are involved in EF, had sufficient statistical power given our sample size, and had sufficient variation in distribution of lesion size within our sample.
Using the group median score for each test, LPFC and RPFC participants were split into higher and lower performing subgroups. We constructed lesion maps for each subgroup for each test and compared differences in lesion volume in white matter (ACR, SLF) or gray matter (PFC, parietal or temporal lobes) structures between each pair of subgroups.
In the LPFC group, which was most impaired in EF, we performed multiple linear regressions for each EF test after adjusting for preinjury intelligence (AFQT), entering gray matter (left prefrontal, parietal, or temporal cortex) and white matter tract (left ACR [LACR] or left SLF [LSLF]) lesion volumes as regressors.
RESULTS
Behavioral results.
The pTBI and HC groups did not differ on demographic or most of the neuropsychological measures (table 1). The pTBI group had a lower postinjury intelligence score than HC, although their scores were within the normal range. The LPFC and RPFC subgroups did not differ on demographic and neuropsychological measures (table 2).
The pTBI and HC groups differed on Verbal Fluency but not on Trail Making or Twenty Questions (table 1). One-way ANOVA between LPFC, RPFC, and HC groups revealed a between-group effect (F2,119 = 4.65, p = 0.015) on Verbal Fluency. Post hoc comparisons with Bonferroni correction indicated that the LPFC group had lower scores than the HC group while the RPFC group did not perform differently from the other groups. There were no differences in performance among the 3 groups on Trail Making (p = 0.39) or Twenty Questions (p = 0.79) (table 2). The 3 groups (LPFC, RPFC, and HC) did not differ in preinjury intelligence scores (χ2 = 3.40, p = 0.18).
VLSM results.
Lesion overlap maps are shown in figure e-1. In the pTBI group, the maximum overlap within 29 patients occurred in frontotemporal regions and underlying white matter tracts.
VLSM results for the 3 EF tests are shown in figure 1. Lesions within a left fronto-temporo-parietal network impaired Verbal Fluency test scores. This network involved gray matter structures such as the left inferior frontal gyrus (IFG), middle frontal gyrus (MFG), superior frontal gyrus (SFG), middle temporal gyrus, superior temporal gyrus, inferior parietal gyrus (IPG), and superior parietal gyrus (SPG), as well as underlying white matter tracts including the SLF, ACR, and superior corona radiata (SCR).
Figure 1. Voxel-based lesion system mapping results for the executive function tests.
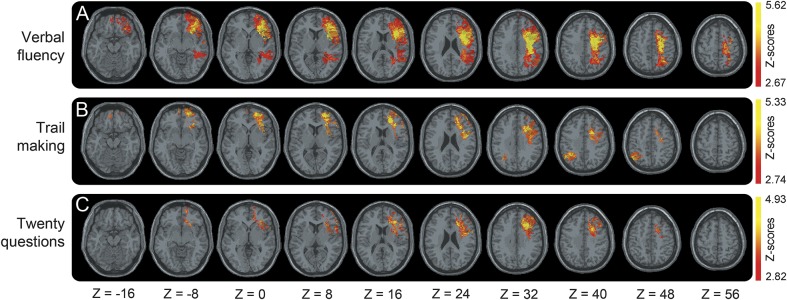
(A) Verbal Fluency, (B) Trail Making, and (C) Twenty Questions. For each test, voxel-based lesion system mapping compares voxel by voxel the performance of penetrating traumatic brain injury with a lesion against those without a lesion in a given voxel. Colored areas indicate a significant association between the presence of a lesion in that location and lower score. The results are overlaid onto an MRI template brain in Montreal Neurological Institute space for visualization purposes. Images are shown in radiologic convention: the right hemisphere is shown on the reader's left side. Color bar indicates Z scores.
Lesions within a fronto-parietal network were found to impair Trail Making test scores. This network included gray matter structures such as the left IFG, MFG, SFG, right IPG, SPG, and white matter structures such as the LSLF, ACR, and SCR.
Lesions within a left frontal network were found to impair Twenty Questions test scores. This network comprised the left IFG, MFG, and SFG, as well as white matter tracts including the LSLF, ACR, and SCR. For a complete list of brain regions and Montreal Neurological Institute coordinates, see table e-1.
Group analysis.
Lesion overlap figures showed that the LPFC and RPFC groups had a maximum overlap of 15 and 16 patients, respectively (figure e-1).
In the LPFC patients, the lower performing subgroup on all 3 tests had more extensive lesions that extended more dorsally compared to the higher performing subgroups (figure 2).
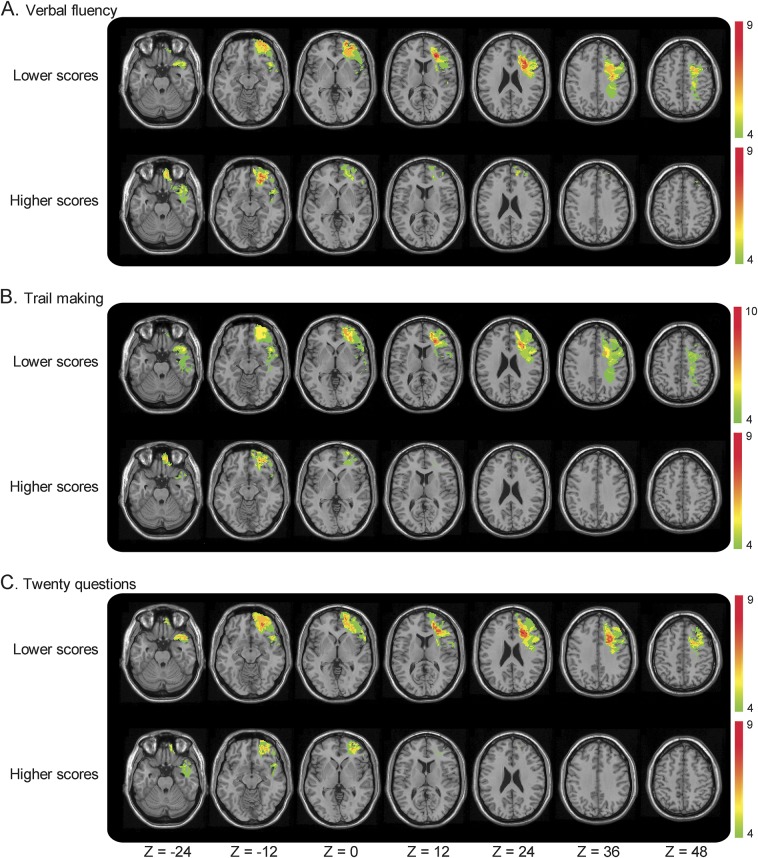
Figure 2. Median split subgroup lesion overlaps in left prefrontal cortex lesions group according to executive function scores.
The left prefrontal cortex lesions group subdivided according to their test score on the (A) Verbal Fluency, (B) Trail Making, and (C) Twenty Questions test. The lower performing penetrating traumatic brain injury group had wider network of gray and white matter damage compared to higher performing individuals. Color denotes the number of patients with overlapping lesion at each voxel. Red indicates the highest density of lesion overlap subjects and blue indicates the least density of lesion overlap. The results are overlaid onto an MRI template brain in Montreal Neurological Institute space for visualization purposes. Images are shown in radiologic convention: the right hemisphere is shown on the reader's left side.
White matter involvement was more pronounced in the lower performing subgroups. Moreover, the largest amount of lesion overlap corresponded to frontal white matter. For each EF test, the percentage of lesion overlap located in white matter was higher in the lower performing subgroup compared to the higher performing subgroup (Verbal Fluency: lower group: 11.25%, higher group: 0.57%; Trail Making: lower group: 7%, higher group: 2.14%; Twenty Questions: lower group: 6.47%, higher group: 1.31%).
For the Verbal Fluency test, the lower performing subgroup had greater LSLF damage than the higher performing subgroup (U = 55, Z = −3.61, p < 0.01). Similarly, for the Trail Making test, we found larger lesions to the left SLF in the lower performing subgroup (U = 85, Z = −2.40, p = 0.04). For the Twenty Questions test, the lower performing subgroup tended to have larger left PFC lesions compared to the higher performing subgroup (U = 90, Z = −1.88, p = 0.06). A similar median split analysis performed for the RPFC group revealed no differences (figures 1 and 2).
Correlation analysis.
Correlations in all pTBI patients yielded modest results between EF scores and lesion size in left PFC and white matter tracts (table e-2). Since PFC is the main cortical region associated with EF performance, we performed detailed analyses on patients with PFC damage to further investigate gray and white matter contributions to recovery of function. We found moderate to large correlations between lesion extent in certain structures and task performance for the LPFC group, but not for the RPFC group (table e-2). For the LPFC group, we found a large correlation between Verbal Fluency and LSLF lesion (rs = −0.698, p < 0.01); a moderate correlation between Trail Making and LSLF lesion (rs = −0.405, p < 0.05); and a moderate correlation between Twenty Questions and lesion size in LPFC (rs = −0.425, p < 0.05), LACR (rs = −0.441, p < 0.01), and LSLF (rs = −0.433, p < 0.05). A Fisher Z test showed that the Verbal Fluency–LSLF correlation was higher than the Verbal Fluency–LPFC (Z = 3.04, p < 0.01) and Verbal Fluency–LACR (Z = 2.28, p < 0.05) correlations.
Multiple regression analysis.
For the Verbal Fluency test, LSLF lesion predicts test scores (β = −0.88, p < 0.02, R2 change = 0.12) after controlling for preinjury intelligence, LACR, and gray matter lesions. For the Trail Making test, LPFC lesion marginally predicted test scores (β = −0.41, p = 0.076, R2 change = 0.06) after controlling for preinjury intelligence, white matter, and other gray matter lesions. For the Twenty Questions test, LPFC lesion marginally predicted test scores (β = −0.38, p = 0.055, R2 change = 0.05) after controlling for preinjury intelligence, white matter, and other gray matter lesions.
DISCUSSION
EF impairments after TBI can become a large source of burden for caregivers. Therefore, understanding the factors crucial to brain plasticity and recovery has important clinical implications. Here, we investigated the relative contribution of white and gray matter brain damage in mediating recovery in focal pTBI patients, using 3 standard tests of EF as outcome measures: Verbal Fluency, Trail Making, and Twenty Questions.
Neuroplastic changes in the brain can facilitate some restoration of function immediately after damage, and long-term outcomes can reflect the extent of recovery. In this study, we used long-term outcomes as a measure of recovery after TBI. Our results show that the relative contribution of white and gray matter depends on the property of the EF tasks. Verbal Fluency performance recruits a large left fronto-temporo-parietal network. Converging evidence from VLSM, group, and regression analyses demonstrated that Verbal Fluency performance can be predicted by the degree of white matter damage to the LSLF, with poorer performance associated with larger lesions. In fact, LSLF lesion volume predicted functional outcome better than gray matter lesion.
Deficits in the Trail Making test are associated with lesions to a network involving left frontal and right parietal regions, and thus the precise contribution of white matter vs gray matter in functional outcomes is more complicated, probably due to the multiple cognitive processes recruited by this task. White matter tracts (LSLF) and gray matter regions (LPFC) were associated with Trail Making performance and LPFC trends to be a better predictor of recovery.
Finally, the Twenty Questions test showed more localized left frontal involvement. While underlying white matter tracts, including LACR and LSLF, were also associated with poorer task performance, white matter damage played a smaller role in predicting functional recovery compared to LPFC gray matter.
In our study, 2 white matter tracts, LSLF and LACR, were associated with impaired EF. The SLF is a major association fiber tract that connects the frontal lobe to temporo-parietal regions,18 and is involved in visuospatial attention,19 working memory,20 and language articulation.21 The ACR is a projection fiber tract that connects frontal and subcortical regions, and has been associated with executive attention.22 The different structural connections and functional relevance of these tracts likely underlie some of the different subprocesses involved in the EF tasks that impact functional outcome when damaged.
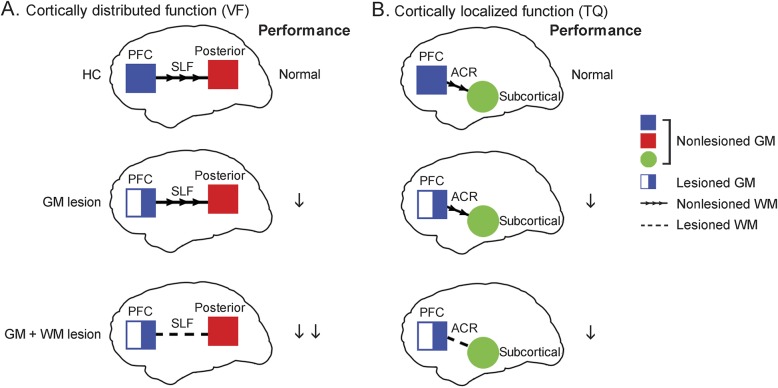
Our results shed light on the relative contributions of gray and white matter damage in EF recovery depending on the network of brain regions recruited (figure 3).
Figure 3. Summary of results.
Summary of executive function results that involve (A) cortically distributed network (Verbal Fluency [VF]) and (B) cortically localized regions (Twenty Questions [TQ]). The 2 tests that yielded the clearest results were chosen for illustrative purposes. Top panels: healthy controls (HCs) have no gray matter (GM) or white matter (WM) lesion and normal performance on both tests. Middle panels: penetrating traumatic brain injury (pTBI) with lesions predominantly in the GM, while WM tracts (superior longitudinal fasciculus [SLF] or anterior corona radiata [ACR]) are spared. Their performance on the tests is impaired (single downward arrow). Bottom panels: pTBI with lesions in both GM and WM. The combination of prefrontal cortex (PFC) lesion plus SLF lesion impairs performance on the Verbal Fluency test (double downward arrows), which requires a broad network of brain regions. The combination of PFC lesion plus ACR lesion did not markedly impact performance on the Twenty Questions test.
In addition to critical involvement of the frontal lobes, a flexible network that includes posterior regions also contributes to successful EF performance.23 EF encompasses a variety of complex processes, and the make-up of the network recruited depends on the various demands in different EF tasks, giving rise to varying recovery mechanisms. White matter contribution appears to be crucial in tasks that involve a large network of cortical regions, such as the PFC and temporoparietal network. We propose that when both cortical gray matter (PFC) and associated white matter (SLF) are damaged, performance on the task will inevitably be impaired. When only PFC is damaged, recovery mechanisms can still occur, possibly through compensatory processes dependent on preserved white matter tracts that connect spared PFC regions to posterior regions. However, when deeper white matter tracts are damaged, functional recovery is limited and EF task performance is more severely impaired.
Since white matter damage reduces connections between the cortical areas in the implicated network, it places additional burden on EF recovery. Adjacent cortical tissue spared from the lesion may be restricted in its ability to receive and respond to signals from other brain regions, thus limiting compensatory functioning in other sectors within the network. In this case, the contribution of white matter damage is more important in functional recovery than gray matter lesions. On the other hand, in the case of more localized functions, the contribution of white matter damage appears to be less relevant compared to gray matter, since recovery and plasticity processes can occur within the PFC and are less dependent on a widespread neural network.
Our study is consistent with previous findings examining diffuse low-grade glioma, a rare tumor condition that evolves along associative white matter tracts, where white matter tract damage was shown to play a critical role in language impairments.24
Recovery after TBI can be varied due to the range of genetic, molecular, psychological, and environmental factors that influence functional outcomes. One such factor is individual genetic variation, which can predispose TBI patients to different functional outcomes.25,26 For example, brain-derived neurotrophic factor, a neurotrophic factor important to synaptic plasticity,27 is involved in the recovery of EF after pTBI.28 Another predictor of functional recovery is preinjury cognitive ability.15 People with more cognitive reserve are less susceptible to adverse outcome of brain damage, due to either preexisting neuroprotective processes or postinjury cognitive compensatory mechanisms.29,30
Besides individual variability, environmental influences also play an important role in recovery of function. Support from family members and significant others help facilitate rehabilitation,31,32 while a stressful family setting may deter recovery.33 Cognitive training intervention can also help improve function34,35 by affecting brain plasticity.36 It would be useful to understand how these other factors interact with the extent of white matter damage in mediating neuroplasticity and recovery of EF after TBI in order to devise better individual-oriented rehabilitation plans.
A limitation of our study is that the white matter atlas used identifies deep white matter tracts, but not different layers within the tracts as can be identified using more refined white matter atlases.37 Other limitations include the use of an all-male sample and the requirement that only CT scans could be used given the retained metal within the brain as a result of the penetrating brain wounds or surgical materials.
Another promising avenue for future research in white matter involvement in functional recovery is the influence of genetic variation. Interestingly, the Val variant of the BDNF gene is associated with reduced white matter integrity in healthy participants,38 while Met allele carriers demonstrate increased structural connectivity,39 despite the greater association of the Met allele with cognitive impairment and clinical pathology.40 It would be interesting to study whether individual genetic variation mediates the contribution of white matter structural integrity in functional outcome after brain injury.
Our results have clinical implications for minimizing the undesirable effects of penetrating brain wounds. A lesion that penetrates deeper and occludes more white matter may have more serious functional consequences than a more superficial lesion comparable in size. This emphasizes the importance of producing helmets that can effectively reduce shrapnel velocity, thus reducing the depth of penetration into the brain. However, helmets do not protect from blast injury, which can affect both gray and white matter; thus, improved forms of wearable shielding would be necessary to reduce the impact of a strong blast on the brain. Our results also suggest that neurosurgeons should be cautious about removing white matter.
Results from the present study indicate an important role of white matter volume loss in predicting recovery of EF after TBI. Particularly in cognitive functions that involve a broadly distributed network of brain regions such as verbal fluency, white matter lesion volume can predict functional outcome above and beyond contributions from gray matter damage. White matter damage may place additional burden on EF recovery by altering and deteriorating signal transmission between cortical areas within a functional network. Thus, white matter lesion volume can be considered as a crucial factor driving clinical outcome in addition to other neural and psychosocial factors.
Supplementary Material
ACKNOWLEDGMENT
The authors thank the Vietnam veterans for their participation in the study; A. Salazar, V. Raymont, S. Bonifant, B. Cheon, C. Ngo, A. Greathouse, K. Reding, and G. Tasick for testing and evaluating participants; and the National Naval Medical Center and the National Institute of Neurological Disorders and Stroke for providing their facilities and supporting the research.
GLOSSARY
- ACR
anterior corona radiata
- ANOVA
analysis of variance
- D-KEFS
Delis-Kaplan Executive Function System
- EF
executive function
- HC
healthy control
- IFG
inferior frontal gyrus
- IPG
inferior parietal gyrus
- LACR
left anterior corona radiata
- LPFC
left prefrontal cortex lesions
- LSLF
left superior longitudinal fasciculus
- MFG
middle frontal gyrus
- PFC
prefrontal cortex
- pTBI
penetrating traumatic brain injury
- RPFC
right prefrontal cortex lesions
- SCR
superior corona radiata
- SFG
superior frontal gyrus
- SLF
superior longitudinal fasciculus
- SPG
superior parietal gyrus
- TBI
traumatic brain injury
- VLSM
voxel-based lesion system mapping
Footnotes
Editorial, page 1390
Supplemental data at Neurology.org
AUTHOR CONTRIBUTIONS
All authors contributed to the manuscript. I.C., W.Z., F.K., and J.G. designed and performed research. I.C. and W.Z. analyzed the data. I.C., W.Z., and J.G. interpreted the data. I.C., W.Z., A.C., F.K., J.S., and J.G. wrote the paper. F.K. and J.G. acquired the data. J.S. implemented analysis algorithms. J.G. supervised the study. All authors read and approved the final version of the manuscript.
STUDY FUNDING
No targeted funding reported.
DISCLOSURE
I. Cristofori receives support from the US Department of Education (M. Switzer Research Fellowship). W. Zhong, A. Chau, J. Solomon, and F. Krueger report no disclosures relevant to the manuscript. J. Grafman serves as coeditor of Cortex and receives research support from the Coleman Foundation and the Frankel Foundation. Go to Neurology.org for full disclosures.
REFERENCES
- 1.CDC. Injury Fact Book. Atlanta: Centers for Disease Control and Prevention; 2006. [Google Scholar]
- 2.Brooks DN, Hosie J, Bond MR, Jennett B, Aughton M. Cognitive sequelae of severe head injury in relation to the Glasgow Outcome Scale. J Neurol Neurosurg Psychiatry 1986;49:549–553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Whitnall L, McMillan TM, Murray GD, Teasdale GM. Disability in young people and adults after head injury: 5-7 year follow up of a prospective cohort study. J Neurol Neurosurg Psychiatry 2006;77:640–645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Draper K, Ponsford J. Cognitive functioning ten years following traumatic brain injury and rehabilitation. Neuropsychology 2008;22:618–625. [DOI] [PubMed] [Google Scholar]
- 5.Levin H, Kraus MF. The frontal lobes and traumatic brain injury. J Neuropsychiatry Clin Neurosci 1994;6:443–454. [DOI] [PubMed] [Google Scholar]
- 6.Miller EK. The prefrontal cortex and cognitive control. Nat Rev Neurosci 2000;1:59–65. [DOI] [PubMed] [Google Scholar]
- 7.Levin HS, Eisenberg HM, Benton AL. Frontal Lobe Function and Dysfunction. New York: Oxford University Press; 1991. [Google Scholar]
- 8.Stuss DT, Benson DF. The Frontal Lobes. New York: Raven Press; 1986. [Google Scholar]
- 9.Barbey AK, Colom R, Solomon J, Krueger F, Forbes C, Grafman J. An integrative architecture for general intelligence and executive function revealed by lesion mapping. Brain 2012;135:1154–1164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Koenigs M, Barbey AK, Postle BR, Grafman J. Superior parietal cortex is critical for the manipulation of information in working memory. J Neurosci 2009;29:14980–14986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mori S, Oishi K, Jiang H, et al. Stereotaxic white matter atlas based on diffusion tensor imaging in an ICBM template. Neuroimage 2008;40:570–582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rudrauf D, Mehta S, Grabowski TJ. Disconnection's renaissance takes shape: formal incorporation in group-level lesion studies. Cortex 2008;44:1084–1096. [DOI] [PubMed] [Google Scholar]
- 13.Ludlow CL, Rosenberg J, Fair C, Buck D, Schesselman S, Salazar A. Brain lesions associated with nonfluent aphasia 15 years following penetrating head injury. Brain 1986;109:55–80. [DOI] [PubMed] [Google Scholar]
- 14.Forkel SJ, Thiebaut de Schotten M, Dell'Acqua F, et al. Anatomical predictors of aphasia recovery: a tractography study of bilateral perisylvian language networks. Brain 2014;137:2027–2039. [DOI] [PubMed] [Google Scholar]
- 15.Raymont V, Greathouse A, Reding K, Lipsky R, Salazar A, Grafman J. Demographic, structural and genetic predictors of late cognitive decline after penetrating head injury. Brain 2008;131:543–558. [DOI] [PubMed] [Google Scholar]
- 16.Delis DC, Kaplan E, Kramer JH. Delis-Kaplan Executive Function System. San Antonio, TX: The Psychological Corporation; 2001. [Google Scholar]
- 17.Tzourio-Mazoyer N, Landeau B, Papathanassiou D, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage 2002;15:273–289. [DOI] [PubMed] [Google Scholar]
- 18.Makris N, Kennedy DN, McInerney S, et al. Segmentation of subcomponents within the superior longitudinal fascicle in humans: a quantitative, in vivo, DT-MRI study. Cereb Cortex 2005;15:854–869. [DOI] [PubMed] [Google Scholar]
- 19.Thiebaut de Schotten M, Dell'Acqua F, Forkel SJ, et al. A lateralized brain network for visuospatial attention. Nat Neurosci 2011;14:1245–1246. [DOI] [PubMed] [Google Scholar]
- 20.Petrides M, Pandya DN. Association pathways of the prefrontal cortex and functional observations. In: Struss DT, Knight RT, eds. Principles of Frontal Lobe Function. Oxford: Oxford University Press; 2002:31–50. [Google Scholar]
- 21.Bernal B, Ardila A. The role of the arcuate fasciculus in conduction aphasia. Brain 2009;132:2309–2316. [DOI] [PubMed] [Google Scholar]
- 22.Yin X, Han Y, Ge H, et al. Inferior frontal white matter asymmetry correlates with executive control of attention. Hum Brain Mapp 2013;34:796–813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Elliott R. Executive functions and their disorders. Br Med Bull 2003;65:49–59. [DOI] [PubMed] [Google Scholar]
- 24.Almairac F, Herbet G, Moritz-Gasser S, de Champfleur NM, Duffau H. The left inferior fronto-occipital fasciculus subserves language semantics: a multilevel lesion study. Brain Struct Funct Epub 2014 Apr 18. [DOI] [PubMed]
- 25.McAllister TW. Genetic factors modulating outcome after neurotrauma. PM R 2010;2:S241–S252. [DOI] [PubMed] [Google Scholar]
- 26.Weaver SM, Portelli JN, Chau A, Cristofori I, Moretti L, Grafman J. Genetic polymorphisms and traumatic brain injury: the contribution of individual differences to recovery. Brain Imaging Behav 2014;8:420–434. [DOI] [PubMed] [Google Scholar]
- 27.Binder DK, Scharfman HE. Brain-derived neurotrophic factor. Growth Factors 2004;22:123–131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Krueger F, Pardini M, Huey ED, et al. The role of the Met66 brain-derived neurotrophic factor allele in the recovery of executive functioning after combat-related traumatic brain injury. J Neurosci 2011;31:598–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stern Y. Cognitive reserve. Neuropsychologia 2009;47:2015–2028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Whalley LJ, Deary IJ, Appleton CL, Starr JM. Cognitive reserve and the neurobiology of cognitive aging. Ageing Res Rev 2004;3:369–382. [DOI] [PubMed] [Google Scholar]
- 31.Jacobs HE. The Los Angeles Head Injury Survey: procedures and initial findings. Arch Phys Med Rehabil 1988;69:425–431. [PubMed] [Google Scholar]
- 32.Levack WM, Siegert RJ, Dean SG, McPherson KM. Goal planning for adults with acquired brain injury: how clinicians talk about involving family. Brain Inj 2009;23:192–202. [DOI] [PubMed] [Google Scholar]
- 33.Sander AM, Caroselli JS, High WM, Jr, Becker C, Neese L, Scheibel R. Relationship of family functioning to progress in a post-acute rehabilitation programme following traumatic brain injury. Brain Inj 2002;16:649–657. [DOI] [PubMed] [Google Scholar]
- 34.Constantinidou F, Thomas RD, Robinson L. Benefits of categorization training in patients with traumatic brain injury during post-acute rehabilitation: additional evidence from a randomized controlled trial. J Head Trauma Rehabil 2008;23:312–328. [DOI] [PubMed] [Google Scholar]
- 35.Wilson BA, Emslie H, Quirk K, Evans J, Watson P. A randomized control trial to evaluate a paging system for people with traumatic brain injury. Brain Inj 2005;19:891–894. [DOI] [PubMed] [Google Scholar]
- 36.Raymont V, Grafman J. Cognitive neural plasticity during learning and recovery from brain damage. Prog Brain Res 2006;157:199–206. [DOI] [PubMed] [Google Scholar]
- 37.Thiebaut de Schotten M, Ffytche DH, Bizzi A, et al. Atlasing location, asymmetry and inter-subject variability of white matter tracts in the human brain with MR diffusion tractography. Neuroimage 2011;54:49–59. [DOI] [PubMed] [Google Scholar]
- 38.Tost H, Alam T, Geramita M, et al. Effects of the BDNF Val66Met polymorphism on white matter microstructure in healthy adults. Neuropsychopharmacology 2013;38:525–532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ziegler E, Foret A, Mascetti L, et al. Altered white matter architecture in BDNF met carriers. PLoS One 2013;8:e69290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Bath KG, Lee FS. Variant BDNF (Val66Met) impact on brain structure and function. Cogn Affect Behav Neurosci 2006;6:79–85. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.